��Ŀ����
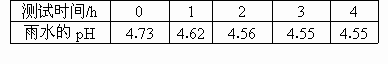
�ҹ�ũҵ������������ÿ����ʧ�ߴ�l5�ڶ�Ԫ��Ϊ����Ч�������꣬����Ժ�����ˡ�����������Ͷ���������Ⱦ���������ַ������ȷ��档(1)������ˮ��Ʒl�ݣ�ÿ��һ��ʱ��ⶨ����ˮ��Ʒ��pH�������������£�
����ʱ��/h | 0 | 1 | 2 | 3 | 4 |
��ˮ��Ph | 4.73 | 4.62 | 4.56 | 4.55 | 4.55 |
�������ݣ��ش��������⣺
����ˮ��Ʒ��pH�仯��ԭ����(�û�ѧ����ʽ��ʾ)_________________________________��
���������ȡ����������ˮ������ˮ���ϣ�pH����_______��ԭ����_________________(�û�ѧ����ʽ��ʾ) ��
(2)����Ϊ�������������;���ɲ��õĴ�ʩ��_________��
������ú��ȼ�ϡ��ڰѹ����̴���ߡ���ȼ�����������ữ�������м�ʯ�ҡ��ݿ�������Դ
A.�٢ڢ� B.�ڢۢܢ�
C.�٢ۢ� D.�٢ۢܢ�
��������ˮ����ʱpH�仯����ԭ������ˮ�е�H2SO3����������������ΪH2SO4���¡���ˮ������ˮ��ϣ�Ӧ����H2SO3��Cl2(��HClO)�ķ�Ӧ����������Ӧ�Ӹ����϶ž������SO2���ŷš�
�𰸣�(1)��2H2SO3+O2![]() 2H2SO4
2H2SO4
����H2SO3+HClO![]() H2SO4+HCl ��SO2+2H2O+Cl2
H2SO4+HCl ��SO2+2H2O+Cl2![]() H2SO4+2HCl
H2SO4+2HCl
(2)C

��ϰ��ϵ�д�
�����Ŀ