��Ŀ����
��10�֣���100 mlŨ�����м���64 gͭƬ������һ��ʱ��������ٷ�ӦΪֹ����ش��������⣺
��1��д����Ӧ�Ļ�ѧ����ʽ�� ��
��2������Ӧ����1.5 mol����ת�ƣ�����ԭ������Ϊ mol����״�������ɵ�SO2�������Ϊ L��
��3������2����Ӧ��Һ����ˣ���ˮϡ����Һ��500 ml��������Zn�ۣ����㹻����ʱ��ų���״����6.72L H2����Ӧ��Zn������Ϊ g��ԭŨ��������ʵ���Ũ��Ϊ mol��L��1��
��1��Cu + 2H2SO4(Ũ)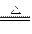 CuSO4 + SO2 ��+ 2 H2O ��2�֣�
CuSO4 + SO2 ��+ 2 H2O ��2�֣�
��2��0.75mol��2�֣��� 16.8L��2�֣�
��3��68.25g��2�֣��� 18 mol��L-1��2�֣�
����

��ϰ��ϵ�д�
 ����ѧҵ���Ե�����ϵ�д�
����ѧҵ���Ե�����ϵ�д�
�����Ŀ