��Ŀ����
5����1��ͬ��ͬѹ�£�ͬ�����CO2��CO�����ǵ�����֮��Ϊ11��7���ܶ�֮��Ϊ11��7����2����50mL1mol•L-1 FeCl3��Һ�У�����100mL NaOH��Һǡ����ȫ��Ӧ�����NaOH��Һ��Ũ��Ϊ1.5mol•L-1��
��3����״���µ�HCl����4.48L����ˮ�γ�100mL��Һ��������������ʵ���Ũ��Ϊ2mol•L-1��������Һ����������м��ַ�Ӧ�����ɵ�H2�ڱ�״���µ����Ϊ2.24L��
���� ��1�����ݰ����ӵ����ɿ�֪��ͬ��ͬѹ��ͬ�����CO2��CO�����ʵ�����ͬ�����ݷ��ӵ���ɺͷ��ӵ���Է������������������ܶȵȹ�ϵ��
��2�����ݹ�ϵʽFeCl3��3NaOH�����c=$\frac{n}{V}$��������⣻
��3������n=$\frac{V}{{V}_{m}}$=c•V����Һ�����㣻���ݷ���ʽ��Fe+2HCl=FeCl2+H2�����㣮
��� �⣺��1�����ݰ����ӵ����ɿ�֪��ͬ��ͬѹ��ͬ�����CO2��CO�����ʵ�����ͬ�����ʵ���֮��Ϊ1��1��
��CO2��CO�����ʵ�����Ϊ1mol��
1molCO2������Ϊ1mol��44g/mol=44g��1molCO������Ϊ1mol��28g/mol=28g������֮��Ϊ44��28=11��7��
��CO2��CO�������ͬ�����ܶ�֮�ȵ�������֮�ȣ�Ϊ11��7��
�ʴ�Ϊ��11��7��11��7��
��2�����ݹ�ϵʽFeCl3��3NaOH������n��NaOH��=3��0.05��1=0.15mol������c=$\frac{n}{V}$=$\frac{0.15mol}{0.1L}$=1.5 mol•L-1���ʴ�Ϊ��1.5 mol•L-1��
��3��n��HCl��=$\frac{4.48L}{22.4L/mol}$=0.2mol��c��HCl��=$\frac{0.2mol}{0.1L}$=2mol/L��
��Fe+2HCl=FeCl2+H2��
2mol 22.4L
0.2mol V
V=2.24L��
�ʴ�Ϊ��2mol•L-1��2.24L��
���� ���⿼�����ʵ������йؼ��㣬��Ŀ�ѶȲ���ע���йؼ��㹫ʽ�����ã�

| A�� | Na2CO3�������� | B�� | Na2CO3�Ǽ� | ||
| C�� | Na2CO3��̼���� | D�� | Na2CO3�Ƿǵ���� |
| A�� | Na2O2��NH4Cl��NaCl��Na2SO4 | B�� | FeO��AlCl3��Na2SiO3������ | ||
| C�� | KHCO3��Ca��OH��2��MgCl2������ | D�� | NaCO3��BaCl2��K2S������ |
| A�� | �ܶ�ʳ�ﺬ��ά���أ����߲ˡ�ˮ��������ʳ�����ṩ�����ά���� | |
| B�� | ѡ��ʳ��ʱ��Ҫ���Ǻ�Ӫ���ص����ࡢ�������Ƿ��ױ��������պ����� | |
| C�� | ����ÿ����ҪӪ���ص���������ȶ��� | |
| D�� | ��������ʳ���У������ṩ��������࣬��ֵҲ��� |
| A�� | ����H+�������������Һ�У�SO${\;}_{3}^{2-}$��Ba2+��K+��NO${\;}_{3}^{-}$ | |
| B�� | ʹ��̪�Ժ�ɫ����Һ�У�NO${\;}_{3}^{-}$��Cl-��K+��Ba2+ | |
| C�� | �������ܷų���������Һ�У�Na+��H+��CH3COO-��Cl- | |
| D�� | �����Ե���ɫ��Һ�У�K+��Fe2+��NO32-��SO42- |
| A�� | �����۷�Ӧ�ų���������ɫ��Һ�У�NO3-��Al3+��Na+��SO42- | |
| B�� | ʹ��ɫʯ����ֽ��������ҺSO42-��HCO3-��Na+��K+ | |
| C�� | ������Һ��Fe3+��Al3+��NO3-��SO42- | |
| D�� | �����£�$\frac{C��{H}^{+}��}{C��O{H}^{-}��}$=10-12����Һ��K+��AlO2-��CO32-��Na+ |
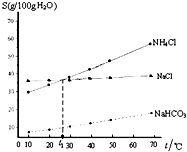
| A�� | t1��ʱ������NaCl��NH4Cl ��Һ�����ʵ���Ũ����ͬ | |
| B�� | ��ͬ�¶��£�NaHCO3��ˮ�е��ܽ�ȱ�NaCl��NH4ClС�ܶ� | |
| C�� | NH4Cl �ܽ�����¶ȱ仯�ϴ� | |
| D�� | ��ҵ���ƴ�������͵�NaCl��Һ��ͨ��NH3��CO2������NaHCO3�������� |
