��Ŀ����
8����Ԫ��������Ԫ�ؼ����γ����Ӽ���Ҳ���γɹ��ۼ����ڻ���������Ԫ�ؿ���Ϊ+1�ۻ�-1�ۣ��ش��������⣺��1��LiH��Li+�İ뾶С��H-�İ뾶������ڡ�����С�ڡ����ڡ�����CaH2��ˮ���ҷ�Ӧ�����ܶ���С�����壬д��CaH2��ϡ���ᷴӦ�Ļ�ѧ����ʽCaH2+2H2O=Ca��OH��2+2H2����
��2������ǽ���Ԫ�������γɹ������⻯��������ڵ���̬�⻯������ȶ�����ǿ������˳��ΪHCl��H2S��PH3��SiH4������̬�⻯�����ʽ����
��3����֪H��B��Si��Cl�ĵ縺�Էֱ�Ϊ2.1��2.0��1.8��3.0�����飨SiH4��������HԪ�صĻ��ϼ�Ϊ-1������������ʱ��������ˮѸ��ˮ������SiO2•nH2O��д���÷�Ӧ�Ļ�ѧ����ʽSiH4+��n+2��H2O=SiO2•nH2O+3H2���������飨B2H6����ǿ��ԭ�ԣ��ڿ���������ȼ���������ǿ��ԭ��������B��
��4���������У��⻯���������鷴Ӧ���������ܻ�ԭ�����⻯�ƣ�NaBH4������д��NaBH4��ˮ��Ӧ�Ļ�ѧ����ʽNaBH4+4H2O�TNa[B��OH��4]+4H2����NaBH4+2H2O�TNaBO2+4H2����
��5����״������ף�N6H6������Ϊ���ܲ��ϵĿ����ԣ���������3�ֻ�ѧ������Nԭ�Ӻ�2�ֻ�ѧ������Hԭ�ӣ���Nԭ�Ӿ��ﵽ8���ӽṹ����Ľṹʽ����Ϊ
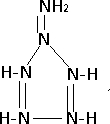 ��
��
���� ��1�����Ӳ�һ���࣬�˵����Խ�뾶ԽС���⻯�ƣ�CaH2����ˮ��Ӧ�����������������غ㶨�ɣ���Ӧǰ��Ԫ������䣬���������������ƣ�д����Ӧ�Ļ�ѧ����ʽ���ɣ�
��2��ͬ����Ԫ�صķǽ�����Խǿ����Ӧ�ĵ��ʵ�������Խǿ���⻯��Ļ�ԭ��Խ����
��3���縺��Խ�õ�������Խǿ������Ԫ����ʾ���ۣ����ݼ�����ˮѸ��ˮ������SiO2•nH2O������������д����ʽ�����ϼ�����Ԫ�ر��ֻ�ԭ�ԣ��������ڿ�����ȼ������B2O3��ˮ��
��4�����⻯�ƣ�NaBH4�����л���ѧ�е�һ�ֳ��û�ԭ��������ˮ��ˮ�����������ƺ��������ݴ���д����ʽ��
��5����״������ף�N6H6������Ϊ���ܲ��ϵĿ����ԣ���������3�ֻ�ѧ������Nԭ�Ӻ�2�ֻ�ѧ������Hԭ�ӣ���Nԭ�Ӿ��ﵽ8���ӽṹ����ԭ����Χ�γ��ĸ����ۼ����ݴ�д���ṹʽ��
��� �⣺��1��LiH��Li+��H-�ĵ��Ӳ���һ���࣬���Ӳ�һ���࣬�˵����Խ�뾶ԽС���ʰ뾶��Li+��H-���⻯�ƣ�CaH2����ˮ��Ӧ�����������������غ㶨�ɣ���Ӧǰ��Ԫ������䣬���������������ƣ���Ӧ�Ļ�ѧ����ʽΪ��CaH2+2H2O=Ca��OH��2+2H2����
�ʴ�Ϊ��С�ڣ�CaH2+2H2O=Ca��OH��2+2H2����
��2��ͬ����Ԫ�صķǽ�����Խǿ����Ӧ�ĵ��ʵ�������Խǿ���⻯��Ļ�ԭ��Խ�����������ڵ���̬�⻯������ȶ�����ǿ������˳��Ϊ��HCl��H2S��PH3��SiH4���ʴ�Ϊ��HCl��H2S��PH3��SiH4��
��3��H�ĵ縺��Խ�õ�������Խǿ��HԪ����ʾ���ۣ�����H��ʾ-1�ۣ�������ˮѸ��ˮ������SiO2•nH2O������ʽΪ��SiH4+��n+2��H2O=SiO2•nH2O+3H2�����������ڿ�����ȼ������B2O3��ˮ����Ӧ����ʽΪ��B2H6+3O2$\frac{\underline{\;��ȼ\;}}{\;}$B2O3+3H2O��BԪ�ػ��ϼ�����-3���ߵ�+3��BԪ�ر��ֻ�ԭ�ԣ��������ǿ��ԭ��������B��
�ʴ�Ϊ��-1��SiH4+��n+2��H2O=SiO2•nH2O+3H2����B��
��4�����⻯�ƣ�NaBH4�����л���ѧ�е�һ�ֳ��û�ԭ��������ˮ��ˮ�����������ƺ���������Ӧ����ʽΪ��NaBH4+4H2O�TNa[B��OH��4]+4H2����NaBH4+2H2O�TNaBO2+4H2�����ʴ�Ϊ��NaBH4+4H2O�TNa[B��OH��4]+4H2����NaBH4+2H2O�TNaBO2+4H2����
��5����������3�ֻ�ѧ������Nԭ�Ӻ�2�ֻ�ѧ������Hԭ�ӣ���Nԭ�Ӿ��ﵽ8���ӽṹ����ԭ����Χ�γ��ĸ����ۼ����ݴ�д���ṹʽ����Ϊ�� ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼��������ˮ�⡢���뾶�Ƚϡ����ʽṹ��������ѧ����ʽ�����ӷ���ʽ��д�����ڱ��еݱ���ɵ�֪ʶ�㣬���ջ����ǽ���ؼ�����Ŀ�ѶȽϴ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�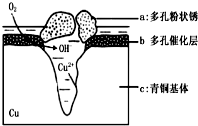 ��ͭ�����������л������Ͷ������ǻ۵Ľᾧ����Ϊһ��ʱ��������������������ͭ������ܵ�������ʴ����ͼΪ��ͭ���ڳ�ʪ�����з����绯ѧ��ʴ��ԭ��ʾ��ͼ�������е� Cl- ��ɢ���ڣ�������缫�����������ɶ��״�� Cu2 �� OH ��3 Cl������˵������ȷ���ǣ�������
��ͭ�����������л������Ͷ������ǻ۵Ľᾧ����Ϊһ��ʱ��������������������ͭ������ܵ�������ʴ����ͼΪ��ͭ���ڳ�ʪ�����з����绯ѧ��ʴ��ԭ��ʾ��ͼ�������е� Cl- ��ɢ���ڣ�������缫�����������ɶ��״�� Cu2 �� OH ��3 Cl������˵������ȷ���ǣ�������| A�� | ��ʴ�����У����� b ������ | |
| B�� | �����е�Cl-��ɢ���ڣ�����������Ӧ���������Ӧ�����������ɶ��״��Cu2��OH��3Cl�������ӷ���ʽΪ2Cu2++3OH-+Cl-=Cu2��OH��3Cl�� | |
| C�� | ������ 2.145gCu 2 �� OH �� 3 Cl�������������ı�״���������Ϊ 0.448L | |
| D�� | �����ĵ缫��ӦʽΪ��������Ӧ�� O2+4e-+2H2O=4OH- |
��CO��g��+$\frac{1}{2}$O2��g���TCO2��g����H=-b kJ/mol
��H2��g��+$\frac{1}{2}$O2��g���TH2O��g����H=-c kJ/mol
��H2��g��+$\frac{1}{2}$O2��g���TH2O��l����H=-d kJ/mol
������������ȷ���ǣ�������
| A�� | �������Ȼ�ѧ����ʽ��֪d��c | |
| B�� | H2��ȼ����Ϊd kJ/mol | |
| C�� | CH3OH��g���TCO��g��+2H2��g����H=��b+2c-a��kJ/mol | |
| D�� | ��CO��H2�����ʵ���֮��Ϊ1��2ʱ������ȫȼ������CO2��H2O��l��ʱ���ų�Q kJ��������������CO�����ʵ���Ϊ$\frac{Q}{b+2c}$mol |
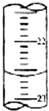 ij�о���ѧϰС����PH=1������ζ�25.00mLδ֪���ʵ���Ũ�ȵ�NaOH��Һ���Ӷ������NaOH��Һ��PHֵ��ѡ�÷�̪��Ϊָʾ����
ij�о���ѧϰС����PH=1������ζ�25.00mLδ֪���ʵ���Ũ�ȵ�NaOH��Һ���Ӷ������NaOH��Һ��PHֵ��ѡ�÷�̪��Ϊָʾ������ʵ���������Ҫʹ�õ�����������̨���ζ��ܼС���ʽ�ζ��ܡ���ͷ�ιܡ���ʽ�ζ��ܡ���ƿ��
����ͼ����ij�εζ�ʱ�ĵζ����е�Һ�棬�����Ϊ22.60mL��
������жϵζ������յ㵱�μ����һ�����ᣬ��Һ����ɫ�ɻ�ɫ��Ϊ��ɫ�����ڰ�����ڲ���ɫ��
�۸����������ݣ�
| �ζ����� | ����Һ�����mL�� | �����������mL�� | |
| �ζ�ǰ������mL�� | �ζ��������mL�� | ||
| ��һ�� | 25.00 | 0.50 | 20.40 |
| �ڶ��� | 25.00 | 4.00 | 24.10 |
��������ʵ���У����в���������������ȷ������ɲⶨ���ƫ�ߵ��У�BD��
A���ζ��յ����ʱ���Ӷ���
B����ʽ�ζ���ʹ��ǰ��ˮϴ��δ��������Һ��ϴ
C����ƿˮϴ��δ����
D����ʽ�ζ��ܼ��첿�������ݣ��ζ�����ʧ��
| A�� | ��Ȼ�� | B�� | ���ȼ� | C�� | ���� | D�� | ��ˮ |
| A�� | 2 LŨ��Ϊ0.5 mol/L ������Һ�к���NA�������� | |
| B�� | 1molFeCl3���ˮ��Ӧ����NA��Fe��OH��3���� | |
| C�� | ��14g CO�к��е�ԭ����ΪNA | |
| D�� | ��H2O2+Cl2�T2HCl+O2��Ӧ�У�ÿ����32 g������ת�Ƶ�����4 NA |
| A�� |  H2S���ռ� | B�� |  NO���ռ� | C�� |  NH3���ռ� | D�� |  Cl2���ռ� |