��Ŀ����
9����������Ca2+��Mg2+��SO42-�Լ���ɳ�����ʣ�ijͬѧ�����һ���Ʊ����ε�ʵ�鷽�����������£����ڳ������Լ��Թ�������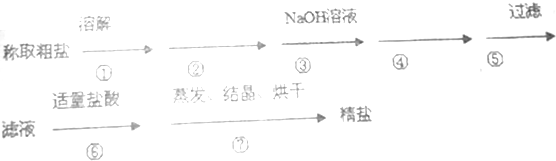
��1���������Ӧ������Լ���BaCl2��Һ���жϴ��Լ��Ѽӹ����ķ�����ȡ������Һ���ϲ���Һ1��2���ڵζ����ϣ��ٵ���1��2��BaCl2��Һ������Һδ����ǣ�����BaCl2�ѹ�����
��2���������Ӧ������Լ���Na2CO3��Һ���ڢܲ�����صĻ�ѧ����ʽ��Ba2++CO32-=BaCO3����Ca2++CO32-=CaCO3����
��3�������ڢݲ��͵ڢ��ߵ������ʵ��������Ӱ�죬��ԭ�����ڴ���������£����в��ֳ����ܽ⣬�Ӷ�Ӱ���Ƶþ��εĴ��ȣ�
��4���ڢۢ��������Լ���˳��Ҳ���Ե���ΪA��
A���ۢڢ�B���ܢۢ�C���ڢܢ�D���ܢڢ�
��5��Ϊ���������Һ��Cl-��SO42-�������������գ�
�����ȼ���SO42-���ӣ�Ӧ�ȼ��������Ba��NO3��2��
�ڽ����û������ˣ�Ȼ������Һ�м����Լ������ữ����������Һ������һ���ӣ�
���� ��ȥ�����е�Ca2+��Mg2+��SO42-����ɳ����ˮ�ܽ������Ҫ����������ڳ�ȥ�������������ƺ�̼���ƣ�Ҫ�ȼӹ������Ȼ�����ȥ��������ӣ�Ȼ����̼����ȥ�������ı����ӣ�����������̼������ҺӦ���ڼ����Ȼ�����Һ֮����������ڹ���֮����룬����м��Ȼ����ɳ�ȥ��������ӣ����м�NaOH��ȥþ���ӣ����м�̼���ƿɳ�ȥ�����ӡ������ӣ����˺���ҪΪ�Ȼ��ƺ�̼���ƣ����м����ᷴӦ����Һ������ΪNaCl��Ȼ�������ᾧ��ʣ������Һ��ֹͣ���ȣ��õ�NaCl���Դ˽��ã�
��1�������Ϸ�����֪������м�����Լ��� BaCl2���Ȼ�������ʱ����������������ɫ������
��2�����м�����Լ���Na2CO3 ��
��3��Mg��OH��2��CaCO3��BaCO3�������ᷴӦ�������Ȼ�þ���Ȼ��ơ��Ȼ��������ʣ���Ӱ���Ƶþ��εĴ��ȣ�
��4����ȥ�����е�Ca2+��Mg2+��SO42-����ɳ����ˮ�ܽ������Ҫ����������ڳ�ȥ�������������ƺ�̼���ƣ�Ҫ�ȼӹ������Ȼ�����ȥ��������ӣ�Ȼ����̼����ȥ�������ı����ӣ�����������̼������ҺӦ���ڼ����Ȼ�����Һ֮����������ڹ���֮����룻
��5���Ȼ��������ᱵ���Dz�����ϡ����İ�ɫ�������ڼ���������ʱ���Ǽ������������ְ�ɫ������֤�������ӵĴ��ڣ�����������������ˮ�ģ�������Ҳ�ͱ�ɳ����ˣ�����Ҫ�ȼ�����������ӣ��ټ��������ӣ�
��� �⣺�����̿�֪�����м��Ȼ����ɳ�ȥ��������ӣ����м�NaOH��ȥþ���ӣ����м�̼���ƿɳ�ȥ�����ӡ������ӣ����˺���ҪΪ�Ȼ��ƺ�̼���ƣ����м����ᷴӦ����Һ������ΪNaCl��Ȼ�������ᾧ�õ�NaCl��
��1�������Ϸ�����֪������м�����Լ��� BaCl2��Һ���жϴ��Լ��Ѽӹ����ķ����ǣ���������������ɫ����������ͨ�����鱵������ȷ���Ȼ����Ƿ����������ȡ������Һ���ϲ���Һ1��2���ڵζ����ϣ��ٵ���1��2��BaCl2��Һ������Һδ����ǣ�����BaCl2�ѹ�����
�ʴ�Ϊ��BaCl2��ȡ������Һ���ϲ���Һ1��2���ڵζ����ϣ��ٵ���1��2��BaCl2��Һ������Һδ����ǣ�����BaCl2�ѹ�����
��2�����м�����Լ���Na2CO3����ȥ�����Ӻͱ����ӣ���Ӧ�����ӷ���ʽΪ��Ba2++CO32-=BaCO3����Ca2++CO32-=CaCO3����
�ʴ�Ϊ��Na2CO3��Ba2++CO32-=BaCO3����Ca2++CO32-=CaCO3����
��3�������ڢݲ��͵ڢ��ߵ������������pH�ٹ��ˣ�����Mg��OH��2��CaCO3��BaCO3�������ᷴӦ������������ˮ���Ȼ�þ���Ȼ��ơ��Ȼ��������ʣ��Ӷ�Ӱ���Ȼ��ƵĴ��ȣ�
�ʴ�Ϊ���ڴ���������£����в��ֳ����ܽ⣬�Ӷ�Ӱ���Ƶþ��εĴ��ȣ�
��4����ȥ�����е�Ca2+��Mg2+��SO42-����ɳ����ˮ�ܽ������Ҫ����������ڳ�ȥ�������������ƺ�̼���ƣ�Ҫ�ȼӹ������Ȼ�����ȥ��������ӣ�Ȼ����̼����ȥ�������ı����ӣ�����������̼������ҺӦ���ڼ����Ȼ�����Һ֮����������ڹ���֮����룬�ڢۢ��������Լ���˳��Ҳ���Ե���Ϊ�ۢڢܣ�
�ʴ�Ϊ��A��
��5�����ڼ���������ʱ���Ǽ������������ְ�ɫ������֤�������ӵĴ��ڣ�����������������ˮ�ģ�������Ҳ�ͱ�ɳ����ˣ�����Ҫ�ȼ�����������ӣ��ټ��������ӣ��ȼ�����������ӵĴ��ڣ�����������Լ������ᱵ��
�ʴ�Ϊ��SO42-��Ba��NO3��2��
�ڹ��˳������������ữ����������Һ���������ӵĴ��ڣ�
�ʴ�Ϊ�������ữ����������Һ��
���� ���⿼����������ᴿ�����ӷ�Ӧ�����Ӽ��飬Ϊ��Ƶ���㣬�������ʵ����ʡ��������뷽��Ϊ���Ĺؼ������ط�����ʵ�������Ŀ��飬ע�����Ӽ��鼰������ϴ�ӣ���Ŀ�Ѷ��еȣ�

 �������ϵ�д�
�������ϵ�д�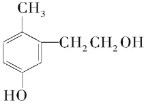
| A�� | ���DZ��ӵ�ͬϵ�� | |
| B�� | 1 mol������������ˮ��Ӧ����2molBr2����ȡ����Ӧ | |
| C�� | 1 mol���л����������������Ʒ�Ӧ����0.5mol H2 | |
| D�� | 1 mol���л�������2molNaOH��Ӧ | |
| E�� | �������ڼ��������¿�����ͭ��Ӧ����һ�־���ȩ���Ļ����� |
| t/s | 0 | 50 | 150 | 250 | 350 |
| n��PCl3��/mol | 0 | 0.16 | 0.19 | 0.20 | 0.20 |
| A�� | ��Ӧ��ǰ50s��ƽ������Ϊv��PCl3��=0.0016mol•L-1•s-1 | |
| B�� | ���������������䣬�����¶ȣ�ƽ��ʱ��c��PCl3��=0.11mol•L-1����Ӧ�ġ�H��0 | |
| C�� | ��ͬ�¶��£���ʼʱ�������г���1.0molPCl5��0.20molPCl3��0.20molCl2���ﵽƽ��ǰv��������v���棩 | |
| D�� | ��ͬ�¶��£���ʼʱ�������г���2.0molPCl3��2.0molCl2���ﵽƽ��ʱ��PCl3��ת����С��80% |
| A�� | ���ࡢ���ס����� | B�� | ���ܲ���ˮ�������� | ||
| C�� | �⡢������ | D�� | �����߲ˡ������ס��ƶ� |
| A�� | �ڲⶨ�к���ʵ������Ҫʹ�õ������У���ƽ����Ͳ���ձ����ζ��ܡ��¶ȼ� | |
| B�� | Ϊ��ȷ�ⶨ��Ӧ�����Һ���¶ȣ�ʵ�����¶ȼ�ˮ����Ӧ��С�ձ��ײ��Ӵ� | |
| C�� | ��50 mL 0.55 mol•L-1��NaOH��Һ��60 mL 0.50 mol•L-1�����ᷴӦ����õ��к�����ֵƫ�� | |
| D�� | ʹ�û��β����������Ϊ�˼Ӵ�Ӧ���ʣ���Сʵ����� |
| A�� | ��40% | B�� | ��40% | C�� | =40% | D�� | ��ȷ�� |