��Ŀ����
��20�֣���18.4mol��L-1��Ũ���ᣬ���� 100 ml 1.0 mol��L-1��ϡ���ᣬ��ش��������⣺
��1����Ҫ18.4mol��L-1��Ũ���� ml��
��2��������������������ʱ����Ҫ�õ� �� ��
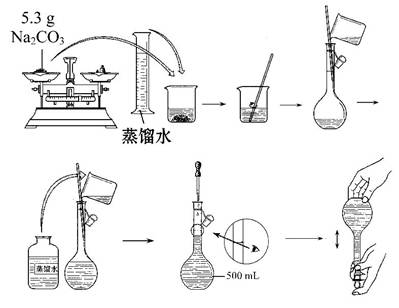
��������ƽ �ڷ�Һ©�� ��250ml����ƿ ���ձ� �ݽ�ͷ�ι�
����Ͳ �߲�����������̨�������У� ��100ml����ƿ
A���ۢܢݢߢ� B���٢ڢݢޢ� C���٢ڢۢ� D���ۢܢݢ�
��3������ʵ�鲽���У���ȷ�IJ���˳��Ӧ���ǣ�
A ����Ͳ��ȡŨ���ᣬ��������װ��Լ50ml����ˮ���ձ�����ò��������衣
B ��Լ30ml����ˮ���ֳ�����ϴ���ձ��Ͳ���������ÿ��ϴ��Һ����������ƿ�У�
C ��ϡ�ͺ������С�ĵص�������ƿ�У�
D.���100ml����ƿƿ���Ƿ���©Һ����
E.������ˮֱ�Ӽ�������ƿ����Һ��ӽ��̶���1����2cm����
F.�ǽ�ƿ���������ߵ���ҡ����Һ��
G.�ý�ͷ�ι�������ƿ����μ�������ˮ����Һ����͵���̶������У�
��4������A���������ʱ��Ӧ��ѡ��
��10 ml��Ͳ ��50 ml��Ͳ ��5000 ml��Ͳ ��1000 ml��Ͳ
��5������A���������� ���ܽ���C��������
��1��5.4
��2��C
��3��D A C B E G F
��4��1
��5������Һ��ȴ������
���������������1�����ܶȦ�=1.84g?mL-1����������Ϊ98%��Ũ�����Ũ��Ϊ��c=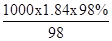 =18.4mol/L
=18.4mol/L
����100mL1mol?L-1��ϡ���ᣬ��Ҫ��Ũ��������Ϊ��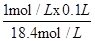 ��0.0054L=5.4ml���𰸣�5.4��
��0.0054L=5.4ml���𰸣�5.4��
��2������100mL1mol/L��ϡ����IJ���Ϊ���������ȡ��ϡ�͡���ȴ����Һ�����ݡ�ҡ�ȡ�װƿ����
ǩ����Ҫʹ�õ������У��ձ� ����Ͳ ��100mL����ƿ��������������ʱ����ʹ�ý�ͷ�ιܣ����Ի�ȱ
�ٵ�����Ϊ��ͷ�ιܣ���Ҫ�õ����Ǣ٢ڢۢ࣬��ѡC��
ʵ��������ȼ������ƿ�Ƿ�©Һ��Ȼ����ݼ������ȡ��ϡ�͡���ȴ����Һ�����ݡ�ҡ�ȡ�װ
ƿ����ǩ�ò���������Һ����ΪDACGEBF��
ͨ��������ҪŨ����5.4ml������ѡȡ��Ͳ�����������ԭ��Ӧѡ��10ml����Ͳ����ѡ�٣�
��5����δ��ȴ�����¼���������ƿ�У���ʹ���������Ũ��ƫ�𰸵���Һ��ȴ�����£��𰸣���
��Һ��ȴ������

��ѧ�����ɳ�����չ������ء�������������������
| A��CO2�ϳɿɽ���ľ�̼���������ϣ�ʵ�֡�̼ѭ���� |
| B���ᳫ���С������г����˹������ȡ���̼�����з�ʽ |
| C�����ϵ������ֹ�ؽ�����Ⱦ |
| D���������ø�������Դ�����ٶԻ�ʯȼ�ϵ����������Խ��Ϳ�����PM2.5�ĺ��� |
��17�֣���ѧ̽��
ijѧϰС�����ͼ����ʾ�ĵ��ˮԭ������ʵ�飬����ʵ��õ�����������ϻ�õ����ݽ��д�������������������һ�����̽�����밴Ҫ����д��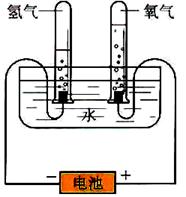
��1���۲첻ͬʱ���Թ��ڵ�H2��O2����ı�ֵ��ԼΪ ��
��2����������1.8g H2O�����ݵ��ˮ�Ļ�ѧ����ʽ��������H2��O2������������������£�
��������£�
| | ����g | ���ʵ���mol | H2��O2���ʵ����ı� |
| H2 | | | |
| O2 | | |
��3���±��г���һЩ���ʵ����
| ���� | ���� | 1 mol���ʵ���� |
| 0��101kPa | H2 | 22.3 L |
| O2 | 22.4 L | |
| CO2 | 22.4 L | |
| 25��101kPa | H2 | 24.4L |
| O2 | 24.5L | |
| CO2 | 24.5L |
�����ϱ����ݣ��ó��Ľ����ǣ�����д�����㣩
������ͬ���¶�ѹǿ�£�
��
��
��
��4������۵ĽǶȽ�����������仯���ɵ�ԭ��
��ÿ��1�֣���8�֣�ʵ������Ҫ����0.1 mol��L��1CuSO4��Һ480 mL��
�����в������������ʵ������֣���ʹ��������������
��1��ѡ����������ɱ�ʵ��������������У�������ƽ(��ȷ��0.1 g)��ҩ�ס��ձ�����������______��________�Լ�����������Ƭ��ֽ��
��2�����㣬Ӧѡ��������ȷ________
| A����ҪCuSO4 ����8g | B����ҪCuSO4��5H2O����12.0 g |
| C����ҪCuSO4��5H2O����12.5 g | D����ҪCuSO4����7.7 g |
��4���ܽ⡢��ȴ���ò�ʵ������Ҫʹ�ò�������Ŀ����_________________ ��
��5��ת�ơ�ϴ�ӡ���ת��ʱӦʹ��________��������Ҫϴ���ձ�2��3����Ϊ��______��
��6�����ݣ�ҡ�ȡ�
��7������õ���Һ����һ��ʱ�����ָ�����Լ�ƿ�����ñ�ǩ��ע�����Ƶ�ʱ �䡢��Һ���Ƽ�Ũ�ȡ�
��8�������ƹ����У�ijѧ���۲춨��ʱҺ�������ͼ��ʾ��������Һ��Ũ�Ȼ�________(�ƫ�ߡ�����ƫ�͡�����Ӱ�족)��

�������ʵķ��뷽������ȷ����
| A���ù��˵ķ�����ȥʳ������ɳ |
| B��������ķ�������ˮ���� |
| C���þƾ���ȡ��ˮ�еĵⵥ�� |
| D���ü��ȵķ�����ȥ̼�����е�̼������ |