��Ŀ����
1���о��Ƽ��仯��������Ҫ���壮��1��NaOH��ʵ��������õ��Լ�֮һ��
ʵ���ҽ���ijʵ����Ҫ0.5mol•L-1������������ҺԼ480mL��������Һ���ƵĹ��̣��ش��������⣺
��ʵ�������Ҫ������ƽ�������룩��ҩ�ס��ձ��Ͳ������⣬����Ҫ����������������500mL����ƿ����ͷ�ιܣ�
�ڸ��ݼ����֪������������ƽ�������룩ȷ����NaOH����10.0g��
�������ƹ����У���������������ȷ�ģ����в���������Ũ��ƫ�����BC��
A��ת����Һʱ������������������ƿ����
B������ʱ���ӿ̶���
C��δ��ȴ�����¾ͽ���Һת�Ƶ�����ƿ������
D�����ݺ�����ƿ������ҡ�Ⱦ��ú�Һ����ڿ̶��ߣ��ټ�ˮ���̶���
��2����֪����ˮ�ܷ�����Ӧ������ʽΪ2Na+2H2O�T2NaOH+H2�������÷�Ӧ��дΪ���ӷ���ʽΪ2Na+2H2O�T2Na++2OH-+H2����
��3��NaNO2����ۺ�ʳ�����ƣ�������ζ������ʹ����ʳ�ж���
��֪NaNO2�ܷ������·�Ӧ��2NaNO2+4HI�T2NO��+I2+2NaI+2H2O
��������Ӧ�У���������NaNO2��ÿ���ɱ�״���µ�NO����2.24L��ת�Ƶ��ӵ���ĿΪ0.1NA��
��ij������Һ�У�����2%��5%��NaNO2��ֱ���ŷŻ������Ⱦ�������Լ���ʹNaNO2ת��Ϊ�����������Ⱦ��N2����B��
A��KMnO4 B��NH4Cl C��O2��
���� ��1������������һ�����ʵ���Ũ�ȵ���Һ����ѡ����Ҫ������
������m=CVM������Ҫ���ʵ�������
�۷������������ʵ����ʵ�������Һ�����Ӱ�죬����C=$\frac{n}{V}$������������
��2�����ʡ�������������Ӧ������ѧʽ����������Ϊǿ��Ӧ���������ʽ��
��3���ٷ�����Ӧ��Ԫ�ػ��ϼ۱仯����Ӧ����������Ԫ�ػ��ϼ۽��͵�Ϊ�����������ݷ���ʽ��Ԫ�ػ��ϼ۱仯����ת�Ƶ�������
��ѡ���Լ�ʱҪ���ǣ���ʹNaNO2ת����������ȾN2���������Ʒ�����ԭ��Ӧ�����Լ��������Ӧ���л�ԭ�ԣ�
��� �⣺��1��������һ�����ʵ���Ũ�ȵ���Һ���裺���㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȣ��õ���������������ƽ�������룩��ҩ�ס��ձ�����������500mL����ƿ����ͷ�ιܣ����Ի�ȱ�ٵ�������500mL����ƿ����ͷ�ιܣ�
�ʴ�Ϊ��500mL����ƿ����ͷ�ιܣ�
����Ҫ0.5mol•L-1������������ҺԼ480mL��Ӧѡ��500mL����ƿ��ʵ������500mL��Һ����Ҫ���ʵ�����m=0.5mol/L��40g/mol��0.5L=10.0g��
�ʴ�Ϊ��10.0��
��A��ת����Һʱ������������������ƿ���棬�������ʵ����ʵ���ƫС��������ҺŨ��ƫ�ͣ���A��ѡ��
B������ʱ���ӿ̶��ߣ�������Һ���ƫС����ҺŨ��ƫ�ߣ���Bѡ��
C��δ��ȴ�����¾ͽ���Һת�Ƶ�����ƿ�����ݣ���ȴ����Һ����ƫС����ҺŨ��ƫ�ߣ���Cѡ��
D�����ݺ�����ƿ������ҡ�Ⱦ��ú�Һ����ڿ̶��ߣ��ټ�ˮ���̶��ߣ�������Һ���ƫ����ҺŨ��ƫ�ͣ���D��ѡ��
��ѡ��BC��
��2������ˮ�ܷ�����Ӧ������ʽΪ2Na+2H2O�T2NaOH+H2�������÷�Ӧ��дΪ���ӷ���ʽΪ 2Na+2H2O�T2Na++2OH-+H2����
�ʴ�Ϊ��2Na+2H2O�T2Na++2OH-+H2����
��3����2NaNO2+4HI�T2NO��+I2+2NaI+2H2O����Ӧ������������+3�۵�N���ϼ۽��ͣ�����������������������
��Ӧ������2molNO��ת��2mol���ӣ���ÿ���ɱ�״���µ�NO����2.24L�����ʵ���$\frac{2.24L}{22.4L/mol}$=0.1mol��ת�Ƶ��ӵ���Ŀ0.1NA��
�ʴ�Ϊ��0.1NA��
��NaNO2��N2�DZ���ԭ������ӻ�ԭ�����������������ǿ�������ԣ��Ȼ�茶��л�ԭ�ԣ�
��ѡ��B��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ơ�������ԭ��Ӧ����ȷ����ԭ������������Ϥ������ԭ��Ӧ�����ǽ���ؼ���ע������ƿ����ѡ���ʹ�÷�������Ŀ�ѶȲ���

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ��������ʴ��������֪����Ҳ��������ʴ���� | |
| B�� | ��Na20���������Ӹ�����Ϊ1��2������֪Na202���������Ӹ�����Ϊ1��1 | |
| C�� | �������巴Ӧ�����廯��������֪���͵ⵥ�ʷ�Ӧ���ɵ⻯�� | |
| D�� | ��������ˮ���ҷ�Ӧ������֪��Ҳ������ˮ���ҷ�Ӧ |
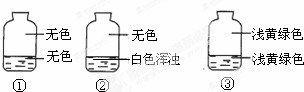 ��ʢ����������������ƿ�У��ֱ�ע��Լ���֮һ������Һ�岢���۲���ͼ����¼�������жϸ�ƿע���Һ��������ʲô����������
��ʢ����������������ƿ�У��ֱ�ע��Լ���֮һ������Һ�岢���۲���ͼ����¼�������жϸ�ƿע���Һ��������ʲô����������| A�� | AgNO3��Һ��NaOH��Һ��ˮ | B�� | NaOH��Һ��AgNO3��Һ��ˮ | ||
| C�� | ˮ��AgNO3��Һ��NaOH��Һ | D�� | AgNO3��Һ��ˮ��NaOH��Һ |
| A�� | �ס��һ�ѧƽ���ƶ�����ͬ | B�� | �ס���ƽ����NO2�����������ͬ | ||
| C�� | �ס���ƽ����N2O4����������ף��� | D�� | �ס�������ʱ�������棩������������С |
��֪�״��Ʊ����йػ�ѧ��Ӧ�Լ��ڲ�ͬ�¶��µĻ�ѧ��Ӧƽ�ⳣ�����±���ʾ��
| ��ѧ��Ӧ | ƽ�ⳣ�� | �¶ȡ� | |
| 500 | 800 | ||
| ��2H2��g��+CO��g��?CH3OH��g�� | K1 | 2.5 | 0.15 |
| ��2H2��g��+CO2��g��?H2O+CO��g�� | K2 | 1.0 | 2.50 |
| ��3H2��g��+CO2��g��?CH3OH��g��+H2O��g�� | K3 | ||
��2��ij�¶��·�Ӧ����H2��ƽ��ת���ʣ�a������ϵ��ѹǿ��P���Ĺ�ϵ��ͼ1��ʾ����ƽ��״̬��A�䵽Bʱ��ƽ�ⳣ��K��A��=K��B�������������������=������
�ݷ�Ӧ����ڿ��Ƶ���K1��K2��K3֮��Ĺ�ϵ����K3=K1•K2����K1��K2��ʾ����

��3����3L�ݻ��ɱ���ܱ������з�����Ӧ�ڣ���֪c��CO���뷴Ӧʱ��t�仯���ߢ���ͼ2��ʾ������t0ʱ�̷ֱ�ı�һ�������������ߢ�ɱ�Ϊ���ߢ�����ߢ����ߢ��Ϊ���ߢ�ʱ���ı�������Ǽ�������������ߢ��Ϊ���ߢ�ʱ���ı�������ǽ���������������٣�ѹ����2L��
��4���״�ȼ�ϵ�����Ź㷺����;�������ò�Ϊ�缫���ϣ������Ϸֱ�ͨ��״���������������������ҺΪ�������Һ����ü���ȼ�ϵ�صĸ�����Ӧʽ��CH3OH-6e-+8OH-=CO32-+6H2O��
| A�� | 6 | B�� | 7 | C�� | 10 | D�� | 14 |
| A�� | �Ʊ�Al��OH��3����Һ����1mol•L-1AlCl3��Һ�мӹ�����6mol•L-1NaOH��Һ | |
| B�� | �ᴿ���������������������������������������������м����������̼������Һ�����÷�Һ������ȥ�л����ˮ | |
| C�� | ������Һ���Ƿ���Fe3+��ȡ������������Һ�������м�������������ˮ���ٵμ�KSCN��Һ���۲�ʵ������ | |
| D�� | ̽��������H2O2�ֽ����ʵ�Ӱ�죺����ͬ�����£���һ�Թ��м���2mL5%H2O2��1mLH2O������һ�Թ��м���2mL 10%H2O2��1mLFeCl3��Һ���۲첢�Ƚ�ʵ������ |
| A�� | 1mol H2O���е�ԭ����ΪNA | |
| B�� | 4gH2�����������4NA | |
| C�� | ���³�ѹ�£�11.2L O2�к��з�����Ϊ0.5NA | |
| D�� | 1L 0.1 mol•L-1NaCl��Һ�к�Na+��0.1NA |
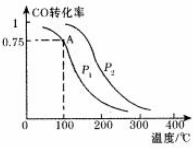 CO��SO2����Ҫ�Ĵ�����Ⱦ���壬���û�ѧ��Ӧԭ����������Ⱦ����Ҫ������
CO��SO2����Ҫ�Ĵ�����Ⱦ���壬���û�ѧ��Ӧԭ����������Ⱦ����Ҫ������