��Ŀ����
16������Ԫ�����ڱ���һ���֣��������е���ĸ�ֱ����һ�ֻ�ѧԪ�أ��Իش��������⣺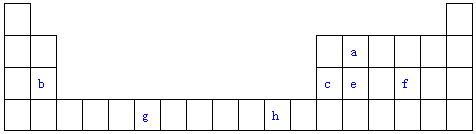
��1��д��Ԫ��g�Ļ�̬ԭ�ӵ����Ų�ʽ[Ar]3d54s1��1s22s22p63s23p63d54s1��
��2��f�ڿ�����ȼ�ղ���ķ��ӹ���Ϊ���ͣ���V���ͣ�������ԭ�ӵ��ӻ���ʽΪsp2�ӻ����÷����Ǽ��Է��ӣ�ѡ����ԡ������Ǽ��ԡ������ӣ�
��3����aԭ�Ӹ�eԭ����1��1������϶��γɵľ��壬�����뾧��e��ͬ����������۵���ߵ���SiC���ѧʽ�����Դӽṹ�Ƕȼ��Խ��Ͷ���ͬΪԭ�Ӿ��壬������C-Si����Si-Si����C-Si�����ι̣�
��4��c���ʾ�����ԭ�ӵĶѻ���ʽ��ͼ����ʾ���侧��������ͼ����ʾ��ԭ��֮���λ�ù�ϵ��ƽ��ͼ��ͼ����ʾ��
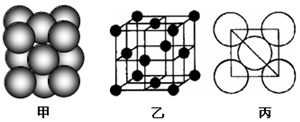
����֪c��ԭ�Ӱ뾶Ϊd���ף�NA���������ӵ�������c�����ԭ������ΪM����ش�
�پ�����cԭ�ӵ���λ��Ϊ12����ʾԭ�ӿռ�ռ���ʵĴ���ʽΪ$\frac{\sqrt{2}��}{6}$��
�ڸþ�����ܶ�Ϊ$\frac{\sqrt{2}M}{8{d}^{3}{N}_{A}}$g/cm3���ú��й���ĸ�Ĵ���ʽ��ʾ����b��h�У���c���ʾ�����ԭ�ӵĶѻ���ʽ��ͬ����Cu����Ԫ�ط��ţ���
���� ��Ԫ�������ڱ���λ�ã���֪aΪC��bΪMg��cΪAl��eΪSi��fΪS��gΪCr��hΪCu��
��1��gΪCrԪ�أ�Ϊ24��Ԫ�أ�ԭ�Ӻ�����24�����ӣ����ݺ�������Ų�������д��
��2������������ȼ�����ɶ��������ݼ۲���Ӷ������ж�����ӹ��ͺ�����ԭ�ӵ��ӻ���ʽ������������������IJ��غϣ�Ϊ���Է��ӣ���֮Ϊ�Ǽ��Է��ӣ�
��3����Cԭ�Ӹ�Siԭ����1��1������϶��γɵľ��壬�����뾧��Si��ͬ����Ϊԭ�Ӿ��壬��Si-C��������Si-Si������
��4���ٸ�����Ϊ�����������ܶѻ����ݴ˼�����λ����������Խ���Ϊ��ԭ�Ӱ뾶��4�����ݴ˼���ԭ�ӿռ�ռ���ʣ�
�ڸ��ݾ�̯�����㾧���к���ԭ����Ŀ�����ݦ�=$\frac{m}{V}$���㣬CuΪ����������
��� �⣺��1��gΪCrԪ�أ�Ϊ24��Ԫ�أ�ԭ�Ӻ�����24�����ӣ����Ժ�������Ų�ʽΪ��[Ar]3d54s1��1s22s22p63s23p63d54s1��
�ʴ�Ϊ��[Ar]3d54s1��1s22s22p63s23p63d54s1��
��2��SԪ�أ��ڿ�����ȼ������SO2��S��1�Թ¶Ե��ӣ�����S=O�����۲���ӶԸ���=�Ҽ�����+�µ��ӶԸ�����SO2�ļ۲���ӶԸ���=2+$\frac{1}{2}$��6-2��2��=3��Sԭ�Ӳ�ȡsp2�ӻ���Ϊ���ͣ���V���ͣ�������������IJ��غϣ�Ϊ���Է��ӣ�
�ʴ�Ϊ�����ͣ���V���ͣ���sp2�ӻ������Է��ӣ�
��3����Cԭ�Ӹ�Siԭ����1��1������϶��γɵ�SiC ���壬�����뾧��Si��ͬ����Ϊԭ�Ӿ��壬��Si-C��������Si-Si��������Si-C���ܴ�C-Si�����ι̣�SiC���۵�ߣ�
�ʴ�Ϊ��SiC������ͬΪԭ�Ӿ��壬������C-Si����Si-Si����C-Si�����ι̣�
��4���ٸ���ͼƬ֪��Al����Ϊ�����������ܶѻ���Alԭ����λ����λ��=3��$\frac{1}{2}$��8=12���� ��ԭ�Ӱ뾶Ϊd���ף������ı߳�Ϊ4dcm��$\frac{\sqrt{2}}{2}$=2$\sqrt{2}$dcm��ÿ���������к���4��ԭ�ӣ�����ԭ�������Ϊ4��$\frac{4}{3}$��d3cm3���������Ϊ��2$\sqrt{2}$dcm��3���ʿռ�������Ϊ$\frac{4��\frac{4}{3}��{d}^{3}}{��2\sqrt{2}d��^{3}}$=$\frac{\sqrt{2}��}{6}$��
�ʴ�Ϊ��$\frac{\sqrt{2}��}{6}$��
����ԭ�Ӹ���=8��$\frac{1}{8}$+6��$\frac{1}{2}$=4�����ԭ������ΪM���ʾ���������=4��$\frac{M}{{N}_{A}}$g��ÿ���������к���4��ԭ�ӣ������ζԽ��߳���Ϊ4d cm������������ⳤΪ��$\frac{\sqrt{2}}{2}$��4dcm=2$\sqrt{2}$d cm���������Ϊ��2$\sqrt{2}$dcm��3=16$\sqrt{2}$d3cm3���ʾ�����ܶȦ�=$\frac{m}{v}$=$\frac{4��\frac{M}{{N}_{A}}}{��2\sqrt{2}d��^{3}}$=$\frac{\sqrt{2}M}{8{d}^{3}{N}_{A}}$���������ܶѻ�������þ��п�����ͣ������������ܶѻ���Cu�ͣ���������c���ʾ�����ԭ�ӵĶѻ���ʽ��ͬ����Cu��
�ʴ�Ϊ��$\frac{\sqrt{2}M}{8{d}^{3}{N}_{A}}$��Cu��
���� ���⿼��Ԫ�����ڱ��Ľṹ����������Ų����ɡ�����ṹ�����ʵȣ���Ŀ�Ѷ��еȣ��������Ԫ�����ڱ��Ľṹ����Ŀ�ۺ��Խ�ǿ������������ѧ�����ۺ�Ӧ��֪ʶ��������

| A�� | NaOH | B�� | NH3 | C�� | AlCl3 | D�� | HCl |
| A�� | ��HnXOmΪǿ�ᣬ��X���⻯������ˮһ�������� | |
| B�� | ��X��OH��nΪǿ���Y��OH��nҲһ��Ϊǿ�� | |
| C�� | ��XԪ���γɵĵ�����X2����YԪ���γɵĵ���һ����Y2 | |
| D�� | ��Y���������Ϊm����X���������һ��Ϊm |
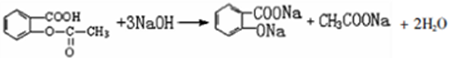
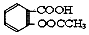 ����������Ӧ����㷺�Ľ��ȡ���ʹ�Ϳ���ҩ������ˮ���������ֽ⣬�ֽ��¶�Ϊ128-135�森ijѧϰС����ʵ������ˮ���ᣨ���ǻ������ᣩ�������[��CH3CO��2O]Ϊ��Ҫԭ�Ϻϳɰ�˾ƥ�֣���Ӧԭ����ͼ��
����������Ӧ����㷺�Ľ��ȡ���ʹ�Ϳ���ҩ������ˮ���������ֽ⣬�ֽ��¶�Ϊ128-135�森ijѧϰС����ʵ������ˮ���ᣨ���ǻ������ᣩ�������[��CH3CO��2O]Ϊ��Ҫԭ�Ϻϳɰ�˾ƥ�֣���Ӧԭ����ͼ��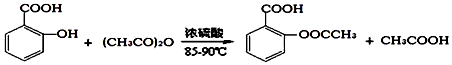
����������ͼ��
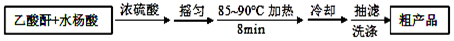
��Ҫ�Լ��Ͳ�Ʒ������������
| �� �� | ��Է������� | �۵��е㣨�棩 | ˮ |
| ˮ���� | 138 | 158���۵㣩 | �� |
| ������ | 102 | 139.4���е㣩 | ��ˮ�� |
| ����ˮ���� | 180 | 135���۵㣩 | �� |
��1���Ʊ���˾ƥ��ʱ��Ҫʹ�ø����������ԭ���Ǵ�������ˮ�⣮
��2���ϳɹ���������ʵļ��ȷ�����ˮԡ���ȣ�
��3���ᴿ�ֲ�Ʒ�������£����Ȼ�����װ����ͼ��ʾ��
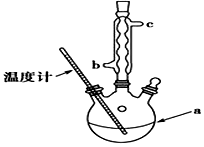
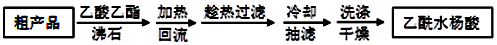
��a����������������ƿ������ˮ������������c���b����c������
�ڳ��ȹ��˵�ԭ���Ƿ�ֹ����ˮ����ᾧ������
�ۼ������ղ�Ʒ���Ƿ���ˮ����Ļ�ѧ������ȡ�����ᾧ���Թ��У�������ˮ�ܽ⣬�μ�FeCl3��Һ����������ɫ��ˮ���ᣮ
������˵����ȷ����ab��
a�������ᴿ�������������������������ܼ�
b�������ᴿ�ֲ�Ʒ�ķ������ؽᾧ
c�����������ᴿ���̿��Եó���˾ƥ�������������е��ܽ�ȵ���ʱ��
��4����ʵ����ԭ��������2.0gˮ���ᡢ5.0mL����������=1.08g/cm3�������ճ�����Ʒ����Ϊ2.2g������������ˮ����IJ���Ϊ84.3%�����ðٷ�����ʾ��������С�����һλ��
| A�� | ������Һ�п��ܴ�������Na+��I-��SO42-��ClO- | |
| B�� | ��������Һ�п��ܴ�������K+��Cl-��HCO3-��AlO2- | |
| C�� | Fe��NO3��3��Һ�м�������HI��Һ�ķ�Ӧ��2Fe3++2I-=2Fe2++I2 | |
| D�� | ���ɫZnS�����ϵμ�CuSO4��Һ����ɫ������ڵ����ӷ���ʽ��ZnS��s��+Cu2+��aq��=CuS��s��+Zn2+��aq�� |
| A�� | ���� | B�� | ��ȩ | C�� | ���� | D�� | ���� |
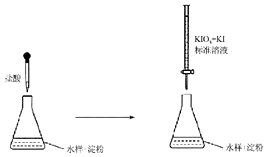 ������������������Ӧ�ù㷺����ѧʹ�ö����彡�����������������ش�
������������������Ӧ�ù㷺����ѧʹ�ö����彡�����������������ش�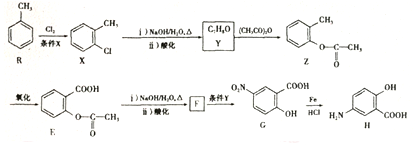
 ��
�� �ױ��������������������ʱ������һ��ȡ��������ȡ����������ڡ���λ���������������Ȼ�ʱ��ȡ���ڼ�λ���ݴ���RΪԭ�Ϻϳɻ�����
�ױ��������������������ʱ������һ��ȡ��������ȡ����������ڡ���λ���������������Ȼ�ʱ��ȡ���ڼ�λ���ݴ���RΪԭ�Ϻϳɻ����� ����������������ƺϳ�·�ߣ�
����������������ƺϳ�·�ߣ� ��
��