��Ŀ����
�ں��º��������£���һ����NO2��N2O4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������з�����Ӧ��N2O4 (g) ![]() 2NO2 (g) ��H �� 0����Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ����ͼ��ʾ��
2NO2 (g) ��H �� 0����Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ����ͼ��ʾ��
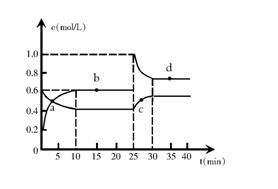
��1�����¶�ʱ���÷�Ӧ��ƽ�ⳣ��Ϊ________________�� ���¶����ߣ�Kֵ��__________�����������С�����䡱����
��2��a��b��c��d�ĸ����У���ѧ��Ӧ����ƽ��״̬����____________�㡣����㿪ʼ�״δﵽƽ��ʱ����NO2��ʾ��ƽ����Ӧ����Ϊ______________________��
��3��25 minʱ��������______mol___________�������ʵĻ�ѧʽ��ʹƽ�ⷢ�����ƶ���
��4��d���ӦNO2�����ʵ���Ũ��_________���>������<����=����0.8mol??L��1��������_______________________________________________________________________��
��1��0.9 mol��L��1 ��2�֣� ���� ��1�֣�
��2��b d ��2�֣� 0.04 mol��L��1��min��1 ��2�֣�
��3��0.8 NO2 ��2�֣�
��4��< ��2�֣� NO2�����ʵ���Ũ��Ϊ0.8mol??L��1ʱ��Q =1.28 mol��L��1������K����ӦӦ����������У���һ������NO2�����ʵ���Ũ���� ��2�֣�
����:
��

 ��ѧ�̸̳����¿α�ϵ�д�
��ѧ�̸̳����¿α�ϵ�д� �ں��º��������£���һ����NO2��N2O4�Ļ������ͨ���ܱ������У�������Ӧ����Ӧ�и����Ũ����ʱ��仯��ϵ��ͼ������˵���У���ȷ���ǣ�������
�ں��º��������£���һ����NO2��N2O4�Ļ������ͨ���ܱ������У�������Ӧ����Ӧ�и����Ũ����ʱ��仯��ϵ��ͼ������˵���У���ȷ���ǣ�������| A��a��b��c��d�ĸ����У�ֻ��b��Ļ�ѧ��Ӧ����ƽ��״̬ | B��ǰ10min���æԣ�NO2����ʾ�Ļ�ѧ��Ӧ����Ϊ0.04 mol/��L?min�� | C��25minʱ������ƽ���ƶ���ԭ���������¶� | D��a�㣬�æԣ�NO2����ʾ�Ļ�ѧ��Ӧ���ʵ����æԣ�N2O4����ʾ�Ļ�ѧ��Ӧ���� |


 HCO3-+OH-
HCO3-+OH- C2D4��g������H��0���ں��º��������£���һ����CD2��C2D4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������У���Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ��ͼ��ʾ��
C2D4��g������H��0���ں��º��������£���һ����CD2��C2D4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������У���Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ��ͼ��ʾ��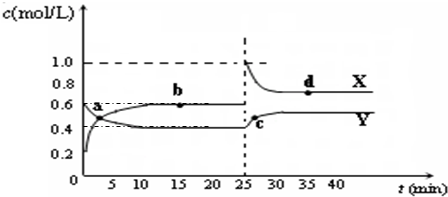
 ��֪��2NO2��g��?N2O4��g����H��O���ں��º��������£���һ����NO2��N2O4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������У���Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ��ͼ��ʾ��
��֪��2NO2��g��?N2O4��g����H��O���ں��º��������£���һ����NO2��N2O4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������У���Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ��ͼ��ʾ��
 A2O4��g������H��0���ں��º��������£���һ����AO2��A2O4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������У���Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ����ͼ��ʾ��
A2O4��g������H��0���ں��º��������£���һ����AO2��A2O4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������У���Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ����ͼ��ʾ��