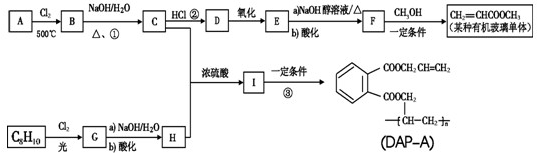
��Ŀ����
�٣�Pd����ż����Ӧ�ǽ����л��ϳɵ��о��ȵ�֮һ�����磺

������I�������ºϳ�·��ã�
CH2=CH-CH2CI
��
��������ʽC3H4O��
��
��1��������I��Br2�ӳɵIJ���Ľṹ��ʽΪ ��
��2���������ķ���ʽΪ ��
��3������������������·���������Ӧ�Ļ�ѧ����ʽΪ ��ע����������
��4����������Ľṹ��ʽΪ ��
��5�����ǻ�����IIһ��ͬ���칹�壬���ķ����б�����������ȡ�������ܷ���������Ӧ���ұ����ϵ�һ�ȴ�����2�֣����Ľṹ��ʽ������ �� ��
��6��1���� ��1��
��1�� ��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ�������л�������������Ľṹ��ʽΪ ��
��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ�������л�������������Ľṹ��ʽΪ ��

������I�������ºϳ�·��ã�
CH2=CH-CH2CI
| NaOH��H2O |
| �� |
| ���� |
| ���� |
��1��������I��Br2�ӳɵIJ���Ľṹ��ʽΪ
��2���������ķ���ʽΪ
��3������������������·���������Ӧ�Ļ�ѧ����ʽΪ
��4����������Ľṹ��ʽΪ
��5�����ǻ�����IIһ��ͬ���칹�壬���ķ����б�����������ȡ�������ܷ���������Ӧ���ұ����ϵ�һ�ȴ�����2�֣����Ľṹ��ʽ������
��6��1����
 ��1��
��1�� ��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ�������л�������������Ľṹ��ʽΪ
��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ�������л�������������Ľṹ��ʽΪ���㣺�л���ĺϳ�,ͬ���칹�����ͬ���칹��
ר�⣺�л�������Ļ����Ӧ��
��������CH2=CH-CH2CI
���֪����ΪCH2=CH-CH2OH����
���Ң��ķ���ʽC3H4O�����Ԣ�ΪCH2=CH-CHO�������Ƶâ�ΪCH2=CH-COOH��
��1�����ݢ�Ľṹ��֪��������I��Br2�ӳɵIJ���Ľṹ��ʽ��
��2����������ķ�����֪�������ķ���ʽ��
��3������������Ӧ��ԭ����д�ʻ���������������·���������Ӧ�Ļ�ѧ����ʽ��
��4����������ķ�����֪����������Ľṹ��ʽ��
��5�����ǻ�����IIһ��ͬ���칹�壬���ķ����б�����������ȡ�����ұ����ϵ�һ�ȴ�����2�֣��ܷ���������Ӧ��˵����ȩ�����ݴ�д�ṹ��ʽ��
��6��1���� ��1��
��1�� ��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ�������л������������
��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ�������л������������ �е���ԭ�ӱ�ȡ�����ݴ�д�����Ľṹ��ʽ��
�е���ԭ�ӱ�ȡ�����ݴ�д�����Ľṹ��ʽ��
| NaOH��H2O |
| �� |
| ���� |
��1�����ݢ�Ľṹ��֪��������I��Br2�ӳɵIJ���Ľṹ��ʽ��
��2����������ķ�����֪�������ķ���ʽ��
��3������������Ӧ��ԭ����д�ʻ���������������·���������Ӧ�Ļ�ѧ����ʽ��
��4����������ķ�����֪����������Ľṹ��ʽ��
��5�����ǻ�����IIһ��ͬ���칹�壬���ķ����б�����������ȡ�����ұ����ϵ�һ�ȴ�����2�֣��ܷ���������Ӧ��˵����ȩ�����ݴ�д�ṹ��ʽ��
��6��1����
 ��1��
��1�� ��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ�������л������������
��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ�������л������������ �е���ԭ�ӱ�ȡ�����ݴ�д�����Ľṹ��ʽ��
�е���ԭ�ӱ�ȡ�����ݴ�д�����Ľṹ��ʽ�����
�⣺��CH2=CH-CH2CI
���֪����ΪCH2=CH-CH2OH����
���Ң��ķ���ʽC3H4O�����Ԣ�ΪCH2=CH-CHO�������Ƶâ�ΪCH2=CH-COOH��
��1�����ݢ�Ľṹ��֪��������I��Br2�ӳɵIJ���Ľṹ��ʽΪCH2Br-CHBr-COOH���ʴ�Ϊ��CH2Br-CHBr-COOH��
��2����������ķ�����֪�������ķ���ʽΪC9H8O2���ʴ�Ϊ��C9H8O2��
��3������������Ӧ��ԭ�������ǻ������⣬��д������������������·���������Ӧ�Ļ�ѧ����ʽΪ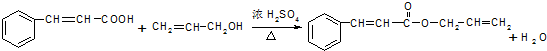 ��
��
�ʴ�Ϊ��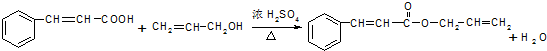 ��
��
��4����������ķ�����֪����������Ľṹ��ʽΪCH2=CH-CHO���ʴ�Ϊ��CH2=CH-CHO��
��5�����ǻ�����IIһ��ͬ���칹�壬���ķ����б�����������ȡ�����ұ����ϵ�һ�ȴ�����2�֣��ܷ���������Ӧ��˵����ȩ�����ݴ�д�ṹ��ʽΪ ��
��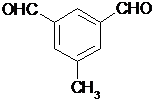 ���ʴ�Ϊ��
���ʴ�Ϊ��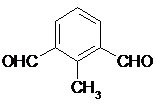 ��
��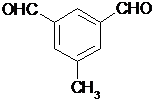 ��
��
��6��1���� ��1��
��1�� ��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ�������л������������
��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ�������л������������ �е���ԭ�ӱ�ȡ�����ݴ�д�����Ľṹ��ʽΪ
�е���ԭ�ӱ�ȡ�����ݴ�д�����Ľṹ��ʽΪ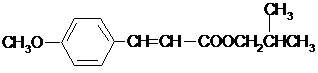 ���ʴ�Ϊ��
���ʴ�Ϊ��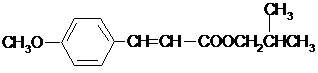 ��
��
| NaOH��H2O |
| �� |
| ���� |
��1�����ݢ�Ľṹ��֪��������I��Br2�ӳɵIJ���Ľṹ��ʽΪCH2Br-CHBr-COOH���ʴ�Ϊ��CH2Br-CHBr-COOH��
��2����������ķ�����֪�������ķ���ʽΪC9H8O2���ʴ�Ϊ��C9H8O2��
��3������������Ӧ��ԭ�������ǻ������⣬��д������������������·���������Ӧ�Ļ�ѧ����ʽΪ
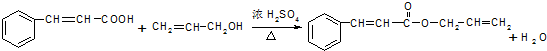 ��
���ʴ�Ϊ��
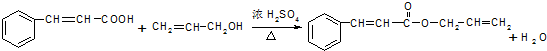 ��
����4����������ķ�����֪����������Ľṹ��ʽΪCH2=CH-CHO���ʴ�Ϊ��CH2=CH-CHO��
��5�����ǻ�����IIһ��ͬ���칹�壬���ķ����б�����������ȡ�����ұ����ϵ�һ�ȴ�����2�֣��ܷ���������Ӧ��˵����ȩ�����ݴ�д�ṹ��ʽΪ
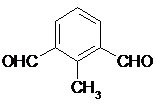 ��
��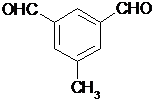 ���ʴ�Ϊ��
���ʴ�Ϊ��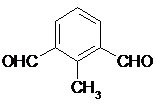 ��
��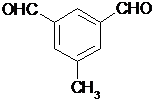 ��
����6��1����
 ��1��
��1�� ��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ�������л������������
��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ�������л������������ �е���ԭ�ӱ�ȡ�����ݴ�д�����Ľṹ��ʽΪ
�е���ԭ�ӱ�ȡ�����ݴ�д�����Ľṹ��ʽΪ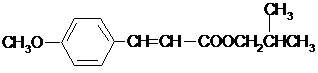 ���ʴ�Ϊ��
���ʴ�Ϊ��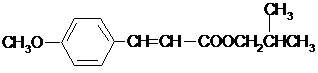 ��
��
���������⿼���л���ϳɣ�Ϊ��Ƶ���㣬���պϳ������еĽṹ�ı仯�������ŵı仯�������������ʵĹ�ϵΪ���Ĺؼ�������±�����������ʵĿ��飬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
�����й�������ԭ��Ӧ��˵����ȷ���ǣ�������
| A��������ԭ��Ӧ��ʵ����Ԫ�صĻ��ϼ۷��������� |
| B���û���Ӧһ����������ԭ��Ӧ |
| C���õ�������Խ������ʣ���������Խǿ |
| D��Ԫ�صĻ���̬������̬����Ԫ��һ�������� |
������ڵ���ʵ���������ȷ���ǣ�������
| A��Һ̬������磬�����Ƿǵ���� |
| B��NaHSO4������״̬�µ������Na+��HSO4-�������� |
| C������ˮ���ܵ���Ļ�����һ���ǵ���� |
| D��������ڵ����������µ���������� |
 ��������ƣ�Na2S2O3���dz��õĻ�ԭ������ά����C����ѧʽC6H8O6����ˮ��Һ�м������I2��Һ��ʹά����C����ȫ������ʣ���I2��Na2S2O3����Һ�ζ����ɲⶨ��Һ��ά����C�ĺ����������ķ�ӦΪ��C6H8O6+I2�TC6H6O6+2H++2I-2S2O32-+I2�TS4O62-+2I-
��������ƣ�Na2S2O3���dz��õĻ�ԭ������ά����C����ѧʽC6H8O6����ˮ��Һ�м������I2��Һ��ʹά����C����ȫ������ʣ���I2��Na2S2O3����Һ�ζ����ɲⶨ��Һ��ά����C�ĺ����������ķ�ӦΪ��C6H8O6+I2�TC6H6O6+2H++2I-2S2O32-+I2�TS4O62-+2I-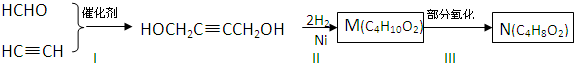
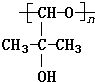
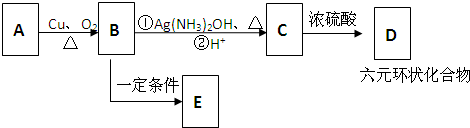
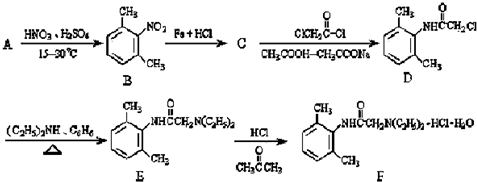
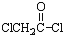 ��������NaOH��Һ���ȳ�ַ�Ӧ�Ļ�ѧ����ʽ��
��������NaOH��Һ���ȳ�ַ�Ӧ�Ļ�ѧ����ʽ��