��Ŀ����
14�������й�ʵ��IJ�����ȷ���ǣ�������| A�� | ����ij����ҩƬ����Ҫ�ɷ�Ϊ����������Ƿ��������ʣ�ʹ����ˮ�����軯����Һ | |
| B�� | �ⶨ��ҺpHʱ������pH��ֽպȡ��������Һ���������ɫ���Ƚ� | |
| C�� | ����ƿ����Ͳ�͵ζ����϶�����ʹ���¶ȣ���Ͳ������ƿ�ޡ�0���̶ȣ��ζ����С�0���̶ȣ�ʹ��ʱ�ζ���ˮϴ������ϴ��������ƿˮϴ������ϴ | |
| D�� | �ⶨ̼������̼�����Ƶĺ�����ȡa����Ʒ������ϡ�����ַ�Ӧ���ݳ�������ͨ����ʯ�Һ�ʯ������b�ˣ�����a��b��ֵ�������̼�����Ƶĺ��� |
���� A��Fe3+�ܺ�SCN-��������ʹ��Һ����Ѫ��ɫ�������ԣ�����Ҫ��ˮ��
B����pH��ֽպȡ��������Һ����Ⱦ�Լ���
C������ƿ����Ͳ�͵ζ����϶�����ʹ���¶ȣ���Ͳ������ƿ�ޡ�0���̶ȣ��ζ����С�0���̶ȣ�ʹ��ʱ�ζ���ˮϴ������ϴ����ֹ�ڱڵ�ˮϡ����Һ����Һ������Ҫ��ˮ���ݣ�����ƿˮϴ������ϴ��
D����ʯ������Ϊ���ɶ�����̼��������̼�л��е�ˮ���������������㣮
��� �⣺A��ʹ����ˮ�����軯����Һ���Լ����������ӣ�ҩƷ���ʣ��������ᱻ����Ϊ��������Fe3+�ܺ�SCN-��������ʹ��Һ����Ѫ��ɫ�������ԣ�����Ҫ��ˮ����A����
B����pH��ֽպȡ��������Һ����Ⱦ�Լ���Ӧ��һС����ֽ���ڱ������ϣ��ò���պȡ����ҺͿ��pH��ֽ�ϣ��������ɫ�����գ���B����
C������ƿ����Ͳ�͵ζ����϶�����ʹ���¶ȣ���Ͳ������ƿ�ޡ�0���̶ȣ��ζ����С�0���̶ȣ�ʹ��ʱ�ζ���ˮϴ������ϴ����ֹ�ڱڵ�ˮϡ����Һ����Һ������Ҫ��ˮ���ݣ�����ƿˮϴ������ϴ����C��ȷ��
D����ʯ������Ϊ���ɶ�����̼��������̼�л��е�ˮ���������������㣬Ӧ�Ƚ��и�����ü�ʯ�����ն�����̼����D����
��ѡ��C��
���� ���⿼��ʵ�鷽�������ۡ�ʵ�������������ѧ�����ȣ�ע�����ճ������Ӽ����Լ����û�ѧ�����ṹ���滭��ʹ�ã�

| A�� | 1mol�������ƺ�����ˮ��ַ�Ӧ��������ת�Ƶĵ�����ԼΪ2��6.02��1023 | |
| B�� | �繤�����н�������ͭ��ֱ���������ᵼ��ͭ�߸��챻���� | |
| C�� | MnS����Һ�еμ�����CuSO4��Һ������CuS��������Ksp��CuS����Ksp��MnS�� | |
| D�� | 0.1mol•L-1������ҺpH=a��0.01mol•L-1������ҺpH=b����a+1��b |
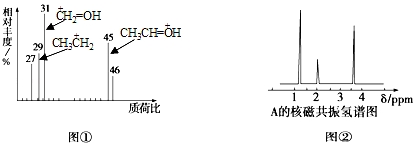
| A�� | HCOOH | B�� | CH3CHO | C�� | CH3CH2OH | D�� | CH3CH2COOH |
| A�� | С��0.8 | B�� | ����1.2 | C�� | ��0.8��1.2֮�� | D�� | ��0.8��1.5֮�� |
| A�� | CH4 | B�� | C2H6 | C�� | C2H5OH | D�� | CH3CHO |
| A�� | CH3COONa | B�� | CH3COOH��CH3COONa | ||
| C�� | CH3COONa��NaOH | D�� | CH3COOH��NaOH |
 ijʵ��С����H2O2�ֽ�Ϊ�����о�Ũ�ȡ���������Һ����ԶԷ�Ӧ���ʵ�Ӱ�죮�ڳ����°�������������ʵ�飮
ijʵ��С����H2O2�ֽ�Ϊ�����о�Ũ�ȡ���������Һ����ԶԷ�Ӧ���ʵ�Ӱ�죮�ڳ����°�������������ʵ�飮| ʵ���� | ��Ӧ�� | ���� |
| �� | 10mL2% H2O2��Һ | �� |
| �� | 10mL5% H2O2��Һ | �� |
| �� | 10mL5% H2O2��Һ | 1mL0.1mol•L-1FeCl3��Һ |
| �� | 10mL5% H2O2��Һ+����HCl��Һ | 1mL0.1mol•L-1FeCl3��Һ |
| �� | 10mL5% H2O2��Һ+����NaOH��Һ | 1mL0.1mol•L-1FeCl3��Һ |
A�������˷�Ӧ�Ļ��B�������˷�Ӧ������
C�������˷�Ӧ���ʱ�D�������˷�Ӧ��ƽ�ⳣ��
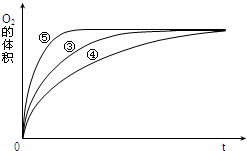
��2��ʵ��ٺ͢ڵ�Ŀ����̽��Ũ�ȶԷ�Ӧ���ʵ�Ӱ�죮ʵ��ʱ����û�й۲쵽������������ó����ۣ�������ʾ��ͨ��������H2O2�ȶ������ֽ⣮Ϊ�˴ﵽʵ��Ŀ�ģ����ԭʵ�鷽���ĸĽ�����Ӧ���м������ͬ�ִ�������ʢ�з�Ӧ����Թܷ���ͬһ��ˮԡ�У���
��3��ʵ��ۡ��ܡ����У�������������������ʱ��仯�Ĺ�ϵ��ͼ������ͼ�ܹ��ó���ʵ������Ǽ��Ի���������H2O2�ֽ�����ʣ����Ի����ܼ�СH2O2�ֽ�����ʣ�
 ij���Թ�ҵ��ˮ�к���K2Cr2O7�������£����ᣨH2C2O4���ܽ����е�Cr2O72-ת��ΪCr3+��ij�������о����֣�����������[Al2Fe��SO4��4•24H2O]���ɶԸ÷�Ӧ������ã�Ϊ��һ���о��й����ضԸ÷�Ӧ���ʵ�Ӱ�죬̽�����£�
ij���Թ�ҵ��ˮ�к���K2Cr2O7�������£����ᣨH2C2O4���ܽ����е�Cr2O72-ת��ΪCr3+��ij�������о����֣�����������[Al2Fe��SO4��4•24H2O]���ɶԸ÷�Ӧ������ã�Ϊ��һ���о��й����ضԸ÷�Ӧ���ʵ�Ӱ�죬̽�����£���1����25���£����ƹ���ǿ�ȡ���ˮ��Ʒ��ʼŨ�Ⱥʹ���������ͬ�����ڲ�ͬ�ij�ʼpH��һ��Ũ�Ȳ�����Һ���������Ա�ʵ�飬�������ʵ����Ʊ������в�Ҫ���ո�
| ʵ���� | ��ʼpH | ��ˮ��Ʒ���/mL | ������Һ���/mL | ����ˮ���/mL |
| �� | 4 | 60 | 10 | 30 |
| �� | 5 | 60 | 10 | 30 |
| �� | 5 | 60 |
��2��������Ӧ����ᱻ���������ӷ���ʽΪCr2O72-+3H2C2O4+8H+=6CO2��+2Cr3++7H2O
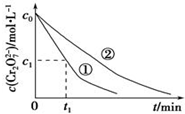
��3��ʵ��ٺ͢ڵĽ��������ҺPH�Ը÷�Ӧ��������Ӱ�죻ʵ�����0��t1ʱ��η�Ӧ����v��Cr3+��=$\frac{2��{c}_{0}-{c}_{1}��}{{t}_{1}}$mol•L-1•min-1���ô���ʽ��ʾ����
��4���ÿ������������[Al2Fe��SO4��4•24H2O]��������õijɷ�������¼��裬������ɼ�����ͼ�������
����һ��Fe2+������ã�
�������Al3+������ã�
��������SO42-������ã�
��
��5���������ʵ����֤��������һ������±������ݣ�
����������ʵ���ṩ���Լ��⣬�ɹ�ѡ���ҩƷ��K2SO4��FeSO4��K2SO4•Al2��SO4��3•24H2O��Al2��SO4��3�ȣ���Һ��Cr2O72-��Ũ�ȿ��������ⶨ��
| ʵ�鷽������Ҫ��д����������̣� | Ԥ��ʵ�����ͽ��� |
| A�� | X��ԭ������һ������Y | B�� | ��һ������Y���ܴ���X | ||
| C�� | �����ӵİ뾶��Xm-��Yn- | D�� | ��̬�⻯����ȶ��ԣ�HmXǿ��HnY |