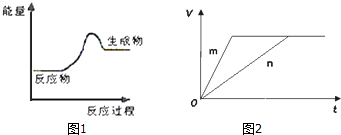
��Ŀ����
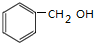
1���ֱ�д�����������������й����ʵĽṹ��ʽ��ѧ����ʽ����1����������Ʒ�Ӧ�����������������Ʒ�Ӧ�ķ����廯����C7H8O��
��2������ʽΪC3H7Br��±��������ȥ��Ӧ��õ����л�����CH3CH=CH2��
��3����C��H��O����Ԫ�ص��л����ȫȼ��ʱ���ĵ����������ɵ�CO2��H2O֮�����ʵ���֮��Ϊ1��1��1��������л��������һ����HCHO
��4�������뱥����ˮ�ķ�Ӧ����ʽΪ
 ���÷�Ӧ���ڱ��ӵĶ��Լ���Ͷ����ⶨ��
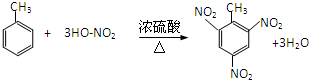
���÷�Ӧ���ڱ��ӵĶ��Լ���Ͷ����ⶨ����5��TNT��2��4��6-�������ױ�����һ������ըҩ���㷺���ڹ�����������·�ȣ�д���Ʊ�TNT�ķ���ʽ
 ��
��
���� ��1����������Ʒ�Ӧ�����������������Ʒ�Ӧ�ķ����廯����C7H8O��˵���������к��д��ǻ��������ǻ���
��2����NaOH�Ĵ���Һ�С����������¸�����鷢����ȥ��Ӧ���ɱ�ϩ��
��3����C��H��O����Ԫ�ص��л��ȼ��ʱ���ĵ����������ɵ�CO2��H2O֮�����ʵ���֮��Ϊ1��1��1�����л���ΪCnH2nOx������CnH2nOx+$\frac{3n-x}{2}$O2=n CO2+nH2O������
��4�����ǻ���λ����λ���巢��ȡ����Ӧ��
��5���ױ���Ũ������Ũ���ᡢ���������·���ȡ����Ӧ����2��4��6-�������ױ���
��� �⣺��1����������Ʒ�Ӧ�����������������Ʒ�Ӧ�ķ����廯����C7H8O��Ϊ���״����ṹ��ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��2������ʽΪC3H7Br��±����CH3CH2CH2Br��CH3CHBrCH3����ȥ��Ӧ��õ����л����ΪCH3CH=CH2���ʴ�Ϊ��CH3CH=CH2��
��3����C��H��O����Ԫ�ص��л��ȼ��ʱ���ĵ����������ɵ�CO2��H2O֮�����ʵ���֮��Ϊ1��1��1�����л���ΪCnH2nOx��ȼ��ͨʽΪCnH2nOx+$\frac{3n-x}{2}$O2=n CO2+nH2O����$\frac{3n-x}{2}$=n�������ɵ�n=x�������ʽΪCH2O��������л��������һ���ǣ�HCHO���ʴ�Ϊ��HCHO��
��4�����ǻ���λ����λ���巢��ȡ����Ӧ�������뱥����ˮ�ķ�Ӧ����ʽΪ�� ���÷�Ӧ�dz����������ڱ��ӵĶ��Լ���Ͷ����ⶨ��
���÷�Ӧ�dz����������ڱ��ӵĶ��Լ���Ͷ����ⶨ��
�ʴ�Ϊ�� �����Լ��飻�����ⶨ��
�����Լ��飻�����ⶨ��
��5���ױ���Ũ������Ũ���ᡢ���������·���ȡ����Ӧ����2��4��6-�������ױ����Ʊ�TNT�ķ���ʽΪ�� ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼���л���ṹ�����ʡ��л����ƶϡ��л���ѧ����ʽ��д�ȣ���3��ע������ԭ���غ���ȼ��ͨʽ�����������������չ����ŵ�������ת����

 �¿α�������������ҵ�������γ�����ϵ�д�
�¿α�������������ҵ�������γ�����ϵ�д� ����ν����Ž̲��㽭���̴�ѧ������ϵ�д�
����ν����Ž̲��㽭���̴�ѧ������ϵ�д� �����Ļ������������������ϵ�д�
�����Ļ������������������ϵ�д�| A�� | ���Ӱ뾶��Na+��Mg2+��Al3+��F- | B�� | ���ȶ��ԣ�HCl��H2S��PH3��AsH3 | ||
| C�� | ����ǿ����H2SiO3��H2CO3��H3PO4 | D�� | �۵㣺SiO2��NaCl��I2��CO2 |
| A�� | пƬ�ܽ���1 mol��ͭƬ������1 mol���� | |
| B�� | �����ܽ�����������ʵ�������� | |
| C�� | пƬ�ܽ���1 g��ͭƬ������1 g���� | |
| D�� | пƬ�ܽ���1 mol������������2mol |
| ���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0�� |
| 2 | �� | �� | �� | |||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� |
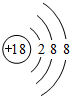 ��
����2��������������ˮ�����У�������ǿ�Ļ�����ķ���ʽ��HClO4��������ǿ�Ļ������������KOH��
��3������������������������Ԫ����Al��д���������������������Ʒ�Ӧ�Ļ�ѧ����ʽAl2O3+2OH-=2AlO2-+H2O��
��4��Ԫ�آ���ߵĻ������������ӣ�����ۡ������ӡ��������
| A�� | ����NaOH��Һ�������� | B�� | ������ȩ̫�� | ||
| C�� | ����CuSO4��Һ�������� | D�� | ������ȩ����̫�� |
| A�� | H2O��H2S�ȶ�������ΪH2O���Ӽ������� | |
| B�� | P�ķǽ�����ǿ��Si��H3PO4��H2SiO3������ǿ | |
| C�� | ���������Ԫ������������Ӧ��ˮ�����У�������ǿ����HNO3 | |
| D�� | I��ԭ�Ӱ뾶����Br��HBr��HI�����ȶ���ǿ |
| A�� | B A D C E | B�� | E C D A B | C�� | A B E D C | D�� | C D E B A |
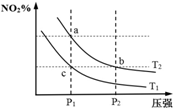 ���ܱ������з�����ӦN2O4��g��?2NO2��g����H=+57kJ•mol-1�����¶�ΪT1��T2ʱ��ƽ����ϵ��NO2�����������ѹǿ�仯������ͼ��ʾ������˵����ȷ���ǣ�������
���ܱ������з�����ӦN2O4��g��?2NO2��g����H=+57kJ•mol-1�����¶�ΪT1��T2ʱ��ƽ����ϵ��NO2�����������ѹǿ�仯������ͼ��ʾ������˵����ȷ���ǣ�������| A�� | ��Ӧ�¶ȣ�T1��T2 | B�� | b��c����ķ�Ӧ���ʣ�v��b����v��c�� | ||
| C�� | a��c�����������ɫ��adz��c�� | D�� | a��b�����ƽ�ⳣ����Kb��Ka |