��Ŀ����
�������ƾ���[CaO2��8H2O]���ȶ����ʰ�ɫ������ˮ��������������Һ���㷺Ӧ���ڻ���ɱ��������������
��.�������ƾ�����Ʊ���
��ҵ������CaO2��8H2O����Ҫ�������£�

��1��������������ȡCaO2��8H2O�Ļ�ѧ����ʽ��_________________________________��
��2������ʱ���ñ�ˮ�����¶���10 �����º�ͨ�������NH3�������ԭ��ֱ�����__________________________����_____________________________��
��.�������ƾ��庬���IJⶨ��
ȷ��ȡ0.300 0 g��Ʒ����ƿ�У�����30 mL����ˮ��10 mL 2.000 mol��L��1 H2SO4����0.020 0 mol��L��1 KMnO4����Һ�ζ����յ㡣�ظ������������Ρ�H2O2��KMnO4��Ӧ�����ӷ���ʽΪ2MnO4-��5H2O2��6H��=2Mn2����5O2����8H2O
�ζ��յ�۲쵽������Ϊ_______________________________________��
��4�����ݱ������ݣ������Ʒ��CaO2��8H2O������������д��������̣���
KMnO4����Һ�ζ�����
�ζ����� | ��Ʒ������/g | KMnO4��Һ�����/mL | |
�ζ�ǰ�̶�/mL | �ζ���̶�/mL | ||
1 | 0.300 0 | 1.02 | 24.04 |
2 | 0.300 0 | 2.00 | 25.03 |
3 | 0.300 0 | 0.20 | 23.24 |
��.��1��CaCl2��H2O2��2NH3��8H2O=CaO2��8H2O����2NH4Cl����2�����¶ȵͿɼ��ٹ�������ķֽ⣬��߹�������������ʣ����ֹ��������ķֽ⣩����ͨ�����NH3ʹ��Һ�ʼ��ԣ�����CaO2��8H2O���ܽ⣨��ʹ��Һ�ʼ��ԣ�����CaO2��8H2O���ܽ⣬����߲�Ʒ�IJ��ʣ�
��.��3�����������һ��KMnO4����Һ����Һ����ɫ��Ϊdz�Ϻ�ɫ����30 s����ɫ
��4�����ݷ�Ӧ����ʽ2MnO4-��5H2O2��6H��=2Mn2����5O2����8H2O��CaCl2��H2O2��2NH3��8H2O=CaO2��8H2O����2NH4Cl����������ʽ��
5��CaO2��8H2O����5H2O2��2KMnO4
n��CaO2��8H2O����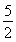 n��KMnO4����
n��KMnO4����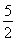 ��0.020 0 mol��L��1��23.03 mL��10��3 L��mL��1��1.151 5��10��3 mol
��0.020 0 mol��L��1��23.03 mL��10��3 L��mL��1��1.151 5��10��3 mol
CaO2��8H2O������������ ��100%��82.91%��
��100%��82.91%��
����������1����������ͼ�еļ�ͷָ��ȷ����Ӧ����CaCl2��H2O2��NH3��������CaO2��8H2O�����÷�Ӧǰ��Ԫ���غ㣬ȷ����һ��ӦΪH2O��������ΪNH4Cl���ʷ�Ӧ�Ļ�ѧ����ʽΪCaCl2��H2O2��2NH3��8H2O=CaO2��8H2O����2NH4Cl����2��30%H2O2�����ֽ⣬��˳���ʱ���ñ�ˮ�����¶���10 �����¼��ٹ�������ķֽ⣬��߹�������������ʣ�����ͨ��NH3�IJ���Ϊ��������������ͨ�����NH3ʹ��Һ�ʼ��ԣ�����CaO2��8H2O���ܽ⣬��ʹ���������3��������0.020 0 mol��L��1 KMnO4����Һ����ɫ�����еζ������Եζ��յ�۲쵽������Ϊ���������һ��KMnO4����Һ����Һ����ɫ��Ϊdz�Ϻ�ɫ����30 s�ڲ���ɫ��

 ��У����ϵ�д�
��У����ϵ�д����ڿ��淴ӦN2��g����3H2��g�� 2NH3��g������H��0�������о�Ŀ�ĺ�ʾ��ͼ������ǣ���������
2NH3��g������H��0�������о�Ŀ�ĺ�ʾ��ͼ������ǣ���������
| A | B |
�о� Ŀ�� | ѹǿ�Է�Ӧ��Ӱ �죨p2��p1�� | �¶ȶԷ�Ӧ��Ӱ�� |
ͼʾ |
|
|
�о� Ŀ�� | ƽ����ϵ����N2 �Է�Ӧ��Ӱ�� | �����Է�Ӧ��Ӱ�� |
ͼʾ |
|
|
W��X��Y��Z���ֶ�����Ԫ����Ԫ�����ڱ��е����λ����ͼ��ʾ��W����̬�⻯���������ۺ����ᷴӦ�������ӻ��������˵������ȷ���ǣ���������
W | X |
|
| Y | Z |
A��X��Y��Z������⻯���ȶ�����������Y
B��ZԪ���������Ӧˮ���������һ��ǿ��Y
C��XԪ���γɵĵ��������ӻ�ԭ������Y
D��YZ2�����и�ԭ�ӵ�����������8e���ȶ��ṹ










