��Ŀ����
�����ۡ�������˷ɴ�����ɹ����ҹ������������϶̡�����3-4�����й��˵�����������̽�������֣��������д��ڽϷḻ��������Ϊ3�ĺ�����������Ϊ�˾۱����Ҫԭ��֮һ������������ȷ���ǣ�������
| A����������Ϊ3�ĺ�ԭ�ӹ��ɵķ�����˫ԭ�ӷ��� |
| B��23He��24He�����ʾ���ͬ |
| C��23He���������24He����������� |
| D��23He��24He �γɵķ����д��ڹ��ۼ� |
���㣺����
ר�⣺ԭ�������ṹר��
������A��ϡ�������Ϊ��ԭ�ӷ��ӣ�
B����Ϊͬλ�صĺ���֮�仯ѧ���ʼ�����ȫ��ͬ���������ʲ�ͬ��
C��ԭ�ӷ���ZAX�����½�����Ϊ�����������Ͻ�����Ϊ�����������������=������+���������
D��23He��24He �γɵķ��Ӿ�Ϊ��ԭ�ӷ��ӣ�������ѧ����
B����Ϊͬλ�صĺ���֮�仯ѧ���ʼ�����ȫ��ͬ���������ʲ�ͬ��
C��ԭ�ӷ���ZAX�����½�����Ϊ�����������Ͻ�����Ϊ�����������������=������+���������
D��23He��24He �γɵķ��Ӿ�Ϊ��ԭ�ӷ��ӣ�������ѧ����
���
�⣺A��ϡ�������Ϊ��ԭ�ӷ��ӣ���A����
B����Ϊͬλ�صĺ���֮�䣬�����������ͬ����������ͬ����˻�ѧ���ʼ�����ȫ��ͬ���������ʲ�ͬ����B����
C��23He�е�������Ϊ2��24He��������Ϊ4-2=2����C��ȷ��
D��23He��24He �γɵķ��Ӿ�Ϊ��ԭ�ӷ��ӣ�������ѧ������D����
��ѡC��
B����Ϊͬλ�صĺ���֮�䣬�����������ͬ����������ͬ����˻�ѧ���ʼ�����ȫ��ͬ���������ʲ�ͬ����B����
C��23He�е�������Ϊ2��24He��������Ϊ4-2=2����C��ȷ��
D��23He��24He �γɵķ��Ӿ�Ϊ��ԭ�ӷ��ӣ�������ѧ������D����
��ѡC��
���������⿼����صı�ʾ����������������������������֮��Ĺ�ϵ���漰������ϵʽ��Ӧ�ã���������A=������Z+������N�ں��������=������=�˵����=ԭ����������Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
����ͼʾ��ʾ���������ữ����������Һ�еμ�����������Һʱ���ɳ���M������������������Һ���V�Ĺ�ϵ�����п�����ȷ���ǣ�������
A��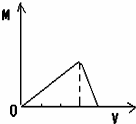 |
B��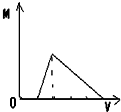 |
C�� |
D��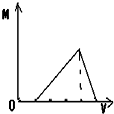 |
���й��ڵ����ʵ������У�����ȷ���ǣ�������
| A��������Һ��ӣ�NH4��2SO4��Һ���ᴿ������ |
| B���ڶ����м���������ʯ�࣬��ʹ��������Ϊ���� |
| C���¶�Խ�ߣ�ø��ijЩ��ѧ��Ӧ�Ĵ�Ч��Խ�� |
| D��������ʳ�ؽ�������ʱ�����Ժȴ�����ţ�̡�����ⶾ |
������þ��Ͷ�뵽���������ͭ�Ļ����Һ�У���ַ�Ӧ��þ���������䣮��ԭ��Һ������������ͭ�����ʵ���Ũ��֮��Ϊ��������
| A��10��3 | B��5��3 |
| C��1��4 | D��1��1 |
��NA��ʾ�����ӵ�������ֵ������˵����ȷ���ǣ�������
| A��100mL 1mol/L NaHCO3��Һ����0.1NA��HCO3- |
| B�����³�ѹ�£�1mol��������2NA��ԭ�� |
| C��7.8g Na2S��Na2O2�Ļ�����к��е�������������0.1NA |
| D�����������£�1molCl2������Fe�۳�ַ�Ӧ��ת�Ƶĵ�����Ϊ3NA |
���³�ѹ�¶�����̼�������Ʒ�Ӧ������������������28g����Ӧ���й����ʵ���������ȷ���ǣ�������
| ������̼ | ̼���� | ת�Ƶĵ��� | |
| �� | 22.4L | 1mol | |
| �� | 1mol | 1mol | |
| �� | 106g | 1mol | |
| �� | 106g | 2mol |
| A���٢� | B���ڢ� | C���ڢ� | D���٢� |
����˵������ȷ���ǣ�������
| A����ƹ����е��Һ���ʵ�Ũ�Ȼ������ֲ��� |
| B�����������Һ���ܱ�ǿ�����Һ��������ǿ |
| C��ʹ�ô������Խ��ͷ�Ӧ�Ļ�� |
| D��ʹ�ô�������ʹ��Ӧ��ƽ�ⳣ������ |
���и������ʻ�Ϻ�ֻ������һ�ֺ��ƻ�������ǣ�������
| A��Na2CO3������ |
| B��NaOH��Һ��CO2 |
| C��NaHCO3��Һ��ʯ��ˮ |
| D��Na2O2��CO2 |
NA��ʾ�����ӵ�������ֵ������˵����ȷ���ǣ�������
| A����ͭ���缫��ⱥ��NaCl��Һ������·��ͨ��NA������ʱ����������11.2L���壨��״���£� |
| B��6.5gпȫ������һ������Ũ���ᣬ����SO2��H2�Ļ�����壬�����������Ϊ0.1 NA |
| C��1.00 L 1.00 mol?L-1 Na2CO3��Һ�У�CO32-�����������Ϊ2NA |
| D����״���£�22.4 L HCl����ˮ����Һ�к���nA��HCl���� |