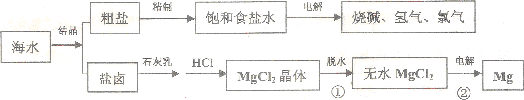
��Ŀ����
14����ѧ��ѧ�����ĵζ��������к͵ζ�����������ԭ�ζ����ȣ�I����1��̽��С���������KMnO4��Һ�ζ�ij��Ѫ��[��Ҫ�ɷ������������壨FeSO4•7H2O��]����Ԫ�صĺ�����
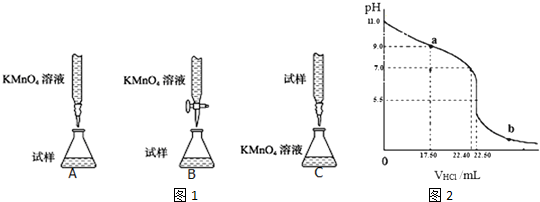
�����еζ�װ���У��гֲ�����ȥ������ͼ1����������ǣ�B������ĸ��ţ���
��ʵ��ǰ������Ҫȷ����һ�����ʵ���Ũ�ȵ�����KMnO4��Һ250mL������ʱ��Ҫ��������������ƽ��ҩ�ס����������ձ�����Ͳ�⣬����Ҫ��ͷ�ιܡ�250mL����ƿ�����������ƣ���
��̽��С���ҲⶨNH3•H2O��Ũ�ȼ�����ƽ�ⳣ��Kb
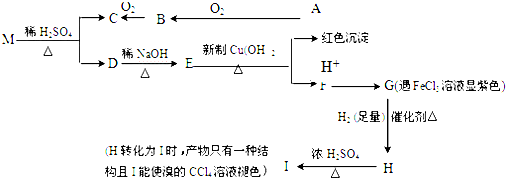
�����£�̽��С����ȡ25.00mL��ˮ����ƿ�У���0.0500mol•L-1HCl�ζ�����pH�Ʋɼ����ݣ���Һ��pH�������������ı仯������ͼ2��ʾ��
��2����ͼ2��������ϡ��������Ϊ22.40mLʱ����ʱ��Һ��c��Cl-��=c��NH4+�����������=����
������ϡ����������b����Һ������Ũ�ȴ�С��ϵΪc��Cl-����c��NH4+������H+����c��OH-����
��4�����ͼ��Ϣ�����㰱ˮ��Ũ��Ϊ0.0448mol•L-1��
��5�����ڸõζ�ʵ���˵���У���ȷ����AC��
A����ƿ������������ˮ��Ӱ��ⶨ���
B����ƿδ�ð�ˮ������ϴ�ᵼ�²�ð�ˮ��Ũ��ƫ��
C����ʽ�ζ���δ��������ϴ�ᵼ�²�ð�ˮ��Ũ��ƫ��
D���ζ��յ�ʱ���Ӷ����ᵼ�²�ð�ˮ��Ũ��ƫ�ߣ�
���� ��1���ٸ�����ؾ���ǿ�����ԣ��������ܣ������ü�ʽ�ζ��ܣ�
��������Һ�IJ����У����㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȵȣ��������Ʋ����ж�ʹ�õ�������Ȼ��ȷ����ȱ�ٵ�������
��2��������ϡ��������Ϊ22.40mL��pH=7���ɵ���غ㣺c��Cl-��+c��OH-��=c��NH4+��+��H+�������b�������������Һ�е�����ΪHCl��NH4Cl����Һ�����ԣ����ݵ����ˮ�������
��4��������ϡ��������Ϊ22.40mL��pH=7��c��Cl-��=c��NH4+�������������ӵ����ʵ������㣻
��5��A����ƿ������������ˮ��Ӱ�찱ˮ�����ʵ�����
B����ƿ������ϴ��
C����ʽ�ζ���δ��������ϴ�ᵼ������Ũ��ƫ�ͣ�
D���ζ��յ�ʱ���Ӷ����ᵼ���������ʵ���ƫС��
��� �⣺��1����������KMnO4��Һ�ζ�ij��Ѫ����������ؾ���ǿ�����ԣ��������ܣ������ü�ʽ�ζ��ܣ�����B���ϣ�
��ѡ��B��
������һ�����ʵ���Ũ�ȵ�����KMnO4��Һ250mL�IJ���Ϊ�����㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ�����ձ����ܽ⣨������Ͳ��ȡˮ������ȴ��ת�Ƶ�250mL����ƿ�У����ò�����������ϴ���ձ���������2��3�Σ���ϴ��Һ��������ƿ����ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�������Ҫ������Ϊ������ƽ��ҩ�ס����������ձ�����Ͳ�����ÿɲ��ã���250mL����ƿ����ͷ�ιܣ�������ʹ�õIJ�������Ϊ����ͷ�ιܡ�250mL����ƿ��
�ʴ�Ϊ����ͷ�ιܡ�250mL����ƿ��
��2��������ϡ��������Ϊ22.40mLʱ����Һ�����ԣ���c��OH-��=��H+�����ɵ���غ�c��Cl-��+c��OH-��=c��NH4+��+��H+����֪��c��Cl-��=c��NH4+����b�������������Һ�е�����ΪHCl��NH4Cl����Һ�����ԣ�����Һ������Ũ�ȴ�С��ϵΪ��c��Cl-����c��NH4+������H+����c��OH-����
�ʴ�Ϊ��=��c��Cl-����c��NH4+������H+����c��OH-����
��4��������ϡ��������Ϊ22.40mL��pH=7��c��Cl-��=c��NH4+�������ߵ����ʵ�����ȣ������ӵ����ʵ���Ϊ0.0224L��0.0500mol•L-1����笠����ӵ����ʵ�������ˮ�����ʵ���Ϊ0.0224L��0.0500mol•L-1������c��NH3•H2O��=$\frac{0.0224L��0.05mol/L}{0.025L}$=0.0448mol/L��
�ʴ�Ϊ��0.0448��
��4��A����ƿ������������ˮ��Ӱ�찱ˮ�����ʵ��������Բ�Ӱ��ⶨ�������A��ȷ��
B����ƿ������ϴ����ϴ��ƿ����ʹ��ƿ�а�ˮ�����ʵ���ƫ��û��ϴ�Բⶨ�����Ӱ�죬��B����
C����ʽ�ζ���δ��������ϴ�ᵼ������Ũ��ƫ�ͣ���ʹ����������ƫ�����Ե��²ⶨ���ƫ�ߣ���C��ȷ��
D���ζ��յ�ʱ���Ӷ����ᵼ���������ƫС�����������ʵ���ƫС�����Ե��²ⶨƫ�ͣ���D����
��ѡAC��
���� ���⿼�����к͵ζ�����������ԭ�ζ��������ؿ���ѧ���������жϼ�ʵ�������������ȷ�ζ�ԭ����������ʵ����֪ʶ���ǽⱾ��ؼ�����Ŀ�Ѷ��еȣ�

 �������Ͽ�ʱͬ��ѵ��ϵ�д�
�������Ͽ�ʱͬ��ѵ��ϵ�д� �������ͬ����ϰϵ�д�
�������ͬ����ϰϵ�д�| A�� | 115.75mL | B�� | 134.48mL | C�� | 143.75mL | D�� | 156.8mL |
| A�� | �ζ�ǰ�ζ����������ݣ��ζ�����ʧ | |
| B�� | ��ʽ�ζ�����ȡNaOH��Һʱ��δ������ϴ���� | |
| C�� | �ζ�ʱ�ﵽ�ζ��յ�ʱ���Ӷ��� | |
| D�� | ��ƿȡ��NaOH����Һǰ������ˮϴ�� |
| A�� | �������ĸ�ԭ�ӹ�ֱ�ߣ���CΪSP�ӻ� | |
| B�� | ������N��C���ļ�������C-C���ļ��� | |
| C�� | �����к���2���Ҽ���4���м� | |
| D�� | ��������������Һ������Ӧ |
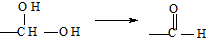
 ����H��I��
����H��I�� ��
�� ��
��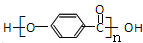 ��
�� �ȣ�����дһ�֣�
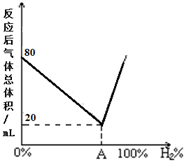
�ȣ�����дһ�֣�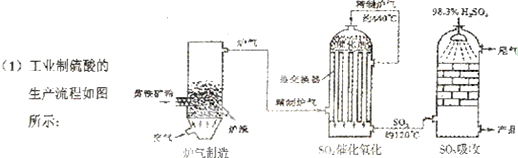
 ��ij��������ʽΪC6H14������������������Ȳ���������ӳɵõ�����������Ľṹ��ʽΪ��CH3��2CHCH2CH2CH3��
��ij��������ʽΪC6H14������������������Ȳ���������ӳɵõ�����������Ľṹ��ʽΪ��CH3��2CHCH2CH2CH3��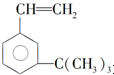 ���������ʣ�
���������ʣ� ��
��