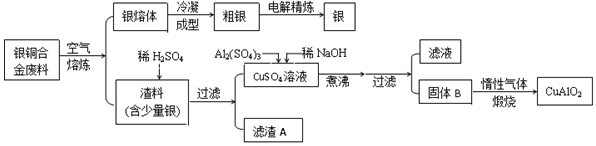
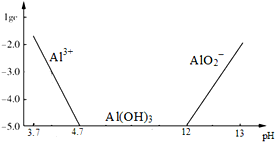
��Ŀ����
6�� ����Cu2O���ھ��������Ĵ����ܶ��ܵ���ע����Ϊ��ȡCu2O�����ַ�����
����Cu2O���ھ��������Ĵ����ܶ��ܵ���ע����Ϊ��ȡCu2O�����ַ�����| ������ | ��̿���ڸ��������»�ԭCuO |
| ������ | ��ⷨ��2Cu+H2O$\frac{\underline{\;���\;}}{\;}$ Cu2O+H2�� |
| ������ | ���£�N2H4����ԭ����Cu��OH��2 |
��2����֪��2Cu��s��+$\frac{1}{2}$O2��g��=Cu2O��s����H=-akJ•mol-1
C��s��+$\frac{1}{2}$O2��g��=CO��s����H=-bkJ•mol-1
Cu��s��+$\frac{1}{2}$O2��g��=CuO��s����H=-ckJ•mol-1
��I�����ķ�Ӧ��2CuO��s��+C��s��=Cu2O��s��+CO��g����H=2c-a-bkJ•mol-1��
��3������II�������ӽ���Ĥ���Ƶ��Һ��OH-��Ũ�ȶ��Ʊ�����Cu2O��װ����ͼ��ʾ���õ�ص�������ӦʽΪ2Cu-2e-+2OH-=Cu2O+H2O��
��4������IIIΪ������������Һ̬ƴ��N2H4����ԭ����Cu��OH��2���Ʊ�����Cu2O��ͬʱ�ų�N2�����Ʒ��Ļ�ѧ����ʽΪ4Cu��OH��2+N2H4$\frac{\underline{\;\;��\;\;}}{\;}$2Cu2O+N2��+6H2O��
��5������ͬ���ܱ������У����������ַ����Ƶõ�Cu2O�ֱ���д��ֽ�ˮ��ʵ�飺
2H2O��g��$?_{Cu_{2}O}^{����}$2H2��g��+O2��g����H��0
ˮ������Ũ����ʱ��t�仯�����ʾ��
| ��� | 0 | 10 | 20 | 30 | 40 | 50 | |
| �� | T1 | 0.050 | 0.0492 | 0.0486 | 0.0482 | 0.0480 | 0.0480 |
| �� | T1 | 0.050 | 0.0488 | 0.0484 | 0.0480 | 0.0480 | 0.0480 |
| �� | T2 | 0.10 | 0.094 | 0.090 | 0.090 | 0.090 | 0.090 |
A��ʵ���¶ȣ�T1��T2
B��ʵ���ǰ20min��ƽ����Ӧ����v��O2��=7��10-5mol/��L•min��
C��ʵ��ڱ�ʵ������õĴ���Ч�ʸ�
��6��25��ʱ����50mL0.018mol/L��AgNO3��Һ�м���50mL0.02mol/L���ᣬ���ɳ���������֪Ksp��AgCl��=1.8��10-10�����ʱ��Һ�е�c��Ag+��=1.8��10-7mol/L��������仯���Բ��ƣ�������������ɺ����Һ�м���100mL0.001mol/L���ᣬ�Ƿ����������������ǡ�����
���� ��1����̿���ڸ��������»�ԭCuO��������Cu��
��2�����ݸ�˹���ɣ�����֪�Ȼ�ѧ����ʽ�����ʵ���ϵ�����мӼ�����Ŀ���Ȼ�ѧ����ʽ����Ӧ��Ҳ������Ӧ��ϵ������Ӧ�ļӼ���
��3����������������Ӧ������������Cu������ʧȥ���ӵõ�Cu2O��H2O��
��4��Һ̬�£�N2H4����ԭ����Cu��OH��2���Ʊ�����Cu2O��ͬʱ�ų�N2�����ݵ�ʧ�����غ������
��5��A�������¶ȶԻ�ѧƽ���ƶ���Ӱ��֪ʶ���ش�
B�����ݷ�Ӧ����v=$\frac{��c}{��t}$�����㣻
C��������������ѧƽ��״̬�ĸı䣬��ʹ��Ӧ���ʼӿ죻
��6��������Һ��Ϻ�������Ӧʱc��Ag+����c��Cl-������ƽ��ʱ��Һ��Ag+��Ũ��Ϊxmol/L�����ݷ�ӦAg+��aq��+Cl-��aq��?AgCl��s������ʾ��ƽ��ʱc��Ag+����c��Cl-���������ܶȻ������з��̼��㣻
�ټ����Ϻ���Һ����������Ӧʱc��Ag+����c��Cl-������������Ũ�Ȼ������ܶȻ��Ƚϣ��ж��Ƿ����������
��� �⣺��1����̿���ڸ��������»�ԭCuO��������Cu����Ӧ����������Cu2O��
�ʴ�Ϊ����Ӧ�����ƣ���ԭ����Cu��
��2����2Cu��s��+$\frac{1}{2}$O2��g��=Cu2O��s����H=-akJ•mol-1��
��C��s��+$\frac{1}{2}$O2��g��=CO��g����H=-bkJ•mol-1��
��Cu��s��+$\frac{1}{2}$O2��g��=CuO��s����H=-ckJ•mol-1
�ɸ�˹���ɿ�֪����-�ۡ�2+�ڵ�2CuO��s��+C��s��=Cu2O��s��+CO��g����H=2c-a-bkJ•mol-1��
�ʴ�Ϊ��2c-a-b��
��3����������������Ӧ������������Cu������ʧȥ���ӵõ�Cu2O��H2O�������缫��ӦʽΪ��2Cu-2e-+2OH-=Cu2O+H2O��
�ʴ�Ϊ��2Cu-2e-+2OH-=Cu2O+H2O��
��4��Һ̬�£�N2H4����ԭ����Cu��OH��2���Ʊ�����Cu2O��ͬʱ�ų�N2���䷴Ӧ�ķ���ʽΪ��4Cu��OH��2+N2H4$\frac{\underline{\;\;��\;\;}}{\;}$2Cu2O+N2��+6H2O��
�ʴ�Ϊ��4Cu��OH��2+N2H4$\frac{\underline{\;\;��\;\;}}{\;}$2Cu2O+N2��+6H2O��
��5��A��ʵ���¶�Խ�ߣ��ﵽ��ѧƽ��ʱˮ����ת����Խ�ں͢���ȣ���ת���ʸߣ�����T2��T1����A��ȷ��
B��ʵ���ǰ20min��ƽ����Ӧ���� v��O2��=$\frac{��c}{��t}$=$\frac{\frac{0.05mol/L-0.0486mol/L}{2}}{20min}$=3.5��10-5 mol•L-1 min-1����B����
C���٢ڻ�ѧƽ��״̬δ�ı䣬�ڷ�Ӧ���ʼӿ죬�ȵ���ƽ�⣬��ʵ��ڱ�ʵ������õĴ���Ч�ʸߣ���C��ȷ��
��ѡAC��
��6����Һ��Ϻ�������Ӧʱc��Ag+��=$\frac{1��}{2}$��0.018mol•L-1=0.009mol•L-1��c��Cl-��=$\frac{1}{2}$��0.02mol•L-1=0.01mol•L-1����ƽ��ʱ��Һ��Ag+��Ũ��Ϊxmol/L��
Ag+��aq��+Cl-��aq��?AgCl��s��
��0.009-x��mol/L ��0.009-x��mol/L
��Ӧ����Һ��c��Cl-��=0.01mol/L-��0.009-x��mol/L=��0.001+x��mol/L����x����0.001+x��=1.8��10-10������0.009��0.01=9��10-5��1.8��10-10����0.001+x��0.001�����x��1.8��10-7��
����Ӧ�����Һ�м���100mL0.001mol•L-1���ᣬ��������Ӧʱc��Ag+��=$\frac{1}{2}$��1.8��10-7mol•L-1=9��10-8mol•L-1��c��Cl-��=$\frac{1}{2}$��[��0.001+1.8��10-7+0.001]mol•L-1��0.001mol•L-1��������Ũ�Ȼ�=0.001��9��10-8=0.9��10-10��1.8��10-10����û�г���������
�ʴ�Ϊ��1.8��10-7mol/L����
���� ���⿼���˹���ɵ�Ӧ�á����ԭ����Ӧ�á�������ԭ��Ӧ����ʽ����д����ƽ����ѧ��Ӧ���ʵļ����Լ���ѧƽ���ƶ���ƽ�ⳣ�����ܶȻ���Ӧ�õȣ���Ŀ�ۺ��Խϴ��Ѷ��еȣ��Ƕ�֪ʶ���ۺ����ã�ע�����֪ʶ���������գ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ��������ˮ�ķ�Ӧ��ʵ��ʱ����ȡ�̶���С�Ľ����ƣ�����ֽ���ɱ���ú�ͣ������ձ��У��������η�̪��Һ���ټ�������ˮ��Ȼ��۲첢��¼ʵ������ | |
| B�� | ��KI��FeCl3��Һ���Թ��л�Ϻ���CCl4�������ã��²�Һ�����Ϻ�ɫ���������ԣ�Fe3+��I2 | |
| C�� | �ⶨ��Ũ�ȵ�Na2CO3��Na2SO3 ��Һ��pH��ǰ��pH �Ⱥ��ߵĴ���ǽ����ԣ�S��C | |
| D�� | �������� NaOH ��Һ���Ⱥ���AgNO3��Һ��δ���ֵ���ɫ������˵��������û��ˮ�� |
 50mL1.0mol/L�����50mL1.1mol/L����������Һ����ͼ��ʾװ���н����кͷ�Ӧ����ͨ���ⶨ��Ӧ���������ų��������������к��ȣ��Իش��������⣺
50mL1.0mol/L�����50mL1.1mol/L����������Һ����ͼ��ʾװ���н����кͷ�Ӧ����ͨ���ⶨ��Ӧ���������ų��������������к��ȣ��Իش��������⣺��1����������β����������Ϊ���ν�������ͭ������������к�����ֵ��Ӱ����ƫ�ͣ��ƫ�ߡ�����ƫ�͡�����Ӱ�족����
��2���������60mL 1.0mol•L-1�����50mL 1.1mol•L-1����������Һ���з�Ӧ����������ʵ����ȣ������������ӣ�����ӡ��������١����䡱���������к�����ֵ���䣨����ӡ��������١����䡱����
��3��������Ba��OH��2������������������������Һ��������H2SO4��Ba��OH��2��Ӧ���ɵ�BaSO4����ʱҲ�������仯������ȣ���
��4����ʵ��С����������ʵ�飬ÿ��ȡ��Һ��50mL������¼���ԭʼ���ݣ�
| ʵ����� | ��ʼ�¶�t1/�� | ��ֹ�¶ȣ�t2���� | �²t2•t1���� | ||
| ���� | NaOH��Һ | ƽ��ֵ | |||
| 1 | 25.1 | 24.9 | 25.0 | 31.7 | 6.7 |
| 2 | 25.1 | 25.1 | 25.1 | 31.9 | 6.8 |
| 3 | 25.1 | 25.1 | 25.1 | 32.0 | 6.9 |
��5������ʵ����ֵ�����57.3kJ/mol��ƫ�����ƫ���ԭ�������acd������ĸ����
a��ʵ��װ�ñ��¡�����Ч����
b����ȡNaOH��Һ�����ʱ���Ӷ���
c���ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ���
d�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�ӲⶨHCl��Һ���¶ȣ�
| ���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| �� | �� | �� | ||||||
| �� | �� | �� | �� | �� | �� | �� | ||
| �� | �� | �� |
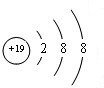
��2���١���Ԫ�ص����������Ծ͵�ˮ������������ǿ����HClO4��������ǿ����KOH��
��3����Ҫ��д�������������ʵĵ���ʽ���ٵ��⻯��
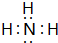 ���������������Ӧ��ˮ����
���������������Ӧ��ˮ����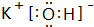 ��
����4���ڢ����ĵ����У���ѧ���ʽϻ��õ���Cl2������1����ѧ��Ӧ˵������ʵ��д����Ӧ�Ļ�ѧ����ʽ����Cl2+2KBr�T2KCl+Br2��
| A�� | Ԫ��C��D��E������������Ӧ��ˮ����֮�����������Է�����Ӧ | |
| B�� | 1mol��Ԫ��A��B����Һ���18 mol e-�Ļ�����ֻ��һ�� | |
| C�� | ��DԪ�ص�����Һֻ�������ԣ��������Լ��� | |
| D�� | ������AE��CE������ͬ���͵Ļ�ѧ�� |
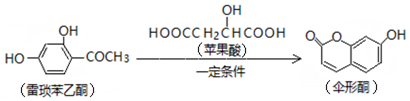
����˵������ȷ���ǣ�������
| A�� | һ������������ͪ����3��̼̼˫�� | |
| B�� | ƻ�����һ�����۲�����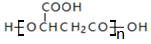 | |
| C�� | 1molɡ��ͪ������NaOH��Һ��Ӧ����������2mol NaOH | |
| D�� | ��������ͪ��ƻ���ᡢɡ��ͪ���ܸ�FeCl3��Һ������ɫ��Ӧ |
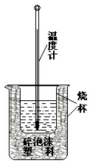 50ml 0.50mol•L-1������50mL 0.55mol•L-1NaOH��Һ������ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ�����зų��������ɼ����к��ȣ��ش��������⣺
50ml 0.50mol•L-1������50mL 0.55mol•L-1NaOH��Һ������ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ�����зų��������ɼ����к��ȣ��ش��������⣺