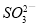��Ŀ����
��14�֣�ij��ѧ��ȤС��ͬѧչ����Ư���������ƣ�NaClO2�����о���
ʵ�����ȡNaClO2����
��֪��NaClO2������Һ���¶ȵ���38��ʱ�����ľ�����NaClO2?3H2O������38��ʱ�����������NaClO2������60��ʱNaClO2�ֽ��NaClO3��NaCl��Ba(ClO)2������ˮ��
������ͼ��ʾװ�ý���ʵ�顣
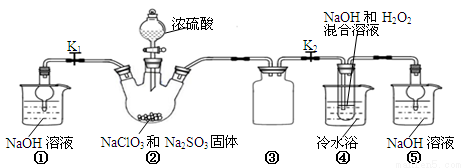
��1��װ�âٵ������� װ�â۵������� ��
��2��װ�â��в���ClO2�Ļ�ѧ����ʽΪ ��
��3����װ�âܷ�Ӧ�����Һ��þ���NaClO2�IJ�������Ϊ��
�ټ�ѹ��55�������ᾧ���ڳ��ȹ��ˣ��� ���ܵ���60�����õ���Ʒ��
��4�����ʵ���������NaClO2�����Ƿ�������Na2SO4�������������ǣ�ȡ����������������ˮ�� ��
ʵ��ⶨij����������Ʒ�Ĵ��ȡ�
�������ʵ�鷽����������ʵ�飺
��ȷ��ȡ��������������Ʒm g���ձ��У�������������ˮ�����ĵ⻯�ؾ��壬�ٵ���������ϡ���ᣬ��ַ�Ӧ����֪��ClO2��+ 4I��+4H+ =2H2O+2I2+Cl�����������û��Һ���100mL������Һ��
����ȡ25.00mL������Һ����ƿ�У���c mol?L-1 Na2S2O3��Һ�ζ������ζ��յ㡣�ظ�2�Σ�������ı���Һ�������ƽ��ֵΪV mL����֪��I2 +2S2O32��=2I��+S4O62������
��5���ζ���ʹ�õ�ָʾ���� ���ﵽ�ζ��յ�ʱ������Ϊ ��
��6�� ��Ʒ��NaClO2����������Ϊ ���ú�m��c��V�Ĵ���ʽ��ʾ��ʽ����NaClO2 90.5����
��1�����ն����ClO2���壬��ֹ��Ⱦ����(1��)
��ֹ����(������ȫƿ��������ȷ˵��) (1��)
��2��2NaClO3+Na2SO3+H2SO4(Ũ)==2ClO2��+2Na2SO4+H2O(2��)
��3����38�桫60�����ˮϴ��(2�֣���д��ϴ�ӡ���1��)
��4���μӼ���BaCl2��Һ�����а�ɫ�������֣�����Na2SO4�����ް�ɫ�������֣���Na2SO4�� (2�֣���һ�����1��)
��5��������Һ��1�֣�����Һ����ɫ��Ϊ��ɫ�Ұ���Ӳ���ɫ (2�֣�������ӡ���д��1)
��6�� 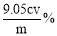 ��
�� 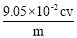 (3��)
(3��)
��������
�����������1��װ������δ��Ӧ��ClO2��װ�âٿ�������δ��Ӧ��ClO2����ֹ�ݳ���Ⱦ������װ�â������巴Ӧ��װ����ѹǿ���ͣ�װ�â۷�ֹ������
��2��װ�â��в���ClO2�ķ�Ӧ����������������Һ��������������Ϊ�����ƣ���������ԭΪ�������ȣ���Ӧ�Ļ�ѧ����ʽΪ��2NaClO3+Na2SO3+H2SO4=2ClO2��+2Na2SO4+H2O��
��3������Һ����ȡ���壬һ����������ᾧ�����ˡ�ϴ�ӡ�����ķ�����Ϊ��ֹ��������NaClO2?3H2O��Ӧ���ȹ��ˣ�����Ŀ��Ϣ��֪��Ӧ�����¶�38�桫60�����ϴ�ӣ�����60����
��4������SO42?��Ba2+�ķ�Ӧ��������NaClO2�����Ƿ�������Na2SO4�������������ǣ�ȡ����������������ˮ���μӼ���BaCl2��Һ�����а�ɫ�������֣�����Na2SO4�����ް�ɫ�������֣���Na2SO4��
��5���������۱���ɫ������ָʾ��Ϊ��������Һ����Ӧ����ʱ���ⷴӦ��ȫ���μ����һ��Na2S2O3��Һʱ��Һ����ɫ��Ϊ��ɫ�Ұ�����ڲ���ɫ��˵������ζ��յ㡣
��6������Ʒ��NaClO2����������Ϊx����NaClO2��2I2��4S2O32-
90.5g 4mol
mxg c mol?L-1��V��10-3L��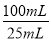
����90.5g��mxg=4mol��c mol?L-1��V��10-3L��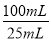 �����x=
�����x=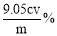 ��
�� 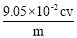 ��
��
���㣺���⿼�����ʵ��Ʊ���ʵ�鷽������������ۡ�������ԭ��Ӧ�ζ���

 ��ɢ˼ά�¿���ϵ�д�
��ɢ˼ά�¿���ϵ�д���֪:������Ũ��Ϊ0.1 mol��L-1��������Һ��pH���:
���� | NaF | NaClO | Na2CO3 |
pH | 7.5 | 9.7 | 11.6 |
�����й�˵����ȷ���ǣ� ����
A������ͬ�¶��£�ͬŨ�ȵ���������Һ�ĵ�������˳��H2CO3��HClO��HF
B������CO2 ͨ��0.1 mol��L-1Na2CO3��Һ������Һ����,����Һ��
2 c (CO32��)+ c (HCO3��)= 0.1 mol��L-1
C�������ϱ�, ˮ�ⷽ��ʽClO��+ H2O HClO + OH����ƽ�ⳣ��K��10��7.6
HClO + OH����ƽ�ⳣ��K��10��7.6
D��������NaClO ��Һ��ͨHF������ǡ����ȫ��Ӧʱ��
c(Na+)��c(F��)��c(H+)��c(HClO)��c(OH��)