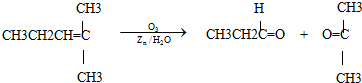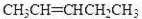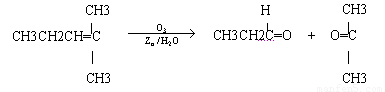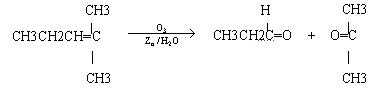��Ŀ����
ϩ��ͨ��������������п��ˮ�����õ�ȩ��ͪ�����磺
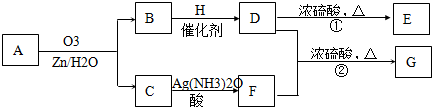
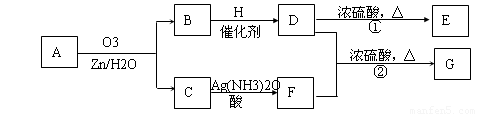
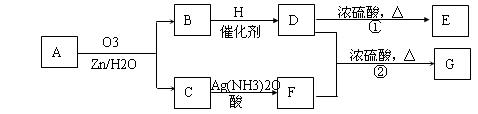
������Ӧ�������ƶ�ϩ���Ľṹ��һ����״��ϩ��Aͨ��������������п��ˮ�����õ�B��C��������B��̼69.8%������11.6%��B��������Ӧ������������D��D��Ũ��������¼��ȣ��ɵõ���ʹ��ˮ��ɫ��ֻ��һ�ֽṹ������E����Ӧͼʾ���£�
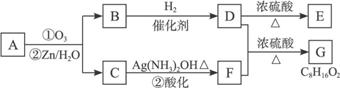
�ش��������⣺
��1��B����Է���������______________��C![]() F�ķ�Ӧ����Ϊ______________��D�к��й����ŵ�����______________��
F�ķ�Ӧ����Ϊ______________��D�к��й����ŵ�����______________��
��2��D+F![]() G�Ļ�ѧ����ʽ�ǣ�_______________________________________________��
G�Ļ�ѧ����ʽ�ǣ�_______________________________________________��
��3��A�Ľṹ��ʽΪ______________________________________________��
��4��������A��ij��ͬ���칹��ͨ��������������п��ˮ����ֻ�õ�һ�ֲ�����ϸ��������칹��Ľṹ��ʽ��____________�֡�
������������һ�������ƶϺͶ����������ϵ���Ŀ��Ҫע������ȷ�ԡ������ԣ�ǡ����������Ŀ���������ʵ����ʣ��жϷ����������Ĺ����ţ��Ӷ��ƶϳ��������ʣ�Ȼ�����Իش����⡣
�𰸣���1��86 ������Ӧ �ǻ�
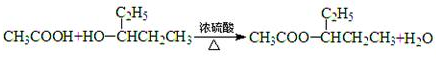
��2��CH3CH2COOH+(C2H5)2CHOH![]() CH3CH2COOCH(C2H5)2+H2O
CH3CH2COOCH(C2H5)2+H2O
��3��(CH3CH2)
��4��3