��Ŀ����
12����ҵ���������ɷ�Ϊ������������������ȡ����������ͼ��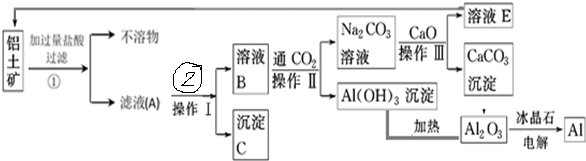
��ش��������⣺
��1�����������������õ��IJ����������ձ���©������������
��2��д����ҺA��һ�����������NaOH��Һ��Ӧ������ҺB�е����ӷ���ʽAl3++4OH-=AlO2-+2H2O��
��3�����������û�й��˲�����ֱ�ӽ��벽��ڣ��Ժ��������������Ӱ���Dz���ٲ�����SiO2�ڲ�����б�NaOH�ܽ⣬���õ���Al2O3�к���SiO2��
��4��д������ҺB����Al��OH��3�����ӷ���ʽ��2AlO2-+CO2+3H2O�T2Al��OH��3��+CO32-��
��5�������������漰������ԭ��Ӧ�Ļ�ѧ����ʽΪ2Al2O3�����ڣ�$\frac{\underline{\;����ʯ\;}}{���}$4Al+3O2����
��6�����������У���NaOH��H2O����ѭ��ʹ���⣬������ѭ��ʹ�õ�������CaO��CO2���ѧʽ����
�ô˷���ȡ���ĸ���Ʒ��Fe2O3��O2���ѧʽ����
���� ���������������������������������м������������Һ��������ӦAl2O3+6H+�T2Al3++3H2O������������������Һ��Fe2O3+6H+=2Fe3++3H2O��������ӦȻ����ˣ��õ���ҺA����AlCl3��FeCl3���������������������Һ�����˵õ���ҺBΪNaOH��NaAlO2�����Һ������CΪFe��OH��3������ҺB��ͨ��CO2��������Ӧ2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��2OH-+CO2=CO32-+H2O��Ȼ����˵õ�����Al��OH��3��Na2CO3��Һ������Һ�м���CaO��������ӦNa2CO3+CaO+H2O�T2NaOH+CaCO3����Ȼ���Ϊ���ˣ����˵õ�����CaCO3����ҺEΪNaOH����NaOHѭ�����ã���Al��OH��3���ȵõ�Al2O3���������Al2O3�õ�Al��
��1�����������������ǹ��ˣ��Դ��ж�ʹ��������
��2����ҺA��һ������AlCl3�����NaOH��Һ��Ӧ������ҺB������ƫ�����ƣ�
��3��������SiO2�ܱ�NaOH�ܽ⣻
��4����ҺBΪNaOH��NaAlO2�����Һ����Һ��ͨ��CO2����Al��OH��3������Na2CO3��
��5��ֻ�е������Al2O3�ķ�Ӧ����������ԭ��Ӧ��
��6�����ɵ�CaCO3���ȷֽ�õ�CaO��CO2���ɼ�CaO��CO2��ѭ��ʹ�ã����������õ�Fe2O3�͵������Al2O3�õ���O2Ϊ����Ʒ��
��� �⣺��1�����������������ǹ��ˣ�������Ҫ�ձ���©������������
�ʴ�Ϊ���ձ�����ͨ©������������
��2����ҺA��һ������AlCl3�����NaOH��Һ��Ӧ������ҺB������ƫ�����ƣ������ӷ���ʽΪAl3++4OH-=AlO2-+2H2O��
�ʴ�Ϊ��Al3++4OH-=AlO2-+2H2O��
��3������ٲ�����SiO2�ڲ�����б�NaOH�ܽ⣬���õ���Al2O3�к���SiO2����������������
�ʴ�Ϊ������ٲ�����SiO2�ڲ�����б�NaOH�ܽ⣬���õ���Al2O3�к���SiO2��
��4����ҺBΪNaOH��NaAlO2�����Һ������ҺB��ͨ��CO2��������Ӧ2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��2OH-+CO2=CO32-+H2O������ҺB����Al��OH��3�����ӷ���ʽ2AlO2-+CO2+3H2O�T2Al��OH��3��+CO32-��
�ʴ�Ϊ��2AlO2-+CO2+3H2O�T2Al��OH��3��+CO32-��
��5��ֻ�е������Al2O3�ķ�Ӧ����������ԭ��Ӧ����Ӧ�ķ���ʽΪ2Al2O3�����ڣ� $\frac{\underline{\;����ʯ\;}}{ͨ��}$4Al+3O2����
�ʴ�Ϊ��2Al2O3�����ڣ�$\frac{\underline{\;����ʯ\;}}{ͨ��}$4Al+3O2����
��6�����ɵ�CaCO3���ȷֽ�õ�CaO��CO2���ɼ�CaO��CO2��ѭ��ʹ�ã����������õ�Fe2O3�͵������Al2O3�õ���O2Ϊ����Ʒ��
�ʴ�Ϊ��CaO��CO2��Fe2O3��O2��
���� ���⿼�����ʷ�����ᴿ�����ؿ���ѧ��������֪ʶ������������ȷ�������ʼ�����������������Ӧԭ�����ɽ��֪������Ԫ�ػ�����֪ʶ���������Ӽ��鷽������Ŀ�Ѷ��еȣ�

| A�� | ����֪Ԫ���о�������ԭ�Ӱ뾶 | B�� | �ڿ�����ȼ��ʱ����������Fr2O | ||
| C�� | �������Ӧ��ˮ�����Ǽ�ǿ�ļ� | D�� | �䵥�ʵ��۵�Ƚ����Ƶ��۵�� |
| A�� | NH4Cl�TNH3+HCl | B�� | H2+Cl2�T2HCl | ||
| C�� | N2+3H2�T2NH3 | D�� | 2Na2O2+2H2O�T4NaOH+O2�� |
| A�� | ԭ�Ӱ뾶��Y��Z��R��T | |
| B�� | XR2��WR2����������R�Ļ��ϼ���ͬ | |
| C�� | ��̬�⻯����ȶ��ԣ�W��R��T | |
| D�� | ����������Ӧ��ˮ������ԣ�X��Z |
| A�� | ���������� | |
| B�� | �������������Сһ�� | |
| C�� | ѹǿ���䣬���뵪��ʹ����������� | |
| D�� | ������� |
| A�� | 0.5 molO2��Լ����6.02��1023��Oԭ�� | |
| B�� | 1 mol H2SO4��������98g/mol | |
| C�� | 0.5 molNa2CO3���1.5 mol CO32- | |
| D�� | H2SO4��Ħ������Ϊ98g |
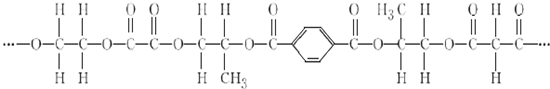
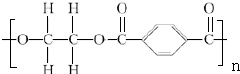 +2nH2O��
+2nH2O��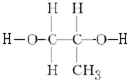 ��
��