��Ŀ����
13��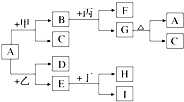 ��ͼ���й������ת���Ĺ�ϵͼ������A�׳����죬��Ϊǿ�ᣬ�ҡ�HΪ��ԭ�����壬��Ϊһ�ֳ����Ļӷ����ᣬGΪ���ɫ������I����ɫ��dz��ɫ��
��ͼ���й������ת���Ĺ�ϵͼ������A�׳����죬��Ϊǿ�ᣬ�ҡ�HΪ��ԭ�����壬��Ϊһ�ֳ����Ļӷ����ᣬGΪ���ɫ������I����ɫ��dz��ɫ����1����F����Na+��SO42-��ɵ���Һ����Ļ�ѧʽ��H2SO4����D����ʹ����ʯ��ˮ����ǵ����壬���ҵĻ�ѧʽΪCO��
��2�����Ļ�ѧ�ɷֿ�����C��
A��Ũ���� B��ϡ���� C������ D��Ũ����
��3������I��Һ�м�������������Һ�����Թ۲쵽�����������ɰ�ɫ������Ѹ�ٱ�Ϊ����ɫ�����ձ�Ϊ���ɫ����Ӧ�Ļ�ѧ����ʽ������Fe2++2OH-=Fe��OH��2����4Fe��OH��2+O2+2H2O=4Fe��OH��3����
��4��д��E��C��Ӧ�ķ���ʽ���õ����ŷ��������ӵ�ת�Ʒ������Ŀ��
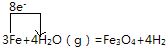 ��
����5����A�л�������Al2O3����ȥ���ʵķ������ȼ��������NaOH��Һ���ٹ��˽��з��루��ʵ�������
���� A�׳����죬��ѧʽΪFe2O3��GΪ���ɫ������ӦΪFe��OH��3��I����ɫ��dz��ɫ������Fe2+���ӣ���F����Na+��SO42-��ɵ���Һ�����Ϊ���ᣬ���ת����ϵ��֪��BΪFe2��SO4��3��CΪˮ��BΪNaOH����D����ʹ����ʯ��ˮ����ǵ����壬��ԭ��������ΪCO��DΪCO2��EΪFe�����Ͷ���Ӧ�������������뻹ԭ������H��˵����Ϊ��������ϡ�ᣬHΪH2����5������Fe2O3�л�������Al2O3�������ù�����NaOH��Һ��ȥ��
��� �⣺A�׳����죬��ѧʽΪFe2O3��GΪ���ɫ������ӦΪFe��OH��3��I����ɫ��dz��ɫ������Fe2+���ӣ���F����Na+��SO42-��ɵ���Һ�����ΪH2SO4�����ת����ϵ��֪��BΪFe2��SO4��3��CΪˮ��BΪNaOH����D����ʹ����ʯ��ˮ����ǵ����壬��ԭ��������ΪCO��DΪCO2��EΪFe�����Ͷ���Ӧ�������������뻹ԭ������H��˵����Ϊ��������ϡ�ᣬHΪH2��
��1����F����Na+��SO42-��ɵ���Һ����Ļ�ѧʽ�ǣ�H2SO4����D����ʹ����ʯ��ˮ����ǵ����壬���ҵĻ�ѧʽΪCO��
�ʴ�Ϊ��H2SO4��CO��
��2��Fe�붡��Ӧ������������������Ũ���ᡢ�����Ϊ�������ᣬ������Ϊ���ᣬ
��ѡ��C��
��3���������Ӻ��������Ʒ�Ӧ���ɰ�ɫ�����������������ڿ����лᱻѸ������Ϊ�����������ɫ�������漰�ķ�Ӧ�����ӷ���ʽΪ��Fe2++2OH-=Fe��OH��2����4Fe��OH��2+O2+2H2O=4Fe��OH��3��
�ʴ�Ϊ�����ɰ�ɫ������Ѹ�ٱ�Ϊ����ɫ�����ձ�Ϊ���ɫ��Fe2++2OH-=Fe��OH��2����4Fe��OH��2+O2+2H2O=4Fe��OH��3��
��4������ˮ��Ӧ�����������������������õ����ŷ��������ӵ�ת�Ʒ������ĿΪ��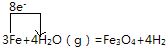 ��
��
�ʴ�Ϊ��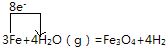 ��
��
��5����Fe2O3�л�������Al2O3�������ù�����NaOH��Һ�ܽ����������ٹ��˳�ȥ����Ӧ���ӷ���ʽΪ��Al2O3+2OH-=2 AlO2-+H2O��
�ʴ�Ϊ��NaOH��Һ�����ˣ�
���� ���⿼�������ƶϣ����ʵ���ɫ������Ϊ�ƶ�ͻ�ƿڣ���Ҫѧ����������Ԫ�ػ��������ʣ��Ƕ�ѧ���ۺ������Ŀ��飬�Ѷ��еȣ�

| A�� | ���� | B�� | �ȶ��� | C�� | ������ | D�� | �ӷ��� |
| A�� | FeO | B�� | Fe3O4 | C�� | Fe2O3 | D�� | CuO |
| A�� | FeS | B�� | Fe2O3 | C�� | FeCl3 | D�� | Fe2��SO��4 |
| A�� | ����ڹ����Ĵ�����ȼ�տ�������SO3 | |
| B�� | ���ñ���ȡ��ˮ�еĵ⣬���ó���©�����з�Һ������ʵ�ַ��� | |
| C�� | ����������������磬������������� | |
| D�� | �����������۷�Ӧ���������� |
| A�� | �Ե��ʳ��ˮΪ������ȡ�����Ȳ�Ʒ�Ĺ�ҵ��Ϊ���ȼҵ�� | |
| B�� | �ö������̺�Ũ������ȡ������ÿ����1 mol�������������Ȼ���Ϊ4 mol | |
| C�� | ����������������Ӧʱ�����Ȼ����� | |
| D�� | ��ҵ����������ʯ��ʯֱ�ӷ�Ӧ����Ư�� |
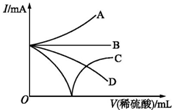
| A�� | A | B�� | B | C�� | C | D�� | D |
����˿��������ȼ��
������������
�ۼ�����������
���������ˮ������Ӧ��
| A�� | �٢� | B�� | �٢� | C�� | �ڢ� | D�� | �ڢ� |