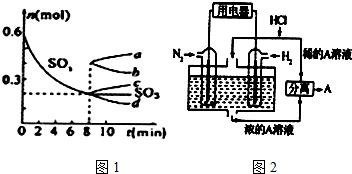
��Ŀ����
19�����ʵ����Ǹ��л�ѧ�г��õ�������������������й������ݵļ��㣮��1��1.2g H2���и�1.2NAHԭ�ӣ�
��2����״���£�������ͬ��ԭ������CO��CO2�����֮��Ϊ2��1��
��3��200mL ijAl2��SO4��3��Һ�У�n��Al3+��=0.20mol��������c��SO${\;}_{4}^{2-}$��=1.5mol/L��
��4����״����6.72L CO��һ������ Fe2O3ǡ����ȫ��Ӧ������Fe��CO2����ʣ����������Ϊ11.2g��
���� ��1�����ݹ�ʽN=nNA������ʵ�ԭ�ӹ��������㣻
��2���ȸ���һ����̼�Ͷ�����̼�ķ�����ɼ�������ߵ����ʵ���֮�ȣ���ͬ���������ʵ���֮�������֮�ȳ����ȣ�
��3���ٸ����������Ļ�ѧʽ��ɼ������������ӵ����ʵ�����������c=$\frac{n}{V}$�������������ӵ�Ũ�ȣ�
��4�����ݷ���ʽ��3CO+Fe2O3$\frac{\underline{\;����\;}}{\;}$2Fe+3CO2���м��㼴�ɣ�
��� �⣺��1��1.2g H2�����ʵ���Ϊ��$\frac{1.2g}{2g/mol}$=0.6mol�����е���ԭ�ӵ���ĿΪ��N��O��=0.6��2��NA=1.2NA��
�ʴ�Ϊ��1.2NA��
��2����״���£�������ͬ��ԭ������CO��CO2�����ʵ���֮��Ϊ��2��1�����֮��Ϊ��2��1��
�ʴ�Ϊ��2��1��
��3������Al2��SO4��3��֪����Һ�к��е���������ӵ����ʵ���Ϊ��n��SO42-��=$\frac{3}{2}$��0.2mol=0.3mol����Һ����������ӵ�Ũ��Ϊ��c��SO42-��=$\frac{0.3mol}{0.2L}$=1.5mol/L��
�ʴ�Ϊ��1.5mol/L��
��4����״����6.72L CO���ʵ�����$\frac{6.72L}{22.4L/mol}$=0.3mol�����ݷ���ʽ��3CO+Fe2O3$\frac{\underline{\;����\;}}{\;}$2Fe+3CO2���õ������������ʵ�����0.2mol��������0.2mol��56g/mol=11.2g���ʴ�Ϊ��11.2��
���� ���⿼�����ʵ����ļ��㣬�������������ʵ����Ĺ�ϵ�����ʵĹ��ɼ������Ļ�ѧ��ӦΪ���Ĺؼ���ע�ػ���֪ʶ�Ŀ��飬��Ŀ�ѶȲ���

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
����˵����ȷ���ǣ�������
| A�� | 1molK����������3molH2�����ӳɷ�Ӧ | |
| B�� | �����������ᷴӦ | |
| C�� | �ʵ������£�1mol����������2mol Br2������Ӧ | |
| D�� | ҡͷ��������� |
| A�� | ��ѧ��Ӧ�У���Ӧ���������������������� | |
| B�� | ��ѧ��Ӧ�У���Ӧ���������������������� | |
| C�� | ��Һ�У�ϡ��ǰ����������ϡ�ͺ��������� | |
| D�� | �ڻ������У������ϼ������븺���ϼ����� |
| A�� | �������Ƶĵ���ʽ��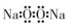 | B�� | ��������ӵĽṹʽ��H-Cl-O | ||
| C�� | C60��ʯī��Ϊͬλ�� | D�� | ����10�����ӵ���ԭ�ӣ�${\;}_{8}^{18}$O |
��1��ú�����������в������к�����H2S��Na2CO3��Һ���գ�����������ʽ�Σ��÷�Ӧ�Ļ�ѧ����ʽΪNa2CO3+H2S�TNaHCO3+NaHS��
��2������ˮú���ϳɶ����ѵ�������Ӧ���£�
��2H2��g��+CO��g��?CH3OH��g������H=-90.8kJ•mol-1
��2CH3OH��g��?CH3OCH3��g��+H2O��g������H=-23.5kJ•mol-1
��CO��g��+H2O��g��?CO2��g��+H2��g������H=-41.3kJ•mol-1
�ܷ�Ӧ��3H2��g��+3CO��g��?��g��+CO2��g���ġ�H=-246.4kJ•mol-1��һ�������µ��ܱ������У����ܷ�Ӧ�ﵽƽ�⣬Ҫ���CO��ת���ʣ����Բ�ȡ�Ĵ�ʩ��a��c��e������ĸ���ţ���
a��ѹ����� b��������� c������CO2��Ũ�� d������CO��Ũ�� e�������������
��3����֪��Ӧ��2CH3OH��g��?CH3OCH3��g��+H2O��g��ij�¶��µ�ƽ�ⳣ��Ϊ400�����¶��£����ܱ������м���CH3OH����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
| ���� | CH3OH | CH3OCH3 | H2O |
| Ũ�ȣ�mol•L-1�� | 0.44 | 0.6 | 0.6 |
���¶����ߣ��÷�Ӧ��ƽ�ⳣ��K��С�����������С�����䡱��
| A�� | ��0.1 mol NaHSO4������������������������Ϊ0.2 NA | |
| B�� | l L 0.1mol•L-1��H2SO3��Һ�к���H+����ĿΪ0.2 NA | |
| C�� | 7.8 g Na2O2�к��е���������ĿΪ0.1 NA | |
| D�� | ���³�ѹ�£�14 g��ϩ�ͱ�ϩ�Ļ����������ԭ����Ϊ 2 NA |