��Ŀ����
9��ʹ������к͵ζ����ⶨ���۰״���������ʵ�鲽�裺��1����ȡ 10.00mLʳ�ð״ף����ձ�����ˮϡ�ͺ�ת�Ƶ�100mL����ƿ�У����
�ý�ͷ�ιܶ��ݣ�ҡ�ȼ��ô���״���Һ��
��2������ʽ�ζ���ȡ����״���Һ20.00mL����ƿ�У������еμ�2�η�̪��ָʾ����
��3����ȡʢװ0.100 0mol/L NaOH��Һ�ļ�ʽ�ζ��ܵij�ʼ������
��4���ζ����ζ��յ����������Һ����ɫ��Ϊdz��ɫ�����ڰ�����ڲ���ɫ
����ʱ��ȡ����ʱ���ӣ�����Լ��������ƫ����ޡ�����ƫ��ƫС����Ӱ�죮
���� ��1������100mL��Һ��Ҫѡ�ù��Ϊ100mL������ƿ�������ʱ��Ҫ�ý�ͷ�ιܣ�
��2������������������Һǡ�÷�Ӧʱ���ɴ����ƣ���Һ�ʼ��ԣ����÷�̪��ָʾ����
��4���ζ�����ǰ��ҺΪ��ɫ���ζ�����ʱ��Һ��Ϊdz��ɫ���ݴ��жϵζ��յ㣻���Ӷ����������ı�Һ���ƫ�ⶨ���ƫ��
��� �⣨1����ȡ10.00mLʳ�ð״ף����ձ�����ˮϡ�ͺ�ת�Ƶ�100mL����ƿ�У�Ȼ���ý�ͷ�ιܶ��ݣ�ҡ�ȼ��ô���״���Һ��
�ʴ�Ϊ������ƿ����ͷ�ιܣ�
��2�����ڴ�������Һ��ʾ���ԣ����÷�̪��ָʾ����������ʽ�ζ���ȡ����״���Һ20.00mL����ƿ�У������еμ�2�η�̪��
�ʴ�Ϊ����̪��
��4����̪��������ʾ��ɫ����������Һ����ʾdz��ɫ����ζ��յ�ʱ��Һ����ɫ��Ϊdz��ɫ�����Եζ��յ������Ϊ����Һ����ɫ��Ϊdz��ɫ�����ڰ�����ڲ���ɫ��
����ʱ��ȡ����ʱ���ӣ����¶����ı�Һ���ƫ�ⶨ���ƫ��
�ʴ�Ϊ����Һ����ɫ��Ϊdz��ɫ�����ڰ�����ڲ���ɫ��ƫ��
���� ���⿼�����к͵ζ�����Һ���ƣ���Ŀ�ѶȲ�����ȷ�к͵ζ�����������������Һ����Ϊ���ؼ���ע������ѡ��ָʾ�����жϵζ��յ�ķ���������������ѧ���Ļ�ѧʵ��������

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | 42g������ͱ�ϩ�Ļ����������ԭ����Ŀһ��Ϊ6NA | |
| B�� | 1L 0.1mol/L��NH4NO3��Һ��ԭ��������0.2NA | |
| C�� | �ڳ��³�ѹ�£�22.4LHF����������ΪNA | |
| D�� | 5.6gFe��������ˮ������ȫ��Ӧ��ת�Ƶ��ӵ���ĿΪ0.2NA |
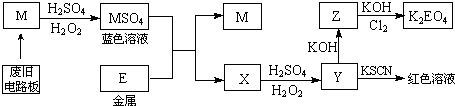
�ݴ˻ش����⣺
��1��M�Ļ�ѧʽ��Cu��
��2��M��MSO4�����У���������H2O2����ԭ����Cu��
��3��д��Z��K2EO4�Ļ�ѧ��Ӧ����ʽ��2Fe��OH��3+10KOH+3Cl2=2K2FeO4+8H2O+6KCl��
��4����Y��Һ�еμ�KI������Һ����Ϊ��ɫ����ԭ�������ӷ���ʽ����Ϊ��2Fe3++2I-�T2Fe2++I2��
��5����������������ͬ��ӡˢ��·��Ľ��� M��10%H2O2��3.0mol/LH2SO4�Ļ����Һ��������ò�ͬ�¶��½���M��ƽ���ܽ����ʣ����±�����
| �¶ȣ��棩 | 20 | 30 | 40 | 50 | 60 | 70 | 80 |
| V��M����x10-3mol��L-1��min-1�� | 7.34 | 8.01 | 9.25 | 7.98 | 7.24 | 6.73 | 5.76 |
| A�� | һ���¶��£�����ˮϡ�ͣ�NH3?H2O �ĵ��������H2O �ĵ����Ҳ���� | |
| B�� | ˮ�м���Ӽ���谭ˮ�ĵ��룬���� c��H+��?c��OH-����10-14 | |
| C�� | ˮ�м��ᣬ�谭ˮ�ĵ��룬���� c��H+��=c��OH-����10-7 mol•L-1 | |
| D�� | ������Һ��ˮϡ�ͣ���Һ����������Ũ�ȶ��ή�� |
| A�� | 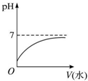 | |
| B�� | ��Һ��$\frac{c��C{H}_{3}CO{O}^{-}��}{c��C{H}_{3}COOH��•c��O{H}^{-}��}$���� | |
| C�� | ��Һ�е������ӵ���Ŀ���� | |
| D�� | �ټ���10 mL pH=11��NaOH��Һ��ǡ����ȫ�к� |
| A�� | H2+Cl2�T2HCl | B�� | CuO+2H+�TCu2++H2O | ||
| C�� | H2O+CaO�TCa��OH��2 | D�� | NaOH+HCl�TH2O+NaCl |
