��Ŀ����
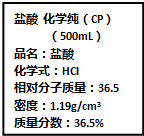 ��Ҫ���������С�⣮
��Ҫ���������С�⣮��1��0.5mol NH3������Ϊ
��2���ڱ�״���£�224mLij���������Ϊ0.64g����������Ħ������Ϊ
��3������100mL 0.1mol/L Na2CO3��Һ����ҪNa2CO3?10H2O������Ϊ
��4����ͼ��ij��ѧʵ���Ҵӻ�ѧ�Լ��̵���ص������Լ���ǩ�ϵIJ������ݣ�
�ٸ�Ũ��������ʵ���Ũ��
���ø�Ũ��������200mL1mol/L��ϡ���������Ũ����������
���㣺���ʵ�������ؼ���
ר�⣺������
��������1������m=nM���㰱������������N=nNA���������Ŀ��ÿ���������Ӻ���10�����ӣ����е�����ĿΪ������Ŀ��10����
��2������n=
������������ʵ������ٸ���M=
��������Ħ��������
��3������n=cV����̼���Ƶ����ʵ���������m=nM����̼���ƾ����������
��4���ٸ���c=
�������������ʵ���Ũ�ȣ�
�ڸ���ϡ�Ͷ��ɣ�ϡ��ǰ�����ʵ����ʵ������䣬�ݴ˼�����ҪŨ����������
��2������n=
| V |
| Vm |
| m |
| n |
��3������n=cV����̼���Ƶ����ʵ���������m=nM����̼���ƾ����������
��4���ٸ���c=
| 1000�Ѧ� |
| M |
�ڸ���ϡ�Ͷ��ɣ�ϡ��ǰ�����ʵ����ʵ������䣬�ݴ˼�����ҪŨ����������
���
�⣺��1��0.5mol��������Ϊ0.5mol��17g/mol=8.5g������N=nNA���������ĿΪ0.5mol��NAmol-1=0.5NA��ÿ���������Ӻ���10�����ӣ����е�����ĿΪ������ĿΪ10��0.5NA=5NA��
�ʴ�Ϊ��8.5��0.5NA��5NA��
��2����������ʵ���Ϊ
=0.01mol��������Ħ������Ϊ
=64g/mol���ʴ�Ϊ��64g/mol��
��3��̼���Ƶ����ʵ���Ϊ0.1L��0.1mol/L=0.01mol����Ҫ̼���ƾ��������Ϊ0.01mol��286g/mol=2.86g���ʴ�Ϊ��2.86g��
��4���ٸ���c=
��֪������������ʵ���Ũ��Ϊ
mol/L=11.9mol/L��
�ڸ���ϡ�Ͷ��ɣ�ϡ��ǰ�����ʵ����ʵ������䣬��ҪŨ��������Ϊ
=16.8mL��
�ʴ�Ϊ��11.9mol/L��16.8��
�ʴ�Ϊ��8.5��0.5NA��5NA��
��2����������ʵ���Ϊ
| 0.224L |
| 22.4L/mol |
| 0.64g |
| 0.01mol |
��3��̼���Ƶ����ʵ���Ϊ0.1L��0.1mol/L=0.01mol����Ҫ̼���ƾ��������Ϊ0.01mol��286g/mol=2.86g���ʴ�Ϊ��2.86g��
��4���ٸ���c=
| 1000�Ѧ� |
| M |
| 1000��1.19��36.5% |
| 36.5 |
�ڸ���ϡ�Ͷ��ɣ�ϡ��ǰ�����ʵ����ʵ������䣬��ҪŨ��������Ϊ
| 200mL��1mol/L |
| 11.9mol/L |
�ʴ�Ϊ��11.9mol/L��16.8��
���������⿼�����ʵ����йؼ��㣬�ѶȲ���ע��Թ�ʽ���������������Ӧ�ã�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
���й��ڻ�ѧƽ�ⳣ����˵���У���ȷ���ǣ�������
| A����ѧƽ�ⳣ������λ |
| B��ƽ�ⳣ��Խ����ת����Խ�� |
| C������һ����ѧ�������̶��ķ�Ӧ��ƽ�ⳣ���Ĵ�Сֻ���¶��й� |
| D����ƽ�ⳣ������ʽ�У���Ӧ���Ũ��Ϊ��ʼŨ�ȣ��������Ũ��Ϊƽ��Ũ�� |
���й���0.05mol?L-1��K2SO4��Һ��˵���У���ȷ���ǣ�������
| A��1L��Һ�к���K+��Ŀ��0.1NA |
| B��1L��Һ�к���K+��SO42-����Ϊ0.1NA |
| C��1 L��Һ��K+��Ũ����0.05 mol?L-1 |
| D��2 L��Һ�к���SO42-��Ũ����0.1mol?L-1 |
���з�Ӧ��Na2O2+SO2�TNa2SO4��Ƚϣ�Na2O2��������ͬ���ǣ�������
| A��2Na2O2+2CO2�T2Na2CO3+O2�� |
| B��2Na2O2+2SO3�T2Na2SO4+O2�� |
| C��Na2O2+H2SO4�TNa2SO4+H2O2 |
| D��3Na2O2+Cr2O3�T2Na2CrO4+Na2O |
���з�Ӧ�����ӷ���ʽ��д��ȷ���ǣ�������
| A���Ȼ�ͭ��Һ�����۷�Ӧ��CuCl2+Fe�TFe2++Cu+2Cl- |
| B��ϡH2SO4�����۷�Ӧ��2Fe+6H+�T2Fe3++3H2�� |
| C������������Һ��ϡH2SO4��Ӧ��Ba2++SO42-�TBaSO4�� |
| D��������������Һ�����Ȼ������壺H++OH-�TH2O |
���������ʷ����У�ǰ�߰������ߵ��ǣ�������
| A������������ ������ |
| B����Һ ���� |
| C������� ������ |
| D������� ���� |
����˵����ȷ���ǣ�������
| A����⾫��ͭʱ��ͬһʱ���������ܽ�ͭ����������������ͭ��������� |
| B���ϳɰ����������н�NH3Һ�����룬�ɼӿ�����Ӧ���ʣ����N2��H2��ת���� |
| C�����ڡ�H��0����S��0�ķ�Ӧ���������Է����У���Ӧ����Ҳ�ܿ� |
| D��S��g��+O2��g��=SO2��g����H1��S��s��+O2��g��=SO2��g����H2�����H1����H2 |
 ������и�С��ļ���
������и�С��ļ���