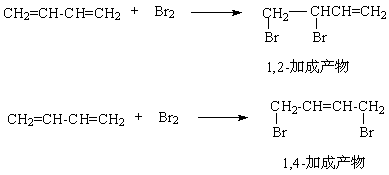��Ŀ����
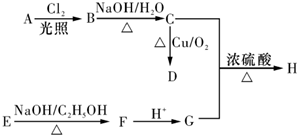
�л���A��F��Ϊ��״�ṹ������֮���ת����ϵ����ͼ��ʾ��
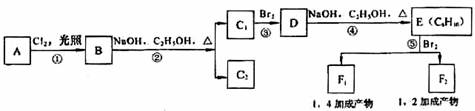
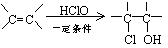
����BΪ���ͻ�����A��һ��ȡ�������Ӧ��ֻ�õ�C1��C2���ֻ�Ϊͬ���칹����л���˴Ź�����ͼ����E������ֻ���������͵��⡣
��1���л���B�Ľṹ��ʽΪ ������ϵͳ������C2������Ϊ ��
��2����Ӧ�ں͢۵ķ�Ӧ����Ϊ �� ��Ӧ��д����D����E�Ļ�ѧ����ʽ ��
��3��������E����Ҫ�Ĺ�ҵԭ�ϣ�д��E�����Ӿ۷�Ӧ�Ļ�ѧ����ʽ
��
��1��
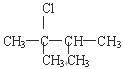 2��3���D�����D1�D��ϩ
2��3���D�����D1�D��ϩ
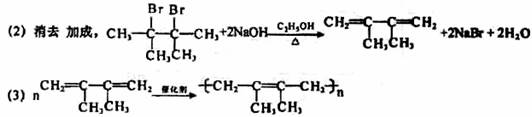

��ϰ��ϵ�д�
�����Ŀ




 RCH2NH2 + HCl����NH2���ױ�����
RCH2NH2 + HCl����NH2���ױ�����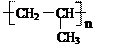 ��
��