��Ŀ����
��֪�Ҵ����Ժ���ˮCaCl2��Ӧ��������ˮ��CaCl2??6C2H5OH ��
�й��Լ��ķе����£�CH3COOC2H5 77.1�棻 C2H5OC2H5 34.5�棻
C2H5OH 78.3�棻 CH3COOH 118�档
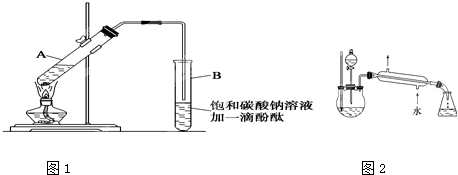
ij����С��������ͼ��ʾװ���Ʊ����������ֲ�Ʒ���Իش��������⣺
��1��������ƿ�м�����Լ��ǣ� �����ᡢ�ƾ���Ũ���ᣬ
����ŨH2SO4�������� �� ��
��2������������������������� �� ��ȥ�ͷе����֡��ռ��е���76��~120��
֮�����֣������㵹�뱥��Na2CO3��Һ�й۲쵽�������ǣ� ��
����ʵ���б���̼������Һ��������_____������ţ�
A���к�������Ҵ��� B���к����Ტ�ܽ��Ҵ���
C�������������ɣ�������IJ��� D�������ڷֲ�������
��3��д������ʵ������CH3COOH��CH3CH218OH��Ũ�����Ʊ����������Ļ�ѧ����ʽ��
��
��4��������ֲ�Ʒ�����������ټ�����ˮCaCl2�������Ŀ���ǣ�
Ϊ�˸�������������ѡ�õ���Ѹ����Ϊ ________ ������ţ�
A������������ B����ˮ������ C����ʯ�� D��������������
��1������, ��ˮ��(��д��ˮ��) (�� 2�֣�
(2) C2H5OC2H5 ��2�֣� �ֲ�,�ϲ�Ϊ��ɫ��״Һ�壨2�֣���������֣� BD( 2�֣�
(3) CH3COOH + CH3CH218OH![]() CH3CO18O CH2CH3+H2O( 2�֣�
CH3CO18O CH2CH3+H2O( 2�֣�
(4) ��ȥ�ֲ�Ʒ�е��Ҵ� ��1�֣� B��1�֣�

 CH3COOCH2CH3+H2O
CH3COOCH2CH3+H2O