��Ŀ����
�����廯����A��ijЩֲ��ӷ����е���Ҫ�ɷ֣��䱽����ֻ��һ��ȡ������A����Է�������������150����ȫȼ��ֻ����CO2��H2O���ҷ�������Ԫ�ص���������Ϊ12.12%��A������ת����ϵ��?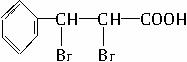
����������Ϣ��գ�?
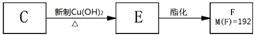
(1)A�ķ���ʽ��____________________��A�й����ŵ�������_________��
(2)C�Ľṹ��ʽ��_______________________��
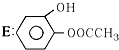
(3)д����Ӧ�ݵĻ�ѧ����ʽ��____________________________________��
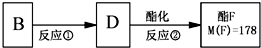
(4)��Ӧ������D��ͬʱ���Ƿ��п������ɱ���л�����У���д����ṹ��ʽ������������������������������������
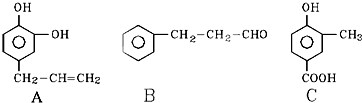
(1)C9H8O��������̼̼˫����ȩ��
(2) 
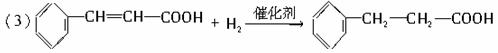
![]()
![]()
������A��������ԭ����ԼΪ![]() ��1?
��1?
��M(A)=![]() =132 g��mol -1
=132 g��mol -1
�������Ϣ��֪��A�б�����ֻ��һ��ȡ����������������Һ��Ӧ˵�������д���ȩ����B�ܷ����ӳɷ�Ӧ������1 mol H2�ӳɣ�˵������![]() �����M=132�����ɵó�A�Ľṹ��ʽΪ
�����M=132�����ɵó�A�Ľṹ��ʽΪ![]() ��
��

��ϰ��ϵ�д�
�����Ŀ




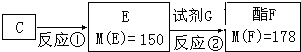

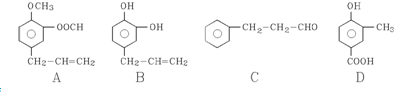
 ��C��
��C��