��Ŀ����
11��ij��ѧС����ʵ��������CaSO4��NH3��CO2�Ʊ���NH4��2SO4���乤���������£�
�ش��������⣺
��1�������������Ϊ���ˣ�������һϵ�в�����������Ũ������ȴ�ᾧ�����ˣ�
��2��X����ΪNH3���ѧʽ����ͬ����Y����ΪCO2����ѭ�����õ����ʵ���NH3��CO2��
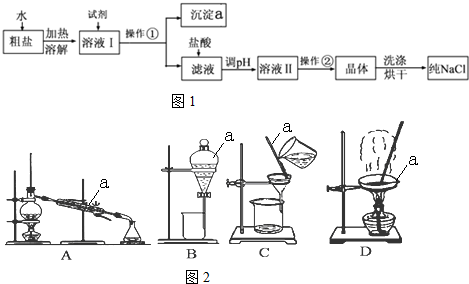
��3��ͼ2��װ�ò�������ʵ�����ư������Ǽף�����ţ���
ѡ��������ȡװ�ú�����ͼ3װ���ռ�����İ���������ȡ�������Һ�����ӵ�˳���ýӿ������ĸ��ʾ���ǣ�a��d��e��g��f��c��b��h��
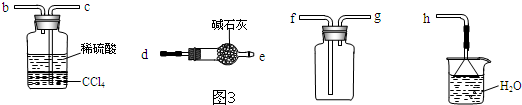
����װ����CCl4�������Ƿ�������
��4��Ҫ�ⶨ���Ƶõ�����林��ȣ�ȡ10.0g��Ʒ����ȫ����ˮ������Һ�еμӹ������Ȼ�����Һ�����ˡ�ϴ�ӡ������������������Ϊ13.98g�����Ƶ�����淋Ĵ���Ϊ79.2%��
���� ���������Һͨ�������İ�����ʹ��Һ�ɼ��ԣ���ͨ�����Ķ�����̼������Һ�еİ�ˮ�����������Һ��Ӧ����̼��Ƴ������������Һ���������˷���õ�̼��Ƴ���������������ʯ�ң�����������茶�������Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����õ�����茶��壻
��1���������ʵķ����ᴿ������
��2������������CO2���ܽ�Ⱥ�С�����ǰ���������ˮ���õ��ļ�����Һ�Ͷ�����̼��Ӧ���ܰ�CaSO4��ȫת��ΪCaCO3��
��3��������ʵ�����Ʊ���������β�����գ�
��4���ⶨ���Ƶõ�����林��ȣ�ʹ�����������ȫ�������������淋����ʵ�����������
��� �⣺���������Һͨ�������İ�����ʹ��Һ�ɼ��ԣ���ͨ�����Ķ�����̼������Һ�еİ�ˮ�����������Һ��Ӧ����̼��Ƴ������������Һ���������˷���õ�̼��Ƴ���������������ʯ�ң�����������茶�������Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����õ�����茶��壻
��1���ɷ���֪��BΪ̼��Ƴ������������Һ��������ߵ�ʵ�����ӦΪ�����ˣ���Һ�������Һ��ʹ���������Һ�������ķ���Ϊ���ȼ����Ƴɱ�����Һ
������Ũ�����ٽ�����ȴ���ᾧ��������ȴ�ᾧ�������ˣ�
�ʴ�Ϊ�����ˣ���ȴ�ᾧ��
��2������������CO2���ܽ�Ⱥ�С�����ǰ���������ˮ���õ��ļ�����Һ�Ͷ�����̼��Ӧ���ܰ�CaSO4��ȫת��ΪCaCO3����CaSO4����Һ�м���NH3������Ӧ���£�CaSO4+2NH3+2H2O?Ca��OH��2+��NH4��2SO4�������м���CO2��������CaCO3��ƽ�����������ƶ��������ڣ�NH4��2SO4�����ɣ���XΪNH3��YΪCO2�����ݷ�Ӧ���̵�֪����������������NH3��CO2��Ҳ��Ҫʹ��NH3��CO2����ѭ�����õ����ʵ���NH3��CO2��
�ʴ�Ϊ��NH3��CO2�� NH3��CO2��
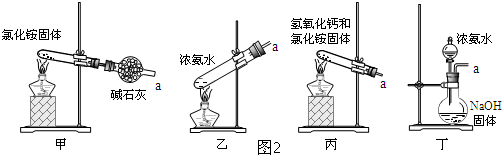
��3���ף��Ȼ�����ȷֽ�ɰ������Ȼ��⣬���������Ȼ���һ��ȴ�����ֿ��Ի��ϳ��Ȼ�泥�������������ư�����
�ң���ˮ�д���ƽ���ϵ��NH3+H2O�TNH3•H2O�TNH4++OH-������Ũ��ˮ�������ܽ�Ƚ��ͣ�����Һ���ݳ���ƽ�����淴Ӧ�����ƶ���������ȡ������
������ʯ�Һ��Ȼ�炙�Ϻ���ȿ�����ȡ��������Ӧ�Ļ�ѧ����ʽΪ��2NH4Cl+Ca��OH��2$\frac{\underline{\;\;��\;\;}}{\;}$CaCl2+2NH3��+2H2O��������ȡ������
��������CaO��H2O���ҷ�Ӧ������Ca��OH��2���ų������ȣ��ٽ�NH3•H2O�ķֽ⼰NH3�Ļӷ��ݳ���������Ca��OH��2��ǿ�Ҳ�ٽ�NH3•H2O�ķֽ⼰NH3�Ļӷ��ݳ�����Ӧ�Ļ�ѧ����ʽΪ��NH3•H2O+CaO�TNH3��+Ca��OH��2��������ȡ��������ѡ�ף�
ѡ��������ȡװ�ú�����ͼ3װ���ռ�����İ���������ȡ�������Һ��˳��Ϊ������װ�á�����װ�á�����װ�á�����װ�á�β������װ�ã����ӵ�˳���ýӿ������ĸ��ʾ���ǣ�de��gf��cb��h��
����װ���У�ˮ���ܶȱ����Ȼ�̼С���������Ȼ�̼�����棬���Ȼ�̼���ܺͰ�����Ӧ��Ҳ���ܽⰱ������װ�ü��ܹ��������������壬���ܹ���ֹ������
�ʴ�Ϊ���ף�d��e��g��f��c��b��h����������
��4��Ҫ�ⶨ���Ƶõ�����林��ȣ�ȡ10.0g��Ʒ����ȫ����ˮ������Һ�еμӹ������Ȼ�����Һ��Ŀ����ʹ�����������ȫ�������������ϲ���Һ�ٵμ��Ȼ����Ͳ����г������ɣ����Լ����Ȼ����Ƿ��ѹ�����
����Һ�еμӹ������Ȼ�����Һ�����ˡ�ϴ�ӡ������Ĺ���Ϊ���ᱵ������Ϊ13.98g��n��BaSO4��=$\frac{m}{M}$=$\frac{13.98g}{233g/mol}$=0.06mol��n����NH4��2SO4��=n��SO42+��=n��BaSO4��=0.06mol��m����NH4��2SO4��=n•M=0.06mol��132g/mol=7.92g������淋Ĵ���=$\frac{7.92g}{10.0g}$��100%=79.2%��
�ʴ�Ϊ��79.2%��
���� ����Ϊ���������⣬�漰ԭ�ϡ���Ʒ���жϡ�����ʽ����д�����顢������Ʊ��ȣ�����ʱҪ���������������Ϣ������Լ���ѧ����֪ʶ�������������Ե��Ʋ⡢���㣬�ѶȽϴ�

 ����˼ά����ѵ����ʱ��ѧ��ϵ�д�
����˼ά����ѵ����ʱ��ѧ��ϵ�д�| ��� | �� | �� | �� | �� | �� | �� |
| ������װ��ͼ |  |  |  |  |  |  |
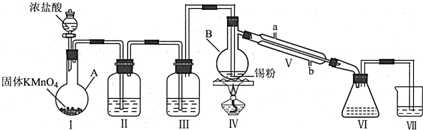
��2��װ�â��з�����Ӧ�Ļ�ѧ����ʽ��MnO2+4HCl$\frac{\underline{\;\;��\;\;}}{\;}$MnCl2+2Cl2��+2H2O��
��3��ʵ�鿪ʼʱ��Ӧ�ȵ�ȼ�ݣ���װ����ţ����ľƾ��ƣ�����CuCl2�к���CuO���ѧʽ�����ʣ�����ַ�Ӧ��װ�âٵIJ�������ʣ������ػ�ɫ��
��4��ijʵ��С����װװ��ʱ���ڢٺ͢�֮��������һ��ʢ��Ũ�����ϴ��ƿ���������Ƿ�ֹ����������Һ�е�ˮ��������װ�â٣�
| A�� | �ܽ����ʱ��Ӧ��������ҺϡЩ����ֹʳ�β���ȫ�ܽ� | |
| B�� | ��ȥ���������ʺ���Һ���������ڼ���Ũ�� | |
| C�� | ���Ƶõľ���ת�Ƶ����ƹ��������ô���ˮ����ϴ�� | |
| D�� | ��������ʣ������Һ��ʱ��ֹͣ���ȣ��������Ƚ�Һ������ |
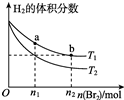 �����ΪV L�ĺ����ܱ�������ʢ��һ����H2��ͨ��Br2��g��������Ӧ��H2��g��+Br2��g��?2HBr��g����H��0�����¶ȷֱ�ΪT1��T2����ƽ��ʱ��H2�����������Br2��g�������ʵ����仯��ϵ��ͼ��ʾ������˵����ȷ���ǣ�������
�����ΪV L�ĺ����ܱ�������ʢ��һ����H2��ͨ��Br2��g��������Ӧ��H2��g��+Br2��g��?2HBr��g����H��0�����¶ȷֱ�ΪT1��T2����ƽ��ʱ��H2�����������Br2��g�������ʵ����仯��ϵ��ͼ��ʾ������˵����ȷ���ǣ�������| A�� | ��ͼ��֪��T1��T2 | |
| B�� | a��b���������Ӧ���ʣ�b��a | |
| C�� | Ϊ�����Br2��g����ת���ʣ�����������������ʱ���������ΪV/2 L | |
| D�� | T1ʱ������Br2��g�����룬ƽ��ʱHBr����������������� |
| A�� | 2NO2?N2O4 ������ӦΪ���ȷ�Ӧ�� | |
| B�� | C��s��+CO2?2CO ������ӦΪ���ȷ�Ӧ�� | |
| C�� | N2+3H2?2NH3 ������ӦΪ���ȷ�Ӧ�� | |
| D�� | H2S?H2+S��s�� ������ӦΪ���ȷ�Ӧ�� |