��Ŀ����
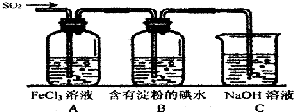 ij��ȤС��̽��SO2��������ʣ�����ʹ�õ�ҩƷ��װ����ͼ��ʾ��
ij��ȤС��̽��SO2��������ʣ�����ʹ�õ�ҩƷ��װ����ͼ��ʾ����1��SO2������Fe3+��Ӧ�Ļ�ԭ������
��2������ʵ�鷽����������ʵ������ȡ����SO2����
A��Na2SO3��Һ��HNO3
B��Na2SO3Ũ��Һ��Ũ����
C���������ڴ�����ȼ��
��3��װ��C��������
��4����Ҫ��A��������Һ��ȡ���壬������е�ʵ��������裺��������ȴ�ᾧ��
A�������� B��ʯ���� C��©�� D��������
��5��������װ����ͨ�������SO2��Ϊ����֤A��SO2��Fe3+������������ԭ��Ӧ������ȡA�е���Һ���ֳ����ݣ������������ʵ�飺
�����٣�����һ����Һ�м�������KMnO4��Һ����ɫ��ȥ
�����ڣ����ڶ�����Һ�м���KSCN��Һ����죬�ټ������Ƶ���ˮ����Һ��죬
�����ۣ�����������Һ�м����������ữ��BaCl2��Һ��������ɫ����
������������������
��6���ܱ���I-�Ļ�ԭ������SO2��������
���㣺����ʵ�鷽�������,��������Ļ�ѧ����
ר�⣺ʵ����
��������1������װ��A��Ӧ�����ӷ���ʽSO2+2Fe3++2H2O�T2Fe2++SO42-+4H+���н�𣻸��ݻ��ϼ۱仯�жϷ�Ӧ��SO2��Fe3+�����ʵ���֮�ȣ�
��2��ʵ������ȡ����Ҫ���Dz������㡢���ơ����ܺ����ʣ�
��3������������д̼�����ζ����Ⱦ������������������Һ���ն�������ֹ������Ⱦ��
��4����A��������Һ��ȡ����Ϊ�̷�����Һ�еõ��̷���ʵ�����������Ũ������ȴ�ᾧ������ϴ�ӣ����ﲽ��õ��������õ�������ʯ�������������������õ�©�����ձ������������ݴ˽��н��
��5�������������������ط���������ԭ��Ӧʹ���������Һ��ɫ��Fe2+Ҳʹ���������Һ��ɫ��
��6������������ʹ���е�ĵ�����Һ��ɫ��˵��������������ԭ��Ӧ�����ݻ�ԭ���Ļ�ԭ�Դ��ڻ�ԭ����Ļ�ԭ�Է�����
��2��ʵ������ȡ����Ҫ���Dz������㡢���ơ����ܺ����ʣ�
��3������������д̼�����ζ����Ⱦ������������������Һ���ն�������ֹ������Ⱦ��
��4����A��������Һ��ȡ����Ϊ�̷�����Һ�еõ��̷���ʵ�����������Ũ������ȴ�ᾧ������ϴ�ӣ����ﲽ��õ��������õ�������ʯ�������������������õ�©�����ձ������������ݴ˽��н��
��5�������������������ط���������ԭ��Ӧʹ���������Һ��ɫ��Fe2+Ҳʹ���������Һ��ɫ��
��6������������ʹ���е�ĵ�����Һ��ɫ��˵��������������ԭ��Ӧ�����ݻ�ԭ���Ļ�ԭ�Դ��ڻ�ԭ����Ļ�ԭ�Է�����
���
�⣺��1�����������������ӷ�Ӧ�����ӷ���ʽΪ��2Fe3++SO2+2H2O=2Fe2++SO42-+4H+����Ӧ�������ӱ��������������ӣ����Ի�ԭ����Ϊ�������ӣ�
�μӷ�Ӧ��SO2��Fe3+�����ʵ���֮��=�������֮��=1��2��
�ʴ�Ϊ��Fe2+��1��2��
��2��A���������ǿ�����ԣ��ܹ����������������������ƣ����õ������������壬��A����
B��Ũ���������ǿ���ԣ���Ũ����ӷ�������������Һ��Ũ�����ܹ���Ӧ���ɶ����������壬��B��ȷ��
C���������ڴ�����ȼ�գ������������ƣ�������ô����Ķ�������C����
�ʴ�Ϊ��B��
��3����������������������������д̼�����ζ��ֱ���ŷŻ���Ⱦ���������ڶ��������ܺͼӦ�����κ�ˮ�����ü�Һ����������������װ��C������Ϊ������SO2β������ֹ��Ⱦ������
�ʴ�Ϊ������SO2β������ֹ��Ⱦ������
��4����Һ�еõ��̷���ʵ�����Ϊ������Ũ������ȴ�ᾧ������ϴ�ӡ����
��������ʹ�������У�A��������D����������������ʹ������Ϊ��C��©����D��������������û��ʹ�õ����ǣ�B��ʯ������
�ʴ�Ϊ�����ˣ�B��
��5�����������л�ԭ�ԣ����������ǿ�����ԣ������������������ط���������ԭ��Ӧʹ���������Һ��ɫ��Fe2+Ҳʹ���������Һ��ɫ�����Է����ٲ�������
�ʴ�Ϊ�������٣���ΪA����Һ�к���SO2��SO2Ҳ��ʹKMnO4��Һ��ɫ��
��6��I2+SO2+2H2O=2HI+H2SO4����������ʹ���е�ĵ�����Һ��ɫ��˵��������������ԭ��Ӧ�����������������������ǻ�ԭ������ԭ���Ļ�ԭ�Դ��ڻ�ԭ����Ļ�ԭ�ԣ�
�ʴ�Ϊ��B����ɫ��Һ��ɫ��
�μӷ�Ӧ��SO2��Fe3+�����ʵ���֮��=�������֮��=1��2��
�ʴ�Ϊ��Fe2+��1��2��
��2��A���������ǿ�����ԣ��ܹ����������������������ƣ����õ������������壬��A����
B��Ũ���������ǿ���ԣ���Ũ����ӷ�������������Һ��Ũ�����ܹ���Ӧ���ɶ����������壬��B��ȷ��
C���������ڴ�����ȼ�գ������������ƣ�������ô����Ķ�������C����
�ʴ�Ϊ��B��
��3����������������������������д̼�����ζ��ֱ���ŷŻ���Ⱦ���������ڶ��������ܺͼӦ�����κ�ˮ�����ü�Һ����������������װ��C������Ϊ������SO2β������ֹ��Ⱦ������
�ʴ�Ϊ������SO2β������ֹ��Ⱦ������
��4����Һ�еõ��̷���ʵ�����Ϊ������Ũ������ȴ�ᾧ������ϴ�ӡ����
��������ʹ�������У�A��������D����������������ʹ������Ϊ��C��©����D��������������û��ʹ�õ����ǣ�B��ʯ������
�ʴ�Ϊ�����ˣ�B��
��5�����������л�ԭ�ԣ����������ǿ�����ԣ������������������ط���������ԭ��Ӧʹ���������Һ��ɫ��Fe2+Ҳʹ���������Һ��ɫ�����Է����ٲ�������
�ʴ�Ϊ�������٣���ΪA����Һ�к���SO2��SO2Ҳ��ʹKMnO4��Һ��ɫ��
��6��I2+SO2+2H2O=2HI+H2SO4����������ʹ���е�ĵ�����Һ��ɫ��˵��������������ԭ��Ӧ�����������������������ǻ�ԭ������ԭ���Ļ�ԭ�Դ��ڻ�ԭ����Ļ�ԭ�ԣ�
�ʴ�Ϊ��B����ɫ��Һ��ɫ��
���������⿼������������ʵ�鷽������ƣ���Ŀ�Ѷ��еȣ������漰������ԭ��Ӧ�ļ��㡢�����Ի�ԭ��ǿ���Ƚϡ�����ʵ�鷽������������۵�֪ʶ������ѧ�����������ͼ��������Ŀ��飬��ȷ����Ũ��������ʡ���������ļ��鷽����֪ʶΪ�����ؼ���

��ϰ��ϵ�д�
�����Ŀ
����þ���ں�ϡ��������Һ����������þ������狀�ˮ����Ӧ��þ��HNO3���ʵ���֮���ǣ�������
| A��3��8 | B��1��4 |
| C��2��5 | D��4��1 |
ijδ֪��Һ�м���AgNO3��Һ�а�ɫ�������ɣ�����ϡ������������ܽ⣬����ɫ��ζ���������ɣ�������ͨ�����ʯ��ˮ�У�ʯ��ˮ����ǣ���֪����Һ�к��������������������е�һ�飮�����ǣ�������
| A��Cl- SO42- |
| B��Cl- NO3- |
| C��Cl- CO32- |
| D��Cl- OH- |
ʵ������һƿ�����Ȼ��Ƶ��������ƹ����Լ������ⶨNaOH����������Ϊ82.0%��Ϊ����֤�䴿�ȣ���Ũ��Ϊ0.2mol?L-1��������еζ������ƫ�ߣ��Է���������������Щʵ���������ģ�������
| A��ת�ƴ���Һ������ƿʱ��δϴ���ձ� |
| B���ζ����յ�ʱ���ζ��ܼ�������Һ�� |
| C���ζ�ʱ����Ӧ����ҡ��̫���ң�������Һ�彦�� |
| D������ʽ�ζ��ܿ�ʼʱ���ӣ����յ�ʱ���� |
���а�װ��ʶ�����˵��ǣ�������
| ѡ�� | A | B | C | D |
| ���� | Ũ���� | �ƾ� | С�մ� | ����� |
| ��ʶ |  |  |  |  |
| A��A | B��B | C��C | D��D |
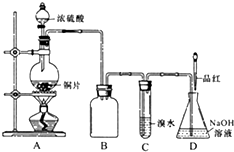 ij��ѧ��ȤС�����ʵ�飬��Ũ������ͭ��Ӧ�Ƶ�SO2���������ʵ��̽����
ij��ѧ��ȤС�����ʵ�飬��Ũ������ͭ��Ӧ�Ƶ�SO2���������ʵ��̽����