��Ŀ����
���ݱ�������˵����ȷ���ǣ���
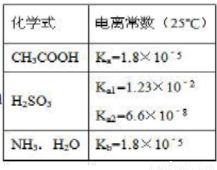
A��25��Cʱ��pH=3�Ĵ����pH=11�İ�ˮ�������Ϻ��Һ��ˮ�ĵ����DZ��ٽ���
B��Na2SO3��Һ�еμ��������ᣬ��Ӧ�����ӷ���ʽΪ��SO3 +2CH3COOH=SO2��+H2O+2CH3COO-
C�� NaHSO3��Һ�����ԣ�����ΪKw/Ka1>Ka2
D��0��1mol/L��CH3COOH��Һ���Ũ�ȵ������CH3COONa ��Ϻ���Һ�е����������¹�ϵ�� c (H+) +c (CH3C OOH) =c (CH3COO-
OOH) =c (CH3COO- ) +2c (OH-)
) +2c (OH-)
��ϰ��ϵ�д�
�����Ŀ
������Ԫ��W��X��Y��Z��ԭ�������������ӡ�m��n��p������ЩԪ����ɵĶ�Ԫ�����W2��X2��Z2�ֱ���Ԫ��W��X��Z�ĵ��ʡ���֪��
I��һ��������ij�ܱ������пɷ�����Ӧ��aX2+bW2 cm����Ӧ���������ʵ�Ũ�ȱ仯���£�
cm����Ӧ���������ʵ�Ũ�ȱ仯���£�
| X2 | W2 | m |
��ʼŨ��/mol��L��1 | 0.4 | 0.4 | 0 |
ƽ��Ũ��/mol��L��1 | 0.3 | 0.1 | 0.2 |
II�����ǿɷ������·�Ӧ��2m(g)+3Z(g)=6n(g)+X2(g)��4n(g)+Y2(g) 2p(l)+2Z2(g)��
2p(l)+2Z2(g)��
����˵����ȷ����
A��ԭ�Ӱ뾶��W��X��Y B��a��b��c=3:1:2
C��X��������һ������ɫ���� D��m��n��p�������ʾ�Ϊ���ۻ�����
9����500mL1.0mol•L-1��CuSO4��Һ�У�����Ϊ100gCuƬ������Ϊ100gZnƬ��ͨ��һ��ʱ�������ȡ���缫�����ZnƬ������Ϊ106.4g����ʱʣ����Һ��CuSO4�����ʵ���Ũ��Ϊ��������
| A�� | 0.25mol•L-1 | B�� | 0.8mol•L-1 | C�� | 1.0mol•L-1 | D�� | 1.2mol•L-1 |
6��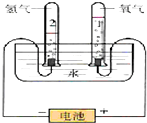 ˮ�����౦�����Ȼ��Դ��Ҳ��һ����Ҫ�Ļ���ԭ�ϣ���ͼ�ǵ��ˮԭ����ʵ��װ��ͼ��
ˮ�����౦�����Ȼ��Դ��Ҳ��һ����Ҫ�Ļ���ԭ�ϣ���ͼ�ǵ��ˮԭ����ʵ��װ��ͼ��
��1��д�����ˮ�Ļ�ѧ����ʽ����Ϊ������ԭ��Ӧ������˫���ű�ʾ�����ӵ�ת�Ʒ������Ŀ��
 ��
��
��2����������3.6gH2O��������±���
��3������47.8gH2O���������м���2.3gNa������ˮ��Ӧ�����ӷ���ʽΪ2Na+2 H2O�T2Na++2OH-+H2��������Ӧ��õ�����Һ��������NaOH�����ʵ���������Ϊ8%����Ҫ�������Һ�����ʵ����ʵ���Ũ�ȣ�����Ҫ��Һ�ܶȣ�������������
��4�����ڵ��ˮ���ռ���O2���Ϊ22.4mL���ڱ�״���£������ռ�����H2������0.004g��
 ˮ�����౦�����Ȼ��Դ��Ҳ��һ����Ҫ�Ļ���ԭ�ϣ���ͼ�ǵ��ˮԭ����ʵ��װ��ͼ��
ˮ�����౦�����Ȼ��Դ��Ҳ��һ����Ҫ�Ļ���ԭ�ϣ���ͼ�ǵ��ˮԭ����ʵ��װ��ͼ����1��д�����ˮ�Ļ�ѧ����ʽ����Ϊ������ԭ��Ӧ������˫���ű�ʾ�����ӵ�ת�Ʒ������Ŀ��
 ��
����2����������3.6gH2O��������±���
| �� �� | ���ʵ��� | O2��H2������ȣ�ͬ��ͬѹ�£� | |
| O2 | 3.2g | 0.1mol | 1��2 |
| H2 | 0.4g | 0.2mol |
��4�����ڵ��ˮ���ռ���O2���Ϊ22.4mL���ڱ�״���£������ռ�����H2������0.004g��
7����ѧ�������һ�����ʡ���ˮ������ÿ��������ˮ95%�������ɳ�ʹ裬��ˮ����CO2����������ͨˮ������������˵����ȷ���ǣ�������
| A�� | ��ˮ�Ǵ����� | B�� | ��ˮ�ɱ���ͬһ������ | ||
| C�� | ��ˮ�е�ˮ���Ӳ����˶� | D�� | �ø�ˮ�ܸ��õ�����CO2 |


 2C(g)+D(g) ��H<0��ƽ���ƶ�ͼ��Ӱ��ƽ���ƶ���ԭ�������
2C(g)+D(g) ��H<0��ƽ���ƶ�ͼ��Ӱ��ƽ���ƶ���ԭ�������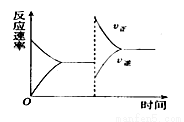
 c����d����
c����d����
 �ԺͲ��ᣨH2C2O4)�����ᷴӦ���ɸ�Ч������ɱ����ClO2��������CO2��KHSO4�����ʡ�д���÷�Ӧ�Ļ�ѧ����ʽ ��
�ԺͲ��ᣨH2C2O4)�����ᷴӦ���ɸ�Ч������ɱ����ClO2��������CO2��KHSO4�����ʡ�д���÷�Ӧ�Ļ�ѧ����ʽ �� �����ܴﵽʵ��Ŀ�ĵ��ǣ���
�����ܴﵽʵ��Ŀ�ĵ��ǣ���
 ��ͬ�����£���һ֧�Թ��м���2ml5%H2O2��1mlH2O������һ֧�Թ��м���2mL5%H2O2��1mLFeCl3��Һ���۲첢�Ƚ�ʵ������
��ͬ�����£���һ֧�Թ��м���2ml5%H2O2��1mlH2O������һ֧�Թ��м���2mL5%H2O2��1mLFeCl3��Һ���۲첢�Ƚ�ʵ������