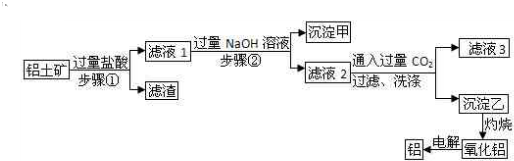
��Ŀ����
�밴Ҫ����գ�
��1����ϩ�ĵ���ʽ ��
��2�����������У�����ͨ����ϩ�ӳɷ�Ӧ�õ����� ��
A��CH3CH3B��CH3CHCl2 C��CH3CH2OHD��CH3CH2Br
��3��ʵ�������õ�ʯ����Ȳ�Ļ�ѧ����ʽ ��
��4����֪ 2CH3CHO+O2
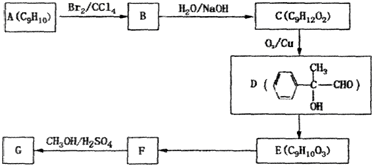
2CH3COOH��������ϩΪ��Ҫԭ�Ϻϳ����ᣬ��ϳ�·����ͼ��ʾ��

��Ӧ�ٵĻ�ѧ����ʽΪ ����Ӧ�ڵĻ�ѧ����ʽΪ ��
��1����ϩ�ĵ���ʽ
��2�����������У�����ͨ����ϩ�ӳɷ�Ӧ�õ�����
A��CH3CH3B��CH3CHCl2 C��CH3CH2OHD��CH3CH2Br
��3��ʵ�������õ�ʯ����Ȳ�Ļ�ѧ����ʽ
��4����֪ 2CH3CHO+O2
| ���� |
| �� |

��Ӧ�ٵĻ�ѧ����ʽΪ
���㣺��ϩ�Ļ�ѧ����,�л���ĺϳ�
ר�⣺
��������1����ϩ�Ǻ���̼̼˫�������ϩ�������ݵ���ʽ������д�ṹ��ʽ��
��2����ϩ�ӳɷ�Ӧ��������ԭ�ӻ�ԭ���ż���˫��̼ԭ���ϣ�˫����Ϊ������
��3��ʵ�������õ�ʯ��ˮ��Ӧ�����������ƺ���Ȳ��
��4����ϩ���Ժ�ˮ�ӳ�����AΪCH3CH2OH��CH3CH2OH���Ա������õ�BΪCH3CHO����ȩ�ױ����������ᣮ
��2����ϩ�ӳɷ�Ӧ��������ԭ�ӻ�ԭ���ż���˫��̼ԭ���ϣ�˫����Ϊ������
��3��ʵ�������õ�ʯ��ˮ��Ӧ�����������ƺ���Ȳ��
��4����ϩ���Ժ�ˮ�ӳ�����AΪCH3CH2OH��CH3CH2OH���Ա������õ�BΪCH3CHO����ȩ�ױ����������ᣮ
���
�⣺��1����ϩ�к�̼��̼֮���Թ���˫����ϣ�����ʽΪ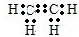 ���ʴ�Ϊ��
���ʴ�Ϊ��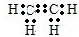 ��
��
��2����ϩ���廯�ⷢ���ӳ����������飬��ˮ�ӳ������Ҵ����������ӳ��������飬�ʴ�Ϊ��B��
��3��ʵ�������õ�ʯ��ˮ��Ӧ�����������ƺ���Ȳ����Ӧ����ʽΪCaC2+2H2O��Ca��OH��2+C2H2�����ʴ�Ϊ��CaC2+2H2O��Ca��OH��2+C2H2����
��4����ϩ���Ժ�ˮ�ӳ�����AΪCH3CH2OH��CH3CH2OH���Ա������õ�BΪCH3CHO����ȩ�ױ����������ᣬ���Է�Ӧ�ٵĻ�ѧ����ʽΪCH2=CH2+H2O
CH3CH2OH����Ӧ�ڵĻ�ѧ����ʽΪ2CH3CH2OH+O2
2CH3CHO+2H2O��
�ʴ�Ϊ��CH2=CH2+H2O
CH3CH2OH��2CH3CH2OH+O2
2CH3CHO+2H2O��
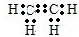 ���ʴ�Ϊ��
���ʴ�Ϊ��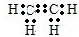 ��
����2����ϩ���廯�ⷢ���ӳ����������飬��ˮ�ӳ������Ҵ����������ӳ��������飬�ʴ�Ϊ��B��
��3��ʵ�������õ�ʯ��ˮ��Ӧ�����������ƺ���Ȳ����Ӧ����ʽΪCaC2+2H2O��Ca��OH��2+C2H2�����ʴ�Ϊ��CaC2+2H2O��Ca��OH��2+C2H2����
��4����ϩ���Ժ�ˮ�ӳ�����AΪCH3CH2OH��CH3CH2OH���Ա������õ�BΪCH3CHO����ȩ�ױ����������ᣬ���Է�Ӧ�ٵĻ�ѧ����ʽΪCH2=CH2+H2O
| ���� |
| Cu |
| �� |
�ʴ�Ϊ��CH2=CH2+H2O
| ���� |
| Cu |
| �� |
������������Ҫ����ѧ����ϩ�Ļ�ѧ���ʣ����Ը��ݽ̲�֪ʶ���ش��ѶȲ�����ʱע�����֪ʶ�����ã�

��ϰ��ϵ�д�
�����Ŀ
2014���ҹ���ѧ���״����㵽ˮ�����ŴصĿռ�ȡ��ͼ��ģ����ͼ�����й���ˮ�� ˵����ȷ���ǣ�������

| A��H2O��D2O��ͬλ�� |
| B����������Ԫ��ֻ�����ˮ |
| C��ˮ����֮�䲻ֻ���ڷ��»��� |
| D��ͼ�����ģ��Ҳ�ɱ�ʾCO2�Ľṹ |
���ڳ����ܽ�ƽ����ܶȻ�����������˵������ȷ���ǣ�������
| A��������Na2SO4��Һ���뵽����ʯ��ˮ�У��а�ɫ����������˵��Ksp[Ca��OH��2]����Ksp[CaSO4] |
| B��Kspֻ�����ܵ���ʵ����ʺ��¶��йأ�������Һ�е�����Ũ���� |
| C����֪25��ʱ��Ksp[Fe��OH��3]=4.0��10-38�����¶��·�ӦFe��OH��3+3H+?Fe3++3H2O��ƽ�ⳣ��K=4.0��104 |
| D����֪25��ʱ��Ksp[Mg��OH��2]=1.8��10-11����MgCl2��Һ�м��백ˮ����û��Һ��pH=11������Һ�е�c��Mg2+��Ϊ1.8��10-3mol��L-1 |
��20mL 0.1mol/L NH4HSO4��Һ�е���0.1mol/L NaOH��Һ��ǡ�÷�Ӧ��ȫ������˵����ȷ���ǣ�������
| A��������Ӧ�����У�c��H+��+c��Na+��+c��NH4+��=c��OH-��+2c��SO42-�� |
| B��������30mL NaOH��Һʱ��pH��7����c��NH4+����c��NH3?H2O����c��OH-����c��H+�� |
| C��������20mL NaOH��Һʱ��2c��SO42-��=c��NH3?H2O��+c��NH4+�� |
| D������Һ������ʱ��c��NH4+����c��SO42-����c��Na+����c��H+��=c��OH-�� |