��Ŀ����
18������������ȷ���ǣ����ڳ����£���������| A�� | pH=3��ǿ����Һ1 mL����ˮϡ����100 mL����ҺpH����2����λ | |
| B�� | 1 L 0.50 mol/L NH4Cl ��Һ��2 L 0.25 mol/L NH4Cl ��Һ��NH4+ ���ʵ������ߴ� | |
| C�� | ���Σ���ҪΪBaCl2������ʳ��ʳ��ʱ������0.5%��Na2SO4��Һ�ⶾ���ܶ���ɺ�����γɣ���ĭ���������ĭ��������ԭ�������ó����ܽ�ƽ��ԭ�������� | |
| D�� | pH=4��Ũ�Ⱦ�Ϊ0.1 mol•L-1 ��CH3COOH��CH3COONa�����Һ�У�c��CH3COO-��-c��CH3COOH��=2����10-4-10-10��mol/L |
���� A����ϡ��ʱ��pH������
B������n=cV�����㣻笠����ӵ�Ũ��ԽС��ˮ��̶�Խ��
C���������ж����������ܺ���������ӷ�Ӧ���ɲ�����ˮ��������ᱵ������̼��������Һˮ��ʼ��ԣ�������ˮ������ԣ�̼�����ƺ�����������ٽ�ˮ�����ɶ�����̼��
D�������£����ҺpH=4��c��H+��=1.0��10-4mol/L������Kw��֪��c��OH-��=1.0��10-10mol/L���ɵ���غ��֪��c��CH3COO-��-c��Na+��=c��H+��-c��OH-�����������غ㣺2c��Na+��=c��CH3COOH��+c��CH3COO-�����ݴ˷�����
��� �⣺A��pH=3��ǿ����Һ1 mL����ˮϡ����100 mL����ҺpH����2����λ����A����
B��1 L 0.50 mol/L NH4Cl ��Һ��2 L 0.25 mol/L NH4Cl ��Һ��NH4+ ���ʵ���ǰ�ߴ�B����
C���������ж������Կ����Եı����ж��������Ӻ���������ӷ�Ӧ���ɲ��������ˮ�����ᱵ���������Ե��������������Ե�����ת�������Կ����ó����ܽ�ƽ��ԭ�������ͣ�̼������ˮ��ʹ����Һ�ʼ��ԣ�������ˮ��ʹ����Һ�����ԣ������Ӻ����������ӷ�Ӧ����ˮ����̼�����ƺ���������ٽ�ˮ�⣬�Ӷ�Ѹ�ٵIJ���������̼�����Կ���������ˮ��ԭ����������ĭ���������ԭ������C����
D�������£����ҺpH=4��c��H+��=1.0��10-4mol/L������Kw��֪��c��OH-��=1.0��10-10mol/L���ɵ���غ��֪����c��CH3COO-��-c��Na+��=c��H+��-c��OH-�����������غ㣺��2c��Na+��=c��CH3COOH��+c��CH3COO-�������2+��ɵã�c��CH3COO-��-c��CH3COOH��=2��c��H+��-c��OH-����=2����10-4-10-10�� mol/L����D��ȷ��
��ѡD��
���� ���⿼��pH�����ϡ�͡�����ˮ�⼰���ԵıȽϵȣ�ע��ˮ�������ԽϡԽˮ�������������Ŀ�Ѷ��еȣ�

| A�� | ���ᡢ��������ƺ���ʯ�ҷֱ������ᡢ��κ������� | |
| B�� | �ƾ������ᱵ��ˮ�ֱ����ڵ���ʡ�ǿ����ʺ�������� | |
| C�� | ����������������ɢϵ�ı��������Ǿ��ж�������� | |
| D�� | ���ð�Ĥ�ɳ�ȥ������Һ�е�����NaCl |
| A�� | ij��Һ����ˮ�������c��H+��=1��10-a mol/L����a��7ʱ������ҺpHһ��Ϊ14-a | |
| B�� | ������������ʵ���Ũ�ȵ�Na2CO3��Һ��NaHCO3��Һ��ϣ�3c��Na+��=2c��CO32-��+2c��HCO3-��+2c��H2CO3�� | |
| C�� | ��0.2 mol/L��ijһԪ��HA��Һ��0.1 mol/L NaOH��Һ�������Ϻ���ҺpH����7����Ӧ��Ļ��Һ��c��HA����c��Na+����c��A-�� | |
| D�� | pH=3�Ķ�Ԫ����H2R��Һ��pH=11��NaOH��Һ��Ϻ��Һ��pH����7����Ӧ��Ļ��Һ��2c��R2-��+c��HR-����c��Na+�� |
| A�� | ���³�ѹ�£��ܽ��С�����β�����ת��Ϊ�ܽ����Դ�Щ������ | |
| B�� | ��ͬ�����£�2mol��ԭ�������е�����С��1mol����������е����� | |
| C�� | ��ͬ�����£�1L0.1mol/L��̼������Һ��pH����2L0.1mol/L�Ĵ�������Һ��pH | |
| D�� | 1L 1mol/L��̼������Һ����S02��������1L 1mol/L������Һ����SO2���� |
| a | ||||
| b | ||||
| c | d | |||
| e |
| A�� | bԪ�س�0���⣬ֻ��һ�ֻ��ϼ� | |
| B�� | cԪ��ֻ���γ�һ���⻯���Һܲ��ȶ� | |
| C�� | dԪ��������Ԫ����ԭ�Ӱ뾶��С��һ�� | |
| D�� | eԪ������������ˮ����Ͳ�ͬ��������������Һ��Ӧ���������������� |
| A�� | ��ȥCO2�е�HCl | |
| B�� | �Ʊ�Fe��OH��2���ܽϳ�ʱ��۲�����ɫ | |
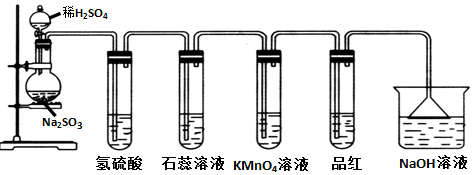
| C�� | ��֤̼�������ǿ�ڹ��� | |
| D�� | ���뱽�;ƾ� |
��$��_{��}^{+��}$��$��_{��}^{+��}$����
| A�� | ��һ���������� | B�� | ��ֻ���� S �� N2 | ||
| C�� | ��һ���������� | D�� | ��Ӧ�ڿ����Ƿ�������ԭ��Ӧ |
 ͼʾװ�ÿ�������ȡ�۲�Fe��OH��2�ڿ����б�����ʱ����ɫ�仯��ʵ��ʱ����ʹ�õ�ҩƷ����м��6mol•L-1�����ᣬ�����Լ���ѡ��
ͼʾװ�ÿ�������ȡ�۲�Fe��OH��2�ڿ����б�����ʱ����ɫ�仯��ʵ��ʱ����ʹ�õ�ҩƷ����м��6mol•L-1�����ᣬ�����Լ���ѡ��