��Ŀ����
�������ӷ���ʽ��д��ȷ���ǣ�������
| A����������Һ�м������������������Һ��2Al3++3SO42-+6OH-+3Ba2+=2Al��OH��3��+3BaSO4�� |
| B���������������м���������ϡ���Fe��OH��2+2H+=Fe2++2H2O |
| C����̼�������Һ�м�������������������Һ��NH4++HCO3-+2OH-=CO32-+NH3?H2O+H2O |
| D����CH2BrCOOH�м�������������������Һ�����ȣ�CH2BrCOOH+OH-��CH2BrCOO-+H2O |
���㣺���ӷ���ʽ����д
ר�⣺���ӷ�Ӧר��
������A����������Һ�м������������������Һ����Ӧ�������ᱵ��ƫ����أ�
B���������������л�ԭ���ܱ����������������������ӣ�
C��̼�������Һ�м����������������ƣ���Ӧ����̼���ƺ�һˮ�ϰ���ˮ��
D��CH2BrCOOH�м�������������������Һ�����ȣ���Ӧ����CH2OHCOONa��H2O��
B���������������л�ԭ���ܱ����������������������ӣ�
C��̼�������Һ�м����������������ƣ���Ӧ����̼���ƺ�һˮ�ϰ���ˮ��
D��CH2BrCOOH�м�������������������Һ�����ȣ���Ӧ����CH2OHCOONa��H2O��
���
�⣺A����������Һ�м������������������Һ����Ӧ�����ӷ���ʽΪ��Al3++SO42-+Ba2++4OH-=AlO2-+BaSO4��+2H2O����A����
B���������������м���������ϡ���ᣬ��Ӧ�����ӷ���ʽΪ��3Fe��OH��2+10��H+��+NO3-=3��Fe3+��+NO��+5H20����B����
C����̼�������Һ�м�������������������Һ����Ӧ�����ӷ���ʽ��NH4++HCO3-+2OH-=CO32-+NH3?H2O+H2O����C��ȷ��
D����CH2BrCOOH�м�������������������Һ�����ȣ���Ӧ�����ӷ���ʽΪ��CH2BrCOOH+2OH-��CH2OHCOO-+H2O+Br-����D����
��ѡ��C��
B���������������м���������ϡ���ᣬ��Ӧ�����ӷ���ʽΪ��3Fe��OH��2+10��H+��+NO3-=3��Fe3+��+NO��+5H20����B����
C����̼�������Һ�м�������������������Һ����Ӧ�����ӷ���ʽ��NH4++HCO3-+2OH-=CO32-+NH3?H2O+H2O����C��ȷ��
D����CH2BrCOOH�м�������������������Һ�����ȣ���Ӧ�����ӷ���ʽΪ��CH2BrCOOH+2OH-��CH2OHCOO-+H2O+Br-����D����
��ѡ��C��
���������⿼�������ӷ���ʽ����д����ȷ��ѧ��Ӧ��ʵ���ǽ���Ĺؼ���ע��Dѡ��Ϊ�״�ѡ�CH2BrCOOH��Br���ױ�ȡ��ת��Ϊ�ǻ���

��ϰ��ϵ�д�
�����Ŀ
���л�ѧ���������ȷ���ǣ�������
| A�����ǵķ���ʽ��C6H12O6 |
B��NH3�ĵ���ʽ��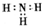 |
C������ı���ģ�ͣ� |
| D��������̼�Ľṹʽ��O-C-O |
����������������ȵĴ���ʯ���ֱ�Ͷ������ļ���Һ��0.1mol/L HCl��������Һ��0.1mol/L���ᣩ�У�������
| A����ʼ�����ʼס�����ͬ�����ɶ�����̼�������ס���Ҳ��ͬ |
| B����ʼ�����ʼ״����ң����ɶ�����̼������Ҳ�Ǽ״����� |
| C����ʼ�����ʼ״����ң����ɶ�����̼�������ס�����ͬ |
| D����Ӧ���ʺ����ɶ�����̼������������ͬ |
���й��ڰ���ӵ�������NA��˵����ȷ���ǣ�������
| A�����³�ѹ�£������ý���ת��2NA������ʱ���ɴ��������û�����22.4LH2 |
| B��1mol�������������������Ʒ�Ӧ����ת����NA�ĵ��� |
| C����״���£�2.24L����Ͱ����Ļ�����������ĵ�����ΪNA |
| D��1mol/L��Na2S��Һ�У����е�S2-����ĿС��NA |
�������ǿ��֮�֣�����������ʵ������ǣ�������
| A������������Һ | B��ˮ |
| C���Ȼ��ƹ��� | D�����ᱵ |
����˵���д�����ǣ�������
| A����������ͬ��ԭ�ӣ����������Ų�Ҳ��ͬ |
| B����������ͬ��ԭ�ӣ��仯ѧ����һ����ͬ |
| C��������Խ���ã���ԭ���ڷ�Ӧ��Խ��ʧȥ���� |
| D���ǽ�����Խ���ã���������Խ��ʧȥ���� |
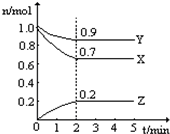 ij�¶�ʱ����һ��2L���ܱ������У�X��Y��Z�������ʵ����ʵ�����ʱ��ı仯������ͼ��ʾ������ͼ�����ݣ�����д���пհף�
ij�¶�ʱ����һ��2L���ܱ������У�X��Y��Z�������ʵ����ʵ�����ʱ��ı仯������ͼ��ʾ������ͼ�����ݣ�����д���пհף�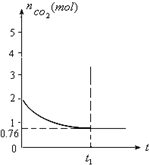 һ���¶���2���ĺ����������У�����2moL̼��2moLCO2�������·�Ӧ��
һ���¶���2���ĺ����������У�����2moL̼��2moLCO2�������·�Ӧ��