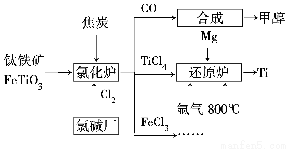
جâؤ؟ؤعبف
ؤ³»¯ر§ر§د°ذ،×éةè¼ئدآح¼تµرé×°ضأ(¼ذ³ض×°ضأآشب¥)ضئ±¸Cl2£¬²¢ج½¾؟آبئّµؤدà¹طذشضت،£

(1)بôA×°ضأضذ¹ججهز©ئ·خھKClO3£¬شٍ·´س¦ضذأ؟ةْ³ة1 mol Cl2ت±×ھزئµç×سµؤخïضتµؤء؟خھ________mol،£
(2)×°ضأB¼بؤـ³ب¥آبئّضذµؤآب»¯ا⣬»¹ؤـ¼ى²âتµرé¹³جضذ×°ضأCتا·ٌ·¢ةْ¶آبû،£بôCضذ·¢ةْ¶آبû£¬شٍBضذ½«¹غ²ىµ½µؤدضدَتا_______________________________________،£
(3)×°ضأCµؤ×÷سأتارéض¤آبئّتا·ٌ¾كسذئ¯°×ذش£¬¢ٌ´¦تاتھبَµؤسذة«²¼جُ£¬شٍ¢ٍ،¢¢َ´¦س¦¼سبëµؤخïضت·ض±ًتا________،¢________،£
(4)ةè¼ئ×°ضأD،¢Eµؤؤ؟µؤتا±ب½دآب،¢نه،¢µâµؤ·ا½ًتôذش،£اë¼ٍتِؤـثµأ÷آب،¢نه،¢µâ·ا½ًتôذشا؟بُµؤتµرé²ظ×÷¼°دضدَ£؛_____________________________________________،£
(5)اëسأ»¯ر§·½³جت½ثµأ÷×°ضأFµؤ×÷سأ£؛_______________________________________،£
(6)¼×ح¬ر§جل³ِ£¬×°ضأFضذµؤتش¼ء؟ة¸ؤسأ×مء؟µؤNa2SO3بـز؛£¬ززح¬ر§بدصوث¼؟¼؛َبدخھ´ث·¨²»؟ةذذ،£اëسأہë×س·½³جت½½âتحززبدخھ²»؟ةذذµؤشزٍ£؛_______________________،£
(1)5/3،،(2)Bضذ³¤¾±آ©¶·ضذز؛أوةدة£¬ذخ³ةز؛ضù،،(3)خقث®آب»¯¸ئ(»ٍ¹è½؛،¢P2O5)،،¸ةشïµؤسذة«²¼جُ
(4)´ٍ؟ھAضذ·ضز؛آ©¶·µؤ»îبû£¬ز»¶خت±¼ن؛َ£¬Dضذµؤخقة«بـز؛±نخھ³بة«(³ب؛ىة«»ٍ»ئة«)£¬ثµأ÷آبµؤ·ا½ًتôذشا؟سعنه£»´ٍ؟ھDضذ»îبû£¬½«Dضذةظء؟بـز؛·إبë×°ضأEضذ£¬صٌµ´£¬دآ²مخھ×د؛ىة«£¬ثµأ÷نهµؤ·ا½ًتôذشا؟سعµâ
(5)Cl2£«2NaOH=NaCl£«NaClO£«H2O
(6)SO32،ھ£«Cl2£«H2O=SO42،ھ£«2Cl££«2H£«،¢SO32،ھ£«2H£«=SO2،ü£«H2O
،¾½âخِ،؟(1)A×°ضأضذ·¢ةْ·´س¦µؤ»¯ر§·½³جت½خھKClO3£«6HCl(إ¨)=KCl£«3Cl2،ü£«3H2O£¬سة»¯ر§·½³جت½؟ةضھ£¬·´س¦ضذأ؟ةْ³ة1 mol Cl2ت±×ھزئ5/3molµç×س،£(2)µ±×°ضأCضذ·¢ةْ¶آبûت±£¬½«µ¼ضآB×°ضأµؤ׶ذخئ؟ضذئّجهر¹ا؟شِ´َ£¬´س¶ّت¹Bضذ³¤¾±آ©¶·ضذز؛أوةدة£¬ذخ³ةز؛ضù،£(3)آبئّؤـت¹تھبَµؤسذة«²¼جُحتة«£¬¹ت¢َ´¦س¦¼سبë¸ةشïµؤسذة«²¼جُ£¬؛ح¢ٌ´¦تھبَµؤسذة«²¼جُ×÷¶ش±ب£¬¢ٍ´¦س¦¼س¹ججه¸ةشï¼ء،£(4)ہûسأآ±ثطµ¥ضتض®¼نµؤضأ»»·´س¦±ب½دشھثط·ا½ًتôذشµؤا؟بُ،£(5)Fخھخ²ئّخüتص×°ضأ£¬·¢ةْ·´س¦µؤ»¯ر§·½³جت½خھCl2£«2NaOH=NaCl£«NaClO£«H2O،£(6)بô½«Fضذµؤتش¼ء»»خھNa2SO3بـز؛£¬شٍ»ل²ْةْخغب¾»·¾³µؤSO2ئّجه،£

 أد½¨ئ½´يجâ±¾دµءذ´ً°¸
أد½¨ئ½´يجâ±¾دµءذ´ً°¸ ³¬ؤـر§µنس¦سأجâجâ؟¨دµءذ´ً°¸
³¬ؤـر§µنس¦سأجâجâ؟¨دµءذ´ً°¸ؤ³ذ،×éب،ز»¶¨ضتء؟µؤFeSO4¹ججه£¬ہûسأدآح¼×°ضأ½ّذذتµرé،£

تµرé | تµرé¹³ج | تµدضدضدَ |
¢ظ | ح¨بëز»¶خت±¼نN2£¬¼سبب | Aضذ¹ججه±نخھ؛ى×طة«£¬Bضذسذ°×ة«³ءµي£¬Dتش¹ـضذسذخقة«ز؛جه |
¢ع | سأ´ّسذ»ًذاµؤؤ¾جُ؟؟½ü×°ضأDµؤµ¼¹ـ؟ع | ؤ¾جُ¸´ب¼ |
¢غ | ³ن·ض·´س¦£¬ح£ض¹¼سبب£¬ہنب´؛َ£¬ب،Aضذ¹ججه£¬¼سرخثل | ¹ججهبـ½â£¬بـز؛³ت»ئة« |
¢ـ | ½«¢غثùµأبـز؛µخبëDتش¹ـضذ | بـز؛±نخھا³آجة« |
زرضھ£؛SO2بغµم£72،و£¬·ذµم£10،و£»SO3بغµم16.8،و£¬·ذµم44.8 ،و،£
(1)تµرé¢غ·´س¦µؤہë×س·½³جت½تا_____________________________________________،£
(2)·ض½â¹³ج³²ْةْت¹ؤ¾جُ¸´ب¼µؤئّجهح⣬½ِسةAضذ¹ججهرصة«±ن»¯حئ²â£¬»¹ز»¶¨سذ________ئّجه£¬زہ¾فتا____________________________________________________
(3)تµرé¢ـ·´س¦µؤہë×س·½³جت½تا_____________________________________________،£
(4)ؤ³ح¬ر§زہ¾فBضذµؤدضدَ£¬بدخھFeSO4·ض½âز»¶¨سذSO3ةْ³ة،£ؤمبدخھتا·ٌصب·£¬²¢ثµأ÷شزٍ£؛____________________________________________(سأ±طزھµؤخؤ×ض؛ح»¯ر§·½³جت½½âتح)،£
خآ¶بخھT،وت±£¬دٍV Lµؤأـ±صبفئ÷ضذ³نبëز»¶¨ء؟µؤA؛حB£¬·¢ةْµؤ·´س¦خھA(g)£«B(g) C(s)£«xD(g)،،¦¤H£¾0£¬بفئ÷ضذA،¢B،¢Dµؤخïضتµؤء؟إ¨¶بثوت±¼نµؤ±ن»¯بç±يثùت¾،£دآءذثµ·¨²»صب·µؤتا(،،،،)
C(s)£«xD(g)،،¦¤H£¾0£¬بفئ÷ضذA،¢B،¢Dµؤخïضتµؤء؟إ¨¶بثوت±¼نµؤ±ن»¯بç±يثùت¾،£دآءذثµ·¨²»صب·µؤتا(،،،،)
ت±¼ن | 0 | 5 min | 10 min | 15 min | 20 min | 25 min | 30 min |
Aµؤخïضتµؤء؟إ¨¶ب(mol،¤L£1) | 3.5 | 2.3 | 2.0 | 2.0 | 2.8 | 3.0 | 3.0 |
Bµؤخïضتµؤء؟إ¨¶ب(mol،¤L£1) | 2.5 | 1.3 | 1.0 | 1.0 | 1.8 | 2.0 | 2.0 |
Dµؤخïضتµؤء؟إ¨¶ب(mol،¤L£1) | 0 | 2.4 | 3.0 | 3.0 | 1.4 | 1.0 | 1.0 |
A.ا°10 minµؤئ½¾ù·´س¦ثظآتv(D)£½0.3 mol،¤L£1،¤min£1
B£®¸أ·´س¦µؤئ½؛â³£ت±ي´ïت½خھK£½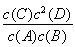
C£®بô´ïµ½ئ½؛âت±±£³ضخآ¶ب²»±ن£¬ر¹ثُبفئ÷جه»£¬ئ½؛â²»زئ¶¯
D£®·´س¦ضء15 minت±£¬¸ؤ±نµؤجُ¼؟ةزشتا½µµحخآ¶ب