��Ŀ����
 �״�����Ϊ2l���͵�����ȼ�ϣ������״����й����⣮
�״�����Ϊ2l���͵�����ȼ�ϣ������״����й����⣮��1����֪�ڳ��³�ѹ�£�
2CH3OH��l��+3O2��g���T2CO2��g��+4H2O��g����H1�T-1275.6kJ/mol
2CO ��g��+O2��g���T2CO2��g����H2�T-566.0kJ/mol
H2O��g���TH2O��l����H3�T-44.0kJ/mol
�״�����ȫȼ�ս�����һ����̼��Һ̬ˮ���Ȼ�ѧ����ʽΪ
CH3OH��l��+O2��g���TCO ��g��+2H2O��l����H�T
��2����ӦCO��g��+2H2��g��?CH3OH��g�����Է����У��ɴ˿����жϡ�H
| ʵ���� | T���棩 | n ��CO��/n��H2�� | P��MPa�� | ||
| 1 | 150 |
| 0.1 | ||
| 2 |
| 5 | |||
| 3 | 350 | 5 |
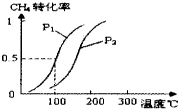
��1.0mol CH4��2.0mol H2O��g��ͨ�뷴Ӧ�ң��ݻ�Ϊ100L������һ�������·�����ӦCH4��g��+H2O��g��?CO��g��+3H2��g����CH4��ת�������¶ȡ�ѹǿ�Ĺ�ϵ����ͼ��ͼ�е�P1
���㣺�Ȼ�ѧ����ʽ,��Ӧ�Ⱥ��ʱ�,��ѧƽ��ļ���
ר�⣺
��������1��������֪���Ȼ�ѧ����ʽ��˹���ɽ��м��㣻
��2����Ӧǰ����������ڷ�Ӧ�����������������Ӧ����Ϊ�ؼ��٣���Ӧ���Է����У����ݡ�H-T?��S��0�����жϣ�
�¶���ͬ���Ƚ�ѹǿ��Ӱ�죻ѹǿ��ͬ���Ƚ��¶ȵ�Ӱ�죻
��3����ͬ�¶��£�����ѹǿ��ƽ�������������С�ķ����ƶ������ݼ���ת���ʴ�Сȷ��ѹǿ��С��
��100��ƽ��CH4��ת����Ϊ0.5����������ʽ����ƽ��ʱ����ֵ�ƽ��Ũ�ȣ�����ƽ�ⳣ������ʽ���㣮
��2����Ӧǰ����������ڷ�Ӧ�����������������Ӧ����Ϊ�ؼ��٣���Ӧ���Է����У����ݡ�H-T?��S��0�����жϣ�
�¶���ͬ���Ƚ�ѹǿ��Ӱ�죻ѹǿ��ͬ���Ƚ��¶ȵ�Ӱ�죻
��3����ͬ�¶��£�����ѹǿ��ƽ�������������С�ķ����ƶ������ݼ���ת���ʴ�Сȷ��ѹǿ��С��
��100��ƽ��CH4��ת����Ϊ0.5����������ʽ����ƽ��ʱ����ֵ�ƽ��Ũ�ȣ�����ƽ�ⳣ������ʽ���㣮
���
�⣺��1������H2O��g��=H2O��l����H1=-44.0kJ/mol
��2CO ��g��+O2��g��=2CO2��g����H2=-566.0kJ/mol
��2CH3OH��l��+3O2��g��=2CO2��g��+4H2O��g����H3=-1275.6kJ/mol��
���ݸ�˹���ɼ��㣬��-��+4���ٵõ��״�����ȫȼ������һ����̼��Һ̬ˮ���Ȼ�ѧ����ʽΪ��CH3OH��l��+O2��g��=CO��g��+2H2O��l����H=-442.8KJ/mol��
�ʴ�Ϊ��-442.8��
��2��CO��g��+2H2��g��?CH3OH��g������Ӧǰ����������ڷ�Ӧ�����������������Ӧ����Ϊ�ؼ��٣���Ӧ���Է����У����ݡ�H-T?��S��0��֪����H��0��
��ȡ���Ʊ�������̽���ϳɼ״����¶Ⱥ�ѹǿ�����������������¶ȡ�ѹǿ�DZ仯�ģ�n��CO��/n��H2�����ֲ��䣬�Ƚ�ʹ��1��2��ѹǿ��ͬ�������¶�Ӧ��ͬ��ӦΪ150�棬�ʴ�Ϊ������150��
��
��3������ͬ�¶��£�����ѹǿ��ƽ�������������С�ķ����ƶ��������ת���ʼ�С������P1��P2��
ƽ��ʱ�����Ũ�ȱ仯��Ϊ
=0.005mol/L����
CH4��g��+H2O��g��?CO��g��+3H2��g��
��ʼ��mol/L����0.01 0.02 0 0
�仯��mol/L����0.005 0.005 0.005 0.015
ƽ�⣨mol/L����0.005 0.015 0.005 0.015
��100��ʱƽ�ⳣ��k=
=2.25��10-4��
�ʴ�Ϊ������2.25��10-4��
��2CO ��g��+O2��g��=2CO2��g����H2=-566.0kJ/mol
��2CH3OH��l��+3O2��g��=2CO2��g��+4H2O��g����H3=-1275.6kJ/mol��
���ݸ�˹���ɼ��㣬��-��+4���ٵõ��״�����ȫȼ������һ����̼��Һ̬ˮ���Ȼ�ѧ����ʽΪ��CH3OH��l��+O2��g��=CO��g��+2H2O��l����H=-442.8KJ/mol��
�ʴ�Ϊ��-442.8��
��2��CO��g��+2H2��g��?CH3OH��g������Ӧǰ����������ڷ�Ӧ�����������������Ӧ����Ϊ�ؼ��٣���Ӧ���Է����У����ݡ�H-T?��S��0��֪����H��0��
��ȡ���Ʊ�������̽���ϳɼ״����¶Ⱥ�ѹǿ�����������������¶ȡ�ѹǿ�DZ仯�ģ�n��CO��/n��H2�����ֲ��䣬�Ƚ�ʹ��1��2��ѹǿ��ͬ�������¶�Ӧ��ͬ��ӦΪ150�棬�ʴ�Ϊ������150��
| 1 |
| 3 |
��3������ͬ�¶��£�����ѹǿ��ƽ�������������С�ķ����ƶ��������ת���ʼ�С������P1��P2��
ƽ��ʱ�����Ũ�ȱ仯��Ϊ
| 1mol��0.5 |
| 100L |
CH4��g��+H2O��g��?CO��g��+3H2��g��
��ʼ��mol/L����0.01 0.02 0 0
�仯��mol/L����0.005 0.005 0.005 0.015
ƽ�⣨mol/L����0.005 0.015 0.005 0.015
��100��ʱƽ�ⳣ��k=
| 0.0153��0.005 |
| 0.015��0.005 |
�ʴ�Ϊ������2.25��10-4��
���������⿼���˹���ɵ�Ӧ�á���ѧƽ���Ӱ�����ء�ƽ�ⳣ������ؼ����ʵ��̽����֪ʶ���ۺ��Խϴ�֪ʶ��㣬�ѶȽϴ�Ӧ��ǿƽʱ֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
 ������ȫ�̼����ĩ���100��ϵ�д�
������ȫ�̼����ĩ���100��ϵ�д�
�����Ŀ
����ȷ��ʾ���з�Ӧ�����ӷ���ʽ�ǣ�������
| A����̼��������Һ�м������HCO3-+H+=CO2��+H2O |
| B���ѽ���������ϡ�����У�2Fe+6H+=2Fe3++3H2�� |
| C�����Ȼ�������Һ��ͨ��������Fe2++Cl2=Fe3++2Cl- |
| D���Ƹ�ˮ��Ӧ��Na+2H2O=Na++2OH-+H2�� |
������0.2mol��Al2��SO4��3��Һ�м���1000mL KOH��Һ�����ɰ�ɫ����7.8g����KOH��Һ�����ʵ���Ũ�ȿ���Ϊ��������
| A��0.3mol/L |
| B��0.7mol/L |
| C��0.8 mol/L |
| D��1.5mol/L |
�����ȷ���ԭ���л�����Ƶý�����1mol�������������ٵ��ǣ�������
| A��MnO2�� |
| B��WO3 |
| C��Fe3O4 |
| D��Cr2O3 |
���ֽ����Ļ����15g����������ϡH2SO4����������0.5mol�������ﲻ�����ǣ�������
| A��Cu��Mg |
| B��Zn��Fe |
| C��Na��Al |
| D��Al��Fe |
����ͼ��ʾװ�ý���ʵ�飬��Һ��A��μ��뵽����B�У�����������ȷ���ǣ�������


| A����AΪŨ���ᣬBΪMnO2��C��ʢƷ����Һ����C����Һ��ɫ |
| B����AΪ���ᣬBΪ���ǣ�C��ʢ����ʯ��ˮ����C����Һ�ޱ仯 |
| C����AΪŨ��ˮ��BΪ��ʯ�ң�C��AlCl3��Һ����C���Ȳ�����ɫ������������ܽ� |
| D��ʵ������D��Ҫ��ֹ��Һ���������� |