��Ŀ����
ͨ�����·�Ӧ���ɻ�ȡH2�������й�˵����ȷ����
��̫������ֽ�ˮ���⣺2H2O(l) 2H2(g)+O2(g)��H1=571.6kJ/mol
2H2(g)+O2(g)��H1=571.6kJ/mol
�ڽ�̿��ˮ��Ӧ���⣺C(s)+H2O(g) CO(g)+H2(g)��H2=131.3kJ/mol
CO(g)+H2(g)��H2=131.3kJ/mol
�ۼ�����ˮ��Ӧ���⣺CH4(g)+H2O(g) CO(g)+3H2(g)��H3=206.1kJ/mol
CO(g)+3H2(g)��H3=206.1kJ/mol
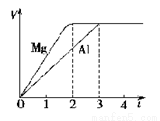
A����Ӧ���е���ת��Ϊ��ѧ��
B����Ӧ��Ϊ���ȷ�Ӧ
C����Ӧ��ʹ�ô�������H3��С
D����ӦCH4(g) C(s)+2H2(g)�Ħ�H3=74.8kJ/mol
C(s)+2H2(g)�Ħ�H3=74.8kJ/mol
��ϰ��ϵ�д�
�����Ŀ
1�����ڽ�����ʴ�������У���ȷ���ǣ�������
| A�� | ��������ʴ�ı����ǣ�M+nH2O=M��OH��n+n/2H2�� | |
| B�� | ������һ������·����ĵ绯ѧ��ʴ��Ҫ��������ʴ | |
| C�� | ���������������Ʋ������ʴʱ�������ǶƲ㱻���� | |
| D�� | �����£����ڿ����еĽ�����Ҫ������ѧ��ʴ |
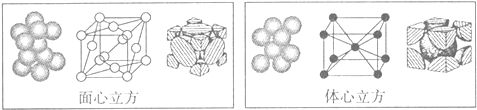
2���������ƣ�NaClO2������Ҫ�ĺ�������������ҵ����Ҫ�����շ��͵�ⷨ������ȡ�������������������ɵ�ClO2���岻�ȶ���Ũ�ȸ�ʱ�ױ�ը������������������ʾ������˵���в���ȷ���ǣ�������

| A�� | ������ֻҪ�������ṩ������������ | |
| B�� | ��ⷨ��ClO2�����������ĵ缫��ӦʽΪ��ClO2+Na++e-=NaClO2 | |
| C�� | ���շ��м���H2O2��Ҫ������ԭ�� | |
| D�� | �ӻ����ĽǶȿ��ǣ���ⷨ�����շ������� |


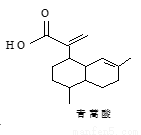
 ��Ϊͬ���칹��
��Ϊͬ���칹��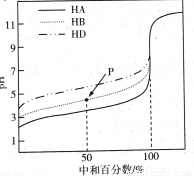
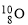
 B.Mg2+�Ľṹʾ��ͼ��
B.Mg2+�Ľṹʾ��ͼ��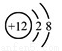
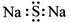 D.��������Ľṹ��ʽ��C2H4O2
D.��������Ľṹ��ʽ��C2H4O2