��Ŀ����
7��ʵ����������480mL0.2mol/L��NaOH��Һ����1��ͨ�������֪��Ӧ��������ƽ����4.0��NaOH���壮
��2����ʵ������Ҫ�IJ�����������Ͳ����ͷ�ιܡ��ձ����500ml����ƿ��
��3��������ѡ������IJ�����д����ȷ��˳����д��Ӧ����ĸ��DHFAE��
A��ϴ�� B������ C����ȡ D������ E������F��ת����Һ G��ϡ�� H���ܽ�
��4����ʵ������г������������������Һ��Ũ���к�Ӱ�죨�ƫ�ߡ���ƫ�͡�����Ӱ�족�� �ٶ���ʱ������ˮ���������˿̶�ƫ��
������ƿ�������һ����ˮ����Ӱ��
�۶���ʱ���ӹ۲쵽Һ��պõ���̶���ƫ��
�� 5����NaOH��Һ��2.24L������STP����ǡ����ȫ��Ӧ��������������Һ1000mL��
��6����18mol/L����������100mL 1.0mol/L���ᣬʵ��ʱӦѡ�õ�������CEFGH����д��ţ���
A��100mL��Ͳ B��������ƽ C�������� D��50mL����ƿ
E��10mL��Ͳ F����ͷ�ι� G��50mL�ձ� H��100mL����ƿ��
���� ��1������m=cVM������Ҫ���ʵ�������
��2����������һ�����ʵ���Ũ����Һ��һ�㲽��ѡ����Ҫ������������������Һ���ѡ����Ҫ����ƿ���
��3������һ�����ʵ���Ũ����Һ��һ�㲽�裺���㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȣ��ݴ�����
��4���������������ʵ����ʵ�������Һ�����Ӱ�죬����c=$\frac{n}{V}$������������
��5��n��Cl2��=0.1mol�����ݷ�Ӧ�����ӷ���ʽCl2+2OH-=CIO-+Cl?+H2O���㣻
��6����������һ�����ʵ���Ũ����Һ�IJ�����
��� �⣺��1������480mL0.2mol/L��NaOH��Һ����Ҫѡ��500mL����ƿ����Ҫ�������Ƶ�����m=0.2mol/L��0.5L��40g/mol=4.0g��
�ʴ�Ϊ��4.0��
��2������һ�����ʵ���Ũ����Һ��һ�㲽�裺���㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȣ��õ���������������ƽ��ҩ�ס��ձ���������������ƿ����ͷ�ιܣ�����480mL0.2mol/L��NaOH��Һ��Ӧѡ��500mL����ƿ�����Ի�ȱ�ٵIJ���������500ml����ƿ��
�ʴ�Ϊ��500ml����ƿ��
��3������һ�����ʵ���Ũ����Һ��һ�㲽�裺���㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȣ�������ȷ��˳��Ϊ��D H B F A E C G��
�ʴ�Ϊ��DHFAE��
��4���ٶ���ʱ������ˮ���������˿̶ȣ�������Һ���ƫ����ҺŨ��ƫ�ͣ�
������ƿ�������һ����ˮ�֣������ʵ����ʵ�������Һ������������Ӱ�죬��ҺŨ�Ȳ��䣻
�۶���ʱ���ӹ۲쵽Һ��պõ���̶��ߣ�������Һ���ƫ����ҺŨ��ƫ�ͣ�
�ʴ�Ϊ����ƫ�� ����Ӱ�� ��ƫ�ͣ�
��5��n��Cl2��=0.1mol���ɷ�Ӧ�����ӷ���ʽ��Cl2+2OH-=CIO-+Cl?+H2O��֪n��NaOH��=0.2mol������������Һ���V��NaOH��=$\frac{0.2mol}{0.2mol/L}$=1L=1000mL��
�ʴ�Ϊ��1000��
��6������100mL 1.0mol/L����IJ���Ϊ�����㡢��ȡ��ϡ�͡���ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȵȣ�ѡ�õ��������Ⱥ�˳��Ϊ��10mL��Ͳ��50mL�ձ�����������100mL����ƿ����ͷ�ιܣ�
�ʴ�Ϊ��CEFGH��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���ȷ����ԭ���Ͳ��������ǽ���ؼ���ע���������ķ�������Ŀ�ѶȲ���

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| �������� | HCl��aq�� | FeCl3 | NaOH | CH3COONa | C2H5OH |
| ��Һ��pH | 3 | 4 | 10 | 11 | δ�ⶨ |
| ˮ�ĵ���̶� | ��1 | ��2 | ��3 | ��4 | ��5 |
| A�� | ��3����1����5����4����2 | B�� | ��4����2����5����3����1 | C�� | ��2����4����5����1����3 | D�� | ��1����3����5����2����4 |
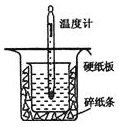 �к��ȵIJⶨ�Ǹ�����Ҫ�Ķ���ʵ�飮ȡ0.55mol/L��NaOH��Һ50mL��0.25mol/L������50mL����ͼ��ʾ��װ���н����к��ȵIJⶨʵ�飬�ش��������⣺
�к��ȵIJⶨ�Ǹ�����Ҫ�Ķ���ʵ�飮ȡ0.55mol/L��NaOH��Һ50mL��0.25mol/L������50mL����ͼ��ʾ��װ���н����к��ȵIJⶨʵ�飬�ش��������⣺��1������ͼʵ��װ�ÿ���������ȱ�ٵ�һ�ֲ�����Ʒ�ǻ��β��������_��
��2��������60mL 0.25mol•L-1H2SO4��50mL 0.55mol•L-1NaOH��Һ���з�Ӧ������ʵ����ȣ����ų�����������ȣ����ȡ���������ȡ�������ʵ���������ȷ���������к���������ȡ�������ȡ���
��3������NaOH��Һ����ȷ�����ǣ�C�� ��������ѡ������
A���ز������������롡 B���������������� C��һ��Ѹ�ٵ���
��4��ʵ�����������
������д�±��еĿհף�
| �¶� ʵ������� | ��ʼ�¶�t1�� | ��ֹ�¶�t2/�� | �¶Ȳ�ƽ��ֵ ��t2-t1��/�� | ||
| H2SO4 | NaOH | ƽ��ֵ | |||
| 1 | 26.2 | 26.0 | 26.1 | 29.5 | |
| 2 | 27.0 | 27.4 | 27.2 | 32.3 | |
| 3 | 25.9 | 25.9 | 25.9 | 29.2 | |
| 4 | 26.4 | 26.2 | 26.3 | 29.8 | |
������ʵ����ֵ�����57.3kJ/mol��ƫ�����ƫ���ԭ������ǣ�����ĸ��abc��
a��ʵ��װ�ñ��¡�����Ч����
b�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�ӲⶨH2SO4��Һ���¶�
c���ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ��У�
| A�� | SO2���������ԣ�������Ư��ֽ�� | |
| B�� | �����������ԣ������ڵ�̲��� | |
| C�� | �������Ⱦ��л�ԭ�ԣ�����������ˮ��ɱ������ | |
| D�� | NH3���л�ԭ�ԣ�����NH3������CuO������ȡ����N2 |
 ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�����������Һʱ��ѡ���̪��ָʾ��������д���пհף�
ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�����������Һʱ��ѡ���̪��ָʾ��������д���пհף���1���ñ���������Һ�ζ����������������Һʱ�����ְ�����ʽ�ζ��ܵĻ���������ҡ����ƿ���۾�ע����ƿ����Һ��ɫ�ı仯��
��2�����в����п���ʹ��������������Һ��Ũ����ֵƫ�͵���D��
A����ʽ�ζ���δ�ñ�������Һ��ϴ��ֱ��ע���������Һ
B���ζ�ǰʢ������������Һ����ƿ������ˮϴ����û�и���
C����ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ
D����ȡ�������ʱ����ʼ���Ӷ������ζ�����ʱ���Ӷ���
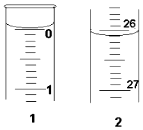
��3�����ζ���ʼ�ͽ���ʱ����ʽ�ζ����е�Һ����ͼ��ʾ����������
��Һ�����Ϊ26.10mL��
��4��ijѧ����������ʵ��ֱ��¼�й����������
| �ζ� ���� | �������������� Һ�����/mL | 0.1000mol•L+1����������mL�� | |
| �ζ�ǰ�̶� | �ζ���̶� | ||
| ��һ�� | 25.00 | 0.00 | 26.11 |
| �ڶ��� | 25.00 | 1.56 | 30.30 |
| ������ | 25.00 | 0.22 | 26.31 |
��1���������Թ���ȼ�ϵ�أ����ط�Ӧԭ��Ϊ4NH3+3O2=2N2+6H2O��NH3Ӧͨ��ȼ�ϵ�صĸ������������������������֪�������ҺΪKOH��Һ�������ĵ缫��ӦʽΪ2NH3+6OH--6e-�TN2+6H2O��
��2����0.5L�����ܱ������У�һ������N2��H2���з�Ӧ��N2��g��+3H2��g��?2NH3��g����H=bkJ•mol-1���仯ѧƽ�ⳣ��K���¶ȵĹ�ϵ�����
| �¶�/�� | 200 | 300 | 400 |
| K | 1.0 | 0.85 | 0.5 |
�������ϣ�Ϊ������ƽ��ʱH2��ת���ʣ��ɲ�ȡ�Ĵ�ʩ��ad������ĸ��ţ���
a������ѹǿ b��ʹ�ú��ʵĴ���
c�������¶� d����ʱ����������е�NH3
��400��ʱ�����ijʱ�̰��������������������ʵ����ֱ�Ϊ3mol��2mol��1molʱ����ʱ�̸÷�Ӧ��v����N2�� С��v�棨N2��������ڡ���С�ڡ����ڡ�����
��3����֪��
��4NH3��g��+3O2��g���T2N2��g��+6H2O��g����H=-1266.8kJ•mol-1
��N2��g��+O2��g���T2NO��g����H=+180.5kJ•mol-1
д�������´��������Ȼ�ѧ����ʽ4NH3��g��+5O2��g��=4NO��g��+6H2O��g����H=-905.8 kJ/mol��
| Ԫ�ش��� | L | M | X | R | T | Q |
| ԭ�Ӱ뾶/nm | 0.160 | 0.143 | 0.102 | 0.089 | 0.074 | 0.078 |
| ��Ҫ���ϼ� | +2 | +3 | +6��-2 | +2 | -2 | +5��-3 |
| A�� | ���⻯��ķе㣺X��T��Q | |
| B�� | ���Ӱ뾶��X2-��T2-��L2+��M3+ | |
| C�� | ��ҵ���õ������״̬��L��T�Ļ�������ȡ����L | |
| D�� | L��M������������Ӧ��ˮ���������ǿ�Ӧ������ǿ�ᷴӦ |