��Ŀ����
18���������Ͻ��ƳɵĴŷ�о�ڸ�Ƶ�¾��нϺõĴ��Ժͽϵ͵���ģ����Ա��������͵��ӽ��ܲ��ϣ�����Ǧ����ĺ���Ӱ������Ǧ�Ͻ�����ܣ�ij�о�С��ͬѧ���ⶨ�úϽ������ĺ������������ͼʵ�飮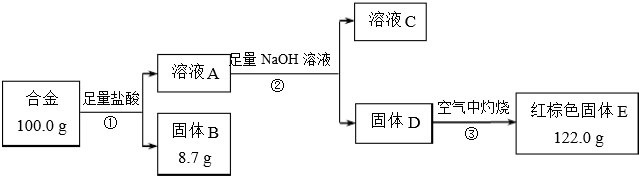
��1���١������漰��ʵ����������У��õ����˵��Ǣ٢ڣ�����ţ�
��2�����з�����Ӧ�����ӷ���ʽ��2Al+6H+=2Al3++3H2����Fe+2H+=2Fe2++H2��
��3������˵����ȷ����a������ĸ����
a����ҺC����3�������� b�����з�����������ԭ��Ӧ
c�����е�����������Һ���ð�ˮ����
��4��������ɫ����EΪ�������Ͻ���������������Ϊ5.9%��
���� �������Ͻ��м�������������Һ���������ܽ⣬����������˵ù���B����ҺA������BΪ�裬��ҺAΪ�Ȼ��������Ȼ�����Һ��A�м���������������Ƶ�ƫ������Һ����DΪ�������������˷��������D����ҺC��������ҺC�к���ƫ�������Ӻ������������Լ������ӣ����������ڿ��������յù���EΪ���������ݴ˴��⣮
��� �⣺�������Ͻ��м�������������Һ���������ܽ⣬����������˵ù���B����ҺA������BΪ�裬��ҺAΪ�Ȼ��������Ȼ�����Һ��A�м���������������Ƶ�ƫ������Һ����DΪ�������������˷��������D����ҺC��������ҺC�к���ƫ�������Ӻ������������Լ������ӣ����������ڿ��������յù���EΪ��������
��1����������ķ�����֪���١������漰��ʵ����������У��õ����˵��Ǣ٢ڣ�
�ʴ�Ϊ���٢ڣ�
��2�����з�����ӦΪ�������ᷴӦ���������ᷴӦ����Ӧ�����ӷ���ʽ�� 2Al+6H+=2Al3++3H2����Fe+2H+=2Fe2++H2����
�ʴ�Ϊ��2Al+6H+=2Al3++3H2����Fe+2H+=2Fe2++H2����
��3��a����������ķ�����֪����ҺC�к���ƫ�������Ӻ������������Լ������ӣ���a��ȷ��
b�������������������ȷֽ⣬�Ƿ�������ԭ��Ӧ����b����
c������Ҫ������������ƫ�������Ӷ��������������룬����ˮ���ܽ�����������ƫ��������ӣ���������������Һ�����ð�ˮ���棬��c����
��ѡa��
��4��������ɫ����EΪ��������������������Ϊ122g������Ԫ�ص�����Ϊ$\frac{112}{160}$��122g=122g=85.4g����Ͻ���������������Ϊ$\frac{100-85.4-8.7}{100}$��100%=5.9%��
�ʴ�Ϊ��5.9%��
���� ���⿼��ѧ����ʵ�������ԭ�������ʵķ����ᴿ�����ʺ����IJⶨ����ѧ����ȣ��Ѷ��еȣ����ʵ��ԭ���ǽ���Ĺؼ����Ƕ���ѧ֪ʶ���ۺ����ã���Ҫѧ���߱���ʵ�Ļ���������֪ʶ���������������������

| A�� | 0.1mol/L NaHCO3��Һ��0.1mol/L NaOH��Һ�������ϣ�������Һ�У�c��Na+����c��CO32-����c��HCO3-����c��OH-�� | |
| B�� | 20mL 0.1mol/L CH3COONa��Һ��10mL 0.1mol/L HCl��Һ��Ϻ���Һ�����ԣ�������Һ�У�c��CH3COO-����c��Cl-����c��CH3COOH����c��H+�� | |
| C�� | �����£�pH=2��������pH=12�İ�ˮ�������ϣ�������Һ�У�c��Cl-��+c��H+����c��NH4+��+c��OH-�� | |
| D�� | 0.1mol/L CH3COOH��Һ��0.1mol/L NaOH��Һ�������ϣ�������Һ�У�c��OH-����c��H+��+c��CH3COOH�� |
| A�� | ��СC ��D ��Ũ�� | B�� | ����ѹǿ | ||
| C�� | ��СB ��Ũ�� | D�� | ���� A ��B��Ũ�� |
| A�� | ��������Ƽ� | B�� | ��������ù�� | C�� | ������������ | D�� | ����ù�ء���Ŷ� |
| A�� | �������е�Ԫ�صĻ��ϼ�Ϊ-3 | |
| B�� | ������Ӧ��ÿ����2molAlN��N2�õ�3mol���� | |
| C�� | �ڵ������ĺϳɷ�Ӧ�У�N2����������Al2O3�ǻ�ԭ�� | |
| D�� | �÷�Ӧ���������ֻ�����Ӧ��������һ�� |
| A�� | H2SO4��K��HCl | B�� | Cl2��Al��H2 | C�� | NO2��Na��Br2 | D�� | O2��SO2��H2O |
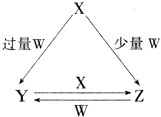 X��Y��Z��W��Ϊ��ѧ��ѧ�г����ĵ��ʻ������֮���ת����ϵ��ͼ��ʾ��ˮ�����ֲ�������ȥ����
X��Y��Z��W��Ϊ��ѧ��ѧ�г����ĵ��ʻ������֮���ת����ϵ��ͼ��ʾ��ˮ�����ֲ�������ȥ����