��Ŀ����
2��H��C��N��O��Na��S��Cl�dz��������ֶ�����Ԫ�أ���1��Nλ��Ԫ�����ڱ��ڶ� ���ڵڢ�A �壬���̬ԭ�Ӻ�����3��δ�ɶԵ��ӣ�S�Ļ�̬ԭ�Ӻ�������Ų�ʽΪ1s22s22p63s23p4��
��2���á�������������գ�
| ��һ������ | �縺�� | �ȶ��� | ���� |
| N��O | C��N | H2S��H2O | H2SO4��HClO4 |
��4��NH3���������ܼ�������Һ����ԭ����NH3���Ӽ����γɷ��Ӽ����������Ĵ���ʹ�ð������Ӽ�������������ǿ���е��������ߣ���Һ������֪25�桢101KPa��NH3���ڴ�����ȼ�գ�ÿת��1mol e-���ų�akJ������д����������NH3ȼ�յ��Ȼ�ѧ����ʽ4NH3��g��+5O2��g��=4NO��g��+6H2O��g����H=-4a kJ/mol��
��5��ͨ������£�N2O5Ϊ��ɫ���壬����Na2O2�ܷ�����Ӧ��������CO2��Na2O2�ܷ�Ӧ����д����Ӧ�Ļ�ѧ����ʽ�����õ����ű������ת�Ƶķ������Ŀ��
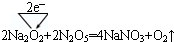 ��
��
���� ��1�����ݵ��Ӳ���=������������������=������������N�Ļ�̬ԭ�Ӻ���δ�ɶԵ��Ӵ���2p�����Sԭ�Ӻ�����ӻ�̬�Ų�ʽΪ1s22s22p63s22p4��
��2��ͬ���ڴ����ҵ�һ�����������������ڵ�IIA����A����������ԣ�
ͬ���ڴ����ҵ縺������Ԫ�صķǽ�����Խǿ����Ӧ���⻯����ȶ���Խǿ��
Ԫ�صķǽ�����Խǿ����Ӧ������������ˮ���������Խǿ��
��3���Ǽ��Թ��ۼ�Ϊͬ�ַǽ���Ԫ�ص�ԭ�Ӽ��γɹ��ۼ������Թ��ۼ����ɲ�ͬ�ַǽ���Ԫ�ص�ԭ�Ӽ����γɵĹ��ۼ���
��4����ԭ�ӷǽ�����ǿ��������֮�������γ������ʹ��е����߶�����Һ����1mol����NH3������O2��Ӧ����ˮ��������һ�����壬��Ӧ����һ��������ˮ�����ݵ���ת�Ƽ��㷴Ӧ�İ�������ϻ�ѧ����ʽ������ϵ���㷴Ӧ���ȣ����Ȼ�ѧ����ʽ��д����д����
��5�����ݶ�����̼��Na2O2�ķ�Ӧ��֪���÷�Ӧ������NaNO3��O2����������ԭ��Ӧ�У����ϼ�����Ԫ��ʧ���ӣ����ϼ۽���Ԫ�صõ����ӣ���ʧ��������ȼ�Ϊת�Ƶ�������
��� �⣺��1��NԪ��λ��Ԫ�����ڱ��ڶ����ڵڢ�A�壬N�Ļ�̬ԭ�Ӻ���δ�ɶԵ��Ӵ���2p�������3��δ�ɶԵ��ӣ�Sԭ�Ӻ��������Ϊ16��������ӻ�̬�Ų�ʽΪ1s22s22p63s23p4��
�ʴ�Ϊ��������A��3��1s22s22p63s23p4��
��2��ͬ���ڴ����ҵ�һ��������������A����������ԣ����Ե�һ������N��O��
ͬ���ڴ����ҵ縺���������Ե縺��C��N��
ͬ����Ԫ�ش��ϵ��·ǽ������������ǽ����ԣ�O��S��Ԫ�صķǽ�����Խǿ����Ӧ���⻯����ȶ���Խǿ����̬�⻯���ȶ��ԣ�H2S��H2O��
ͬ����Ԫ�ش�����Ԫ�طǽ���������ǿ���ǽ����ԣ�Cl��S��Ԫ�طǽ�����Խǿ����Ӧ������������ˮ���������Խǿ�����ԣ�H2SO4��HClO4��
�ʴ�Ϊ������������������
��3��H2O2��N2H4��C2H2��C2H4��C2H6��C6H6�мȺ����Լ��ֺ��Ǽ��Լ���
�ʴ�Ϊ��H2O2��N2H4��C2H2��C2H4��C2H6��C6H6����ѡ��һ��
��4����ԭ�ӷǽ�����ǿ��������֮�������γ����������Ĵ���ʹ�ð������Ӽ�������������ǿ���е��������ߣ���Һ������1mol����NH3������O2��Ӧ����ˮ��������һ�����壬4NH3��g��+5O2��g��=4NO��g��+6H2O��g������Ӧ������4mol������Ӧ����ת��20mol��ÿת��1mol e-���ų�akJ��������Ӧ4mol��������4a kJ��д���Ȼ�ѧ����ʽΪ��4NH3��g��+5O2��g��=4NO��g��+6H2O��g����H=-4a kJ/mol��
�ʴ�Ϊ��NH3���Ӽ����γɷ��Ӽ����������Ĵ���ʹ�ð������Ӽ�������������ǿ���е��������ߣ���Һ����4NH3��g��+5O2��g��=4NO��g��+6H2O��g����H=-4a kJ/mol��
��5����Na2O2��N2O5�ķ�Ӧ�У�����NaNO3��O2�����ϼ�������=���ϼ۽�����=ת�Ƶ�����=2������ת�����Ϊ��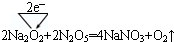 ���ʴ�Ϊ��
���ʴ�Ϊ��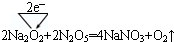 ��
��
���� ������Ҫ�����˺�������Ų���Ԫ�����ʵıȽϡ���ѧ���Լ�������ԭ��Ӧ���ѶȲ���ע���Ӧ֪ʶ�����ã�

| A�� | �ö����ЧӦ����NaCl��Һ��KCl��Һ | |
| B�� | ��ij��Һ�м���BaCl2��Һ���ְ�ɫ�����������Һ�п϶���SO42- | |
| C�� | ��KSCN��Һ����FeCl2��Һ��Fe2��SO4��3��Һ | |
| D�� | ����ɫ��Ӧ�ɼ����NaCl��KCl��Na2SO4�������� |
| ���ữѧʽ | HSCN | CH3COOH | HCN | H2CO3 |
| ����ƽ�ⳣ�� | 1.3��10 -1 | 1.7��10 -5 | 6.2��10 -10 | K1=4.3��10-7 K2=5.6��10-11 |
��4��д��̼��ĵ�һ������ƽ�ⳣ������ʽ��K1=$\frac{[HC{{O}_{3}}^{-}]•[{H}^{+}]}{[{H}_{2}C{O}_{3}]}$��
��2��25��ʱ�������ʵ���Ũ�ȵ� a��CH3COONa��b��NaCN��c��Na2CO3��d��NaHCO3��Һ�� pH �ɴ�С��˳��Ϊcbda ������ĸ����
��3�������£�0.1mol•L-1�� CH3COOH ��Һ��ˮϡ�����У����б���ʽ�����ݱ�����BD������ţ�
A��[H+]B.$\frac{{H}^{+}}{C{H}_{3}COOH}$ C��[H+]•[OH-]D��$\frac{O{H}^{-}}{{H}^{+}}$
��4��25��ʱ���� 20mL 0.1 mol•L-1 CH3COOH ��Һ�� 20mL 0.1 mol•L-1HSCN ��Һ�ֱ��� 20mL 0.1 mol•L-1NaHCO3��Һ��ϣ�ʵ���ò��������������V����ʱ�䣨t���ı仯��ͼ1��ʾ����Ӧ��ʼ��������Һ���� CO2��������ʴ������Բ����ԭ����HSCN�����Ա�CH3COOHǿ������Һ��c��H+���ϴ�����Һ��NaHCO3��Һ�ķ�Ӧ���ʿ죬��Ӧ��������������Һ�У�c��CH3COO-����c��SCN-�������������������=������

��5�������Ϊ 100mL pH=2 �� CH3COOH ��һԪ�� HX����ˮϡ������ pH ����Һ����Ĺ�ϵ��ͼ2��ʾ���� HX �ĵ���ƽ�ⳣ��С�ڣ�����ڡ�����С�ڡ����ڡ���CH3COOH �ĵ���ƽ�ⳣ����
��6��25��ʱ���� CH3COOH �� CH3COONa �Ļ����Һ�У������ pH=6��
����Һ�� c��CH3COO-��-c��Na+��=9.9��10-7 mol•L-1���ȷֵ����$\frac{c��C{H}_{3}CO{O}^{-}��}{c��C{H}_{3}COOH��}$=18��
��7��д������ CO2ͨ�����������Һ�е����ӷ���ʽ��CO2+H2O+ClO-=HCO3-+HClO��
| A�� | CCl4����CH4�Ƶã�����ȡ��ˮ�еĵ� | |
| B�� | �Ҵ������ụΪͬ���칹�� | |
| C�� | ���ȼ���ֻ��һ�ֽṹ | |
| D�� | �����ʵ������Ҵ�����ϩ��ȫȼ��ʱ����������������� |
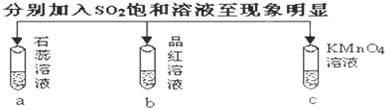
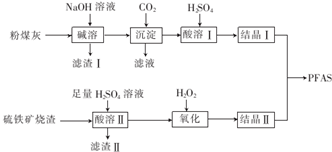
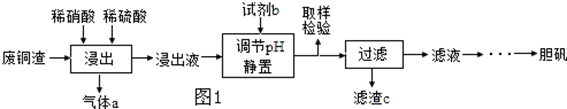
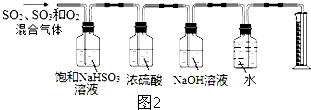
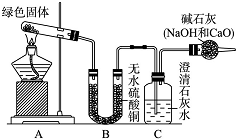 ͭ�����ã����������һ����ɫ���壬Ϊ�˽�ͭ�ڿ����еĸ�ʴ�����ij��ѧ��ȤС���ռ�����ͭ���������ɫ�������̽��������������Ϻ������ɫ���ʿ�����ͭ��̼���Σ�
ͭ�����ã����������һ����ɫ���壬Ϊ�˽�ͭ�ڿ����еĸ�ʴ�����ij��ѧ��ȤС���ռ�����ͭ���������ɫ�������̽��������������Ϻ������ɫ���ʿ�����ͭ��̼���Σ�