��Ŀ����
10�� �Ըߴ�H2Ϊȼ�ϵ����ӽ���Ĥȼ�ϵ�ؾ�������Ч�ʸߡ�����Ⱦ���ŵ㣬��ȼ����������CO���������̵���������Լ״�Ϊԭ����ȡ�ߴ�H2����Ҫ�о�����
�Ըߴ�H2Ϊȼ�ϵ����ӽ���Ĥȼ�ϵ�ؾ�������Ч�ʸߡ�����Ⱦ���ŵ㣬��ȼ����������CO���������̵���������Լ״�Ϊԭ����ȡ�ߴ�H2����Ҫ�о�������1���״��ڴ����������ѽ�ɵõ�H2����Ԫ�������ʴ�100%����Ӧ�Ļ�ѧ����ʽΪCH3OH $\frac{\underline{\;����\;}}{\;}$CO+2H2���÷�����ȱ���Dz���H2��CO������ߣ�
��2���״�ˮ��������������Ҫ��������������Ӧ��
����Ӧ��CH3OH��g��+H2O��g���TCO2��g��+3H2��g����H=+49kJ•mol-1
����Ӧ��H2��g��+CO2��g���TCO��g��+H2O��g����H=+41kJ•mol-1
�ټ��ܼӿ췴Ӧ�����������CH3OHƽ��ת���ʵ�һ�ִ�ʩ�������¶ȣ�
�ڷ����ʵ�����ˮ���ȣ�nH2O��nCH3OH���Լ״�ˮ������������ĺô���״��������ʣ�����������CO�����ɣ�
��ij�¶��£���nH2O��nCH3OH=1��1��ԭ������������ܱ������У���ʼѹǿΪp1����Ӧ�ﵽƽ��ʱ��ѹǿΪp2����ƽ��ʱ�״���ת����Ϊ��$\frac{{P}_{2}}{{P}_{1}}$-1����100%�������Ը���Ӧ��
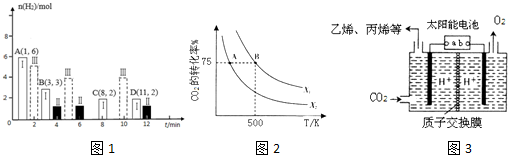
�ܹ�ҵ�����У���λʱ���ڣ���λ����Ĵ��������������������������[��λΪm3/��m3����•h������Ϊh-1]��һ�������£��״���ת�������¶ȡ����ٵĹ�ϵ��ͼ������Խ�״���ת�������¶�Ӱ��Խ������������ͬ���Ƚ�230��ʱ1200h-1��300h-1���ֿ�������ͬʱ����H2�IJ�����ǰ��ԼΪ���ߵ�3.2���������Ը���Ӧ������2λ��Ч���֣�
��3���״�ˮ���������������Ĵ������ܣ���ѧ�������ԭ�����в���һ���������������Ͽ�ʵ�ּ״�ˮ���������������⣮
��֪��CH3OH��g��+$\frac{1}{2}$O2��g���TCO2��g��+2H2��g����H=-193kJ•mol-1
��5CH3OH��g��+4H2O��g��+$\frac{1}{2}$O2��g���T5CO2��g��+14H2��g���ġ�H=+3 kJ•mol-1��
���� ��1���״��ڴ����������ѽ�ɵõ�H2����Ԫ�������ʴ�100%����Ӧ����CO���������õ����������CO�������ȥ��
��2��������ӦΪ���������������ȷ�Ӧ�������¶�������ƽ�������ƶ���������Ӧ���ʣ�
������ˮ���ȣ��״�������������
�ۺ��º����£������ѹǿ֮�ȵ��������ʵ���֮�ȣ�����ƽ��ʱ����������ʵ����������ò���������μӷ�Ӧ�״������ʵ�������������״���ת���ʣ�
����ͼ��֪������Խ�״���ת�������¶�Ӱ��Խ��
��ͼ��֪��230��ʱ1200h-1��300h-1���ֿ����¼״���ת���ʷֱ�Ϊ75%��95%�������IJ���֮�ȵ��ڲμӷ�Ӧ�״������֮�ȣ�
��3����֪���٣�CH3OH��g��+H2O��g��?CO2��g��+3H2��g������H=+49 kJ•mol-1
�ڣ�CH3OH��g��+$\frac{1}{2}$O2��g��?CO2��g��+2H2��g����H=-193kJ•mol-1
���ݸ�˹���ɣ��١�4+�ڿɵã�5CH3OH��g��+4H2O��g��+$\frac{1}{2}$O2��g��?5CO2��g��+14H2��g����
��� �⣺��1���״��ڴ����������ѽ�ɵõ�H2����Ԫ�������ʴ�100%����Ӧ����CO����������Ӧ����ʽΪ��CH3OH $\frac{\underline{\;����\;}}{\;}$CO+2H2��ȱ���ǵõ����������CO�������ȥ������H2��CO������ߣ�
�ʴ�Ϊ��CH3OH $\frac{\underline{\;����\;}}{\;}$CO+2H2������H2��CO������ߣ�
��2��������ӦΪ���������������ȷ�Ӧ�������¶�����Ӧ���ʣ�������ƽ�������ƶ����״���ת�������ʴ�Ϊ�������¶ȣ�
������ˮ���ȣ�nH2O��nCH3OH���������ڷ�Ӧ������У���״��������ʣ�����������CO�����ɣ��ʴ�Ϊ����״��������ʣ�����������CO�����ɣ�
������ʼ n��H2O��=n��CH3OH��=1mol�����º����£������ѹǿ֮�ȵ��������ʵ���֮�ȣ�ƽ��ʱ����������ʵ���2mol��$\frac{{P}_{2}}{{P}_{1}}$��
CH3OH��g��+H2O��g��?CO2��g��+3H2��g�����ʵ�������
1 2
�� $\frac{{P}_{2}}{{P}_{1}}$-1��mol 2mol��$\frac{{P}_{2}}{{P}_{1}}$-2mol=2��$\frac{{P}_{2}}{{P}_{1}}$-1��mol
�ʼ״���ת����Ϊ$\frac{��\frac{{P}_{2}}{{P}_{1}}-1��}{1mol}$��100%=��$\frac{{P}_{2}}{{P}_{1}}$-1����100%��
�ʴ�Ϊ����$\frac{{P}_{2}}{{P}_{1}}$-1����100%��
����ͼ��֪������Խ�״���ת�������¶�Ӱ��Խ��
��ͼ��֪��230��ʱ1200h-1��300h-1���ֿ����¼״���ת���ʷֱ�Ϊ75%��95%�������IJ���֮�ȵ��ڲμӷ�Ӧ�״������֮�ȣ�����ͬʱ����H2�IJ�����ǰ��ԼΪ���ߵ�$\frac{1200��75%}{300��95%}$��3.2����
�ʴ�Ϊ����3.2��
��3����֪���٣�CH3OH��g��+H2O��g��?CO2��g��+3H2��g������H=+49 kJ•mol-1
�ڣ�CH3OH��g��+$\frac{1}{2}$O2��g��?CO2��g��+2H2��g����H=-193kJ•mol-1
���ݸ�˹���ɣ��١�4+�ڿɵã�5CH3OH��g��+4H2O��g��+$\frac{1}{2}$O2��g��?5CO2��g��+14H2��g������H=4��49 kJ•mol-1-193kJ•mol-1=+3kJ•mol-1��
�ʴ�Ϊ��+3��
���� ���⿼�黯ѧƽ�������Ӱ�����ء���Ӧ�ȼ���ȣ����ؿ���ѧ����������������ע���˹�����ڷ�Ӧ�ȼ�����Ӧ�ã�

| NaOH��ʼ���� | NaOH�յ���� | |
| ��һ�� | 0.50mL | 18.60mL |
| �ڶ��� | 0.70mL | 19.00mL |
��2���ﵽ�ζ��յ�ı�־����ɫ��Ϊ��ɫ����30s�ڲ���ɫ��
��3�����²�����ɲⶨ���ƫ�ߵ�ԭ�������AD��
A��δ�ñ�Һ��ϴ��ʽ�ζ���
B���ζ��յ����ʱ�����ӵζ��ܵĿ̶ȣ�������������ȷ
C��ʢװδ֪Һ����ƿ������ˮϴ����δ�ô���Һ��ϴ
D���ζ����յ����ʱ���ֵζ��ܼ��촦����һ����Һ��
�״�����Ҫ�Ļ���ԭ�ϣ��ֿɳ�Ϊȼ�ϣ���ҵ�����úϳ�������Ҫ�ɷ�ΪCO��CO2��H2���ڴ����������ºϳɼ״�������������Ӧ���£�
��CO��g��+2H2��g���TCH3OH��g����H=��
��CO2��g��+3H2��g���TCH3OH��g��+H2O��g����H=-58kJ/mol
��CO2��g��+H2��g���TCO��g��+H2O��g����H=+41kJ/mol
�ش��������⣺
��1����֪��Ӧ���е���صĻ�ѧ�������������£�
| ��ѧ�� | H-H | C-O | C $\frac{\underline{\;��\;}}{\;}$O | H-O | C-H |
| E/��kJ•mol-1�� | 436 | 343 | 1076 | 465 | x |
��2����T��ʱ��6molCO2��8molH2����2L�ܱ������з�����Ӧ�ڣ����H2�����ʵ�����ʱ��仯��ͼ1��״̬��ͼ1��ʵ�ߣ���ʾ��ͼ1������A��1��6��������1minʱH2�����ʵ�����6mol��
��T��ʱ��״̬�������£�ƽ�ⳣ��K=0.5��
�������������䣬���ı��¶�ʱ�����H2�����ʵ�����ʱ��仯��ͼ��״̬����ʾ����״̬���Ӧ���¶ȣ����������������=����T�棻
��һ���¶��£��˷�Ӧ�ں��������н��У����жϸ÷�Ӧ�ﵽ��ѧƽ��״̬���ݵ���bc��
a�� 2��C=O���ѵ�ͬʱ��2��H-O���� b�������л������ƽ��Ħ����������
c��v�棨H2��=3v����CH3OH�� d���״���ˮ����������ȱ��ֲ���
������̼�Ļ��������ǻ��������о����ȵ���⣮
��1��CO2�������⻯�ϳɵ�̼ϩ������2L�����ܱ������г���2moI CO2��nmol H2����һ�������·�����Ӧ��2C02��g��+6H2��g��?CH2=CH2��g��+4H20��g������H=-128kJ/mol��CO2��ת�������¶ȡ�Ͷ�ϱ�[X=$\frac{n��{H}_{2}��}{n��C{O}_{2}��}$]�Ĺ�ϵ��ͼ2��ʾ��
��X2�� X1�����������������=����
����500Kʱ����B���Ͷ�ϱ�Ϊ3.5���Ҵӷ�Ӧ��ʼ��B����Ҫ10min����v��H2��=0.225mol/��L��min����
��2����ϡ����Ϊ�������Һ������̫���ܽ�CO2ת��Ϊ��̼ϩ��������ԭ��ͼ3���£�����������ϩ�ĵ缫��ӦʽΪ2CO2+12e-+12H+=CH2=CH2+4H2O��
 2SO2��g��+2O2��g��?2SO3��g���������������Ҫ��Ӧ֮һ���±���ԭ������V��SO2����V��O2����V��N2��=7��11��82Ͷ�ϣ���1.01��105Paʱ����ͬ�¶���SO2��ƽ��ת���ʣ�
2SO2��g��+2O2��g��?2SO3��g���������������Ҫ��Ӧ֮һ���±���ԭ������V��SO2����V��O2����V��N2��=7��11��82Ͷ�ϣ���1.01��105Paʱ����ͬ�¶���SO2��ƽ��ת���ʣ�| �¶�/ | 400 | 500 | 600 |
| SO2ת����/% | 99.2 | 93.5 | 73.7 |
��2��400�棬1.01��105Paʱ������10 mol SO2��ԭ����ͨ��һ�ܱ������н��з�Ӧ��ƽ��ʱSO2�����ʵ�����0.08mol��
��3�����᳧β������Ҫ�ɷ�SO2��O2��N2���е�Ũ��SO2�������кܶ����
���ð�ˮ��������β������SO2�백ˮǡ�÷�Ӧ�õ����Եģ�NH4��2SO3��Һʱ�����йظ���Һ�����й�ϵ��ȷ����ac������ţ���
a�� c��NH4+��+c��NH3•H2O��=2[c��SO32-��+c��HSO3-��+c��H2SO3��]
b�� c��NH4+��+c��H+��=c��SO32-��+c��HSO3-��+c��OH-��
c�� c��NH4+����c��SO32-����c��OH-����c��H+��
���� MnO2��ˮ������Һ��������β��������MnSO4��
a�� �õ�MnSO4�Ļ�ѧ����ʽ��H2O+SO2=H2SO3��MnO2+H2SO3=MnSO4+H2O��
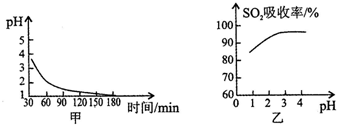
b�������չ�������MnSO4ʱ����Һ��pH�仯������ͼ�ף�SO2����������ҺpH�Ĺ�ϵ��ͼ�ң� ͼ����pH�仯����Ϊ�������в���SO2����������ת��ΪH2SO4������H2SO4��Ӧ�Ļ�ѧ����ʽ��2SO2+O2+2H2O=2H2SO4����ͼ�ҿ�֪pH�Ľ��Ͳ�����SO2�����գ�������ڡ������ڡ������û�ѧƽ���ƶ�ԭ��������ԭ������Һ�д���SO2+H2O?H2SO3?H++HSO3-������Һ��������ǿ��ƽ�������ƶ���ʹSO2�������ϵ���ݳ�����
| A�� | �ղ��ù��������������ó����ֺ���ɫ�� | |
| B�� | ��ú����ȼ����Ȼ��Ϊ�����ṩ���� | |
| C�� | ţ�̾��ÿ����б��ʸ��� | |
| D�� | ���ˮ�е���FeCl3������Һ���ʵ����ȣ��Ʊ����� |
| A�� | c��Fe3+��=0.1 mol•L-1����Һ�У�K+��ClO-��SO42-��SCN- | |
| B�� | $\frac{c��{H}^{+}��}{c��O{H}^{-}��}$=1012����Һ�У�NH4+��Al3+��NO3-��Cl- | |
| C�� | ��ˮ���������c��OH-��=1��10-13mol/L����Һ�У�Ca2+��K+��Cl-��HCO3- | |
| D�� | pH=1����Һ�У�Fe2+��NO3-��SO42-��Na+ |