��Ŀ����
 �����£�ȡһ������PbI2������ɱ�����Һ��Tʱ�̸ı�ijһ���������ӵ�Ũ�ȱ仯��ͼ��ʾ����ע����һ��ƽ��ʱc��I-��=2��10-3mol/L��c��Pb2+��=2��10-3mol/L�������й�˵����ȷ���ǣ�������
�����£�ȡһ������PbI2������ɱ�����Һ��Tʱ�̸ı�ijһ���������ӵ�Ũ�ȱ仯��ͼ��ʾ����ע����һ��ƽ��ʱc��I-��=2��10-3mol/L��c��Pb2+��=2��10-3mol/L�������й�˵����ȷ���ǣ�������| A�������£�Ksp=2��10-6 |
| B���¶Ȳ��䣬��PbI2������Һ�м�����������ǦŨ��Һ��PbI2���ܽ�Ȳ��䣬c��Pb2+������ |
| C��Tʱ�̸ı�������������¶ȣ�PbI2�ģ�Ksp ���� |
| D��������Ksp[PbS]=8��10-28����PbI2������Һ�м���Na2S��Һ��PbI2��s��+S2-��aq��?PbS��s��+2I-��aq����Ӧ�Ļ�ѧƽ�ⳣ��Ϊ5��1018 |
���㣺���ܵ���ʵ��ܽ�ƽ�⼰����ת���ı���
ר�⣺
������A����Ksp��PbI2��=[Pb2+]?[I-]2���������
B����PbI2������Һ�м�����������ǦŨ��Һ��c��Pb2+������PbI2���ܽ�ȼ�С��
C����ͼ������Tʱ�̸ı�����������º����������Ũ�ȣ�
D����Ksp[PbS]=8��10-28������c��Pb2+����c��S2-�����پ�Ksp��PbI2�����c��Pb2+������c��I-�����ɵø÷�Ӧ��ƽ�ⳣ��K=
��
B����PbI2������Һ�м�����������ǦŨ��Һ��c��Pb2+������PbI2���ܽ�ȼ�С��
C����ͼ������Tʱ�̸ı�����������º����������Ũ�ȣ�
D����Ksp[PbS]=8��10-28������c��Pb2+����c��S2-�����پ�Ksp��PbI2�����c��Pb2+������c��I-�����ɵø÷�Ӧ��ƽ�ⳣ��K=
| [I-]2 |
| [S2-] |
���
�⣺A����һ��ƽ��ʱc��I-��=2��10-3mol/L��c��Pb2+��=2��10-3mol/L��Ksp��PbI2��=[Pb2+]?[I-]2=2��10-3����2��10-3��2=8��10-9����A����
B����PbI2������Һ�м�����������ǦŨ��Һ��c��Pb2+������PbI2���ܽ�ȼ�С����B����
C����ͼ������Tʱ�̣�������Ũ��Ѹ���������С��Ǧ����������˵�������������º����������Ũ�ȣ�PbI2��Ksp ����C��ȷ��
D��Ksp[PbS]=8��10-28����c��Pb2+��=c��S2-��=
��10-14��Ksp��PbI2��=[Pb2+]?[I-]2=4��10-9������[I-]2=
��105���÷�Ӧ��ƽ�ⳣ��K=
=
=1��1019����D����
��ѡC��
B����PbI2������Һ�м�����������ǦŨ��Һ��c��Pb2+������PbI2���ܽ�ȼ�С����B����
C����ͼ������Tʱ�̣�������Ũ��Ѹ���������С��Ǧ����������˵�������������º����������Ũ�ȣ�PbI2��Ksp ����C��ȷ��
D��Ksp[PbS]=8��10-28����c��Pb2+��=c��S2-��=
| 8 |
| 8 |
| [I-]2 |
| [S2-] |
| ||
|
��ѡC��
���������⿼���˾��ܶȻ��ļ��㣬ע��̶ȵ�������������Ӹ����ȶ��ܶȻ���Ӱ�죬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ��Уͨ��֤��Ч��ҵϵ�д�
��Уͨ��֤��Ч��ҵϵ�д�
�����Ŀ
��ѧ��Դ�����Ҳ��������������й������еĻ�ѧ������ȷ���ǣ�������
| A��������ʹ�õĺ��������й¶�����ӿ����е�PM2.5 |
| B��Ũ�������ǿ��ʴ�ԣ�����Ũ�����ʴʯӢ������Ʒ |
| C��ʩ��ʱ��ũ�ҷʲ�ľ�ң���Ч�ɷ�K2CO3�������뵪��NH4Cl���ʹ�� |
| D�������ͻ���̿������ΪƯ������ͬʱʹ�ã�Ư��Ч�������Լ�ǿ |
115��Cʱ��������̬�������������ϣ�1L���������9L������ϣ����ȼ�պ�ָ���ԭ״̬�����������������10L�����и������������ϴ��������ǣ�������
| A��CH4��C2H4 |
| B��CH4��C3H4 |
| C��C2H4��C3H4 |
| D��C2H2��C3H6 |
���з�Ӧ�����ӷ���ʽ����ȷ���ǣ�������
| A��п������ͭ��Һ��Ӧ��Zn+Cu2+=Zn2++Cu |
| B������������Һ�����ᷴӦ��OH-+H+=H2O |
| C������ϡ���ᷴӦ��2Fe+6H+=2Fe3++3H2�� |
| D���Ȼ�����Һ��ϡ���ᷴӦ��Ba2++SO42-=BaSO4�� |
���õĵ����һ��Һ�����ʽ���������缫����������-��Na/S8��������෴�������ɹ������ʽ�����Һ��缫��������ͼ����һ������-��-�����������������ɵ����Ĺܣ������ҵ������ƣ��۵�98�棩�����ҵ��������۵�119�棩������������Na+ͨ���������й�˵������ȷ���ǣ�������
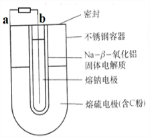

| A��aΪ��ص����� |
| B�����ʱb�ӵ�Դ������ |
| C��������ӦΪ��nS8+16ne-=8Sn2- |
| D��Na+���Դ����������ʺ���Ӧ�Ӷ����ݵ��� |
 Fe2+��Fe3+��O22-��CN-��F-���л����ӵ��γɵĻ�������й㷺��Ӧ�ã�
Fe2+��Fe3+��O22-��CN-��F-���л����ӵ��γɵĻ�������й㷺��Ӧ�ã�

 �ĺ�����·�������Լ���ѡ��������ͼ��ʾ��д����Ӧ����P��Ҫ��Ӧ������
�ĺ�����·�������Լ���ѡ��������ͼ��ʾ��д����Ӧ����P��Ҫ��Ӧ������