��Ŀ����
������������Ҫ�Ĺ�ҵԭ�ϣ�̽�����Ʊ����������ʾ��зdz���Ҫ�����壮
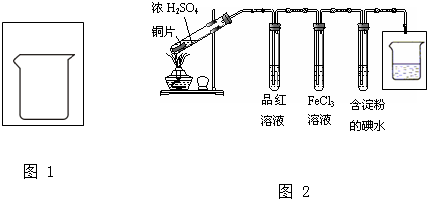
��1��ʵ��������ͼ1��װ�òⶨSO2ת��ΪSO3��ת���ʣ�����֪SO3�۵�Ϊ16.8�棬�����������װ��ʱ�ֱ���ȫ���գ��Һ��Կ�����CO2��Ӱ�죮��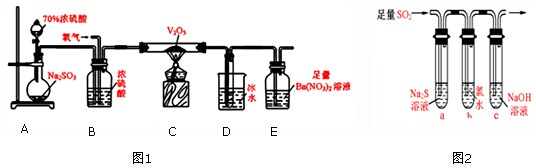
�ټ���ʹ�÷�Һ©����Բ����ƿ�еμ�Ũ����IJ����� ��
�ڵ�ֹͣͨ��SO2��Ϩ��ƾ��ƺ���Ҫ����ͨһ��ʱ�����������Ŀ���� ��
��ʵ���������װ��D���ӵ�����Ϊm g��װ��E�в�����ɫ����������Ϊn g����������¶��������ת������ ���ú���ĸ�Ĵ���ʽ��ʾ�����û���
��2��ijѧϰС�������ͼ2װ����֤��������Ļ�ѧ���ʣ�
����˵������������������Ե�ʵ������Ϊ ��
��Ϊ��֤��������Ļ�ԭ�ԣ���ַ�Ӧ��ȡ�Թ�b�е���Һ�ֳ����ݣ��ֱ��������ʵ�飺
���� I�����һ����Һ�м���AgNO3��Һ���а�ɫ��������
��������ڶ�����Һ����Ʒ����Һ����ɫ��ȥ
���������������Һ����BaCl2��Һ��������ɫ����
���������������Ƿ��� ���I�����������Թ�b�з�����Ӧ�����ӷ�Ӧ����ʽΪ ��
�۵�ͨ������������Թ�c����Һ������ʱ����Һ��c��Na+��= ���ú�����Ũ�ȵĴ���ʽ��ʾ����
��1��ʵ��������ͼ1��װ�òⶨSO2ת��ΪSO3��ת���ʣ�����֪SO3�۵�Ϊ16.8�棬�����������װ��ʱ�ֱ���ȫ���գ��Һ��Կ�����CO2��Ӱ�죮��

�ټ���ʹ�÷�Һ©����Բ����ƿ�еμ�Ũ����IJ�����
�ڵ�ֹͣͨ��SO2��Ϩ��ƾ��ƺ���Ҫ����ͨһ��ʱ�����������Ŀ����
��ʵ���������װ��D���ӵ�����Ϊm g��װ��E�в�����ɫ����������Ϊn g����������¶��������ת������
��2��ijѧϰС�������ͼ2װ����֤��������Ļ�ѧ���ʣ�
����˵������������������Ե�ʵ������Ϊ
��Ϊ��֤��������Ļ�ԭ�ԣ���ַ�Ӧ��ȡ�Թ�b�е���Һ�ֳ����ݣ��ֱ��������ʵ�飺
���� I�����һ����Һ�м���AgNO3��Һ���а�ɫ��������
��������ڶ�����Һ����Ʒ����Һ����ɫ��ȥ
���������������Һ����BaCl2��Һ��������ɫ����
���������������Ƿ���
�۵�ͨ������������Թ�c����Һ������ʱ����Һ��c��Na+��=
���㣺̽������������ˮ��Ʒ����Һ�ķ�Ӧ
ר�⣺ʵ�������
��������1���ٸ��ݷ�Һ©����ʹ�÷������ش�
��Ϊȷ��ʵ���ȷ�ȣ�Ҫ��֤���������������������IJⶨȷ��
�۸��������غ������ܵĶ�����������ʵ���������װ��D������������������Լ������Ӧ���ĵĶ�����������ʵ�����Ȼ���������������ת���ʣ�
��2���ٶ���������������ԣ������ⷴӦ�������ʣ�
���������������ԣ�������������ˮ����������ԭ��Ӧ�������ᣬ����ͨ����������������ж϶���������л�ԭ�ԣ���Ҫ�ų��������ӣ�
�۷�Ӧ�����ҺΪ���ԣ���c��H+��=c��OH-����������Һ�еĵ���غ���н��
��Ϊȷ��ʵ���ȷ�ȣ�Ҫ��֤���������������������IJⶨȷ��
�۸��������غ������ܵĶ�����������ʵ���������װ��D������������������Լ������Ӧ���ĵĶ�����������ʵ�����Ȼ���������������ת���ʣ�
��2���ٶ���������������ԣ������ⷴӦ�������ʣ�
���������������ԣ�������������ˮ����������ԭ��Ӧ�������ᣬ����ͨ����������������ж϶���������л�ԭ�ԣ���Ҫ�ų��������ӣ�
�۷�Ӧ�����ҺΪ���ԣ���c��H+��=c��OH-����������Һ�еĵ���غ���н��
���
�⣺��1���ٷ�Һ©����ʹ�÷�������Һ©���ϿڵĻ�����������Һ©���������������μӣ�
�ʴ�Ϊ����Һ©���ϿڵĻ�����������Һ©���������������μӣ�
��Ϊȷ��ʵ���ȷ�ȣ�Ҫ��֤���������������������IJⶨȷ����ֹͣͨ��SO2��Ϩ��ƾ��ƺ���Ҫ����ͨһ��ʱ���������������װ���еĶ�����������������ֱ������װ�����գ�
�ʴ�Ϊ��ʹ������װ���еĶ��������������������գ�
��װ��D���ӵ�����Ϊmg��˵���������������������mg����ת������������Ķ�����������ʵ���Ϊ��
=
mol��װ��E�в�����ɫ����Ϊ���ᱵ�����������غ㣬�����յĶ�����������ʵ���Ϊ��
=
mol��
���Զ��������ת����Ϊ��
��100%��
�ʴ�Ϊ��
��100%��
��2���ٶ���������������ԣ����Ժ���ͼ۵����Ʒ���������ԭ��Ӧ�����ɵ���ɫ�ij���S��
�ʴ�Ϊ���Թ�a�г��ֵ���ɫ���ǣ�
�ڷ���I�����һ����Һ�м���AgNO3��Һ���а�ɫ�������ɣ���������ˮ�е������Ӳ��������ã��÷�����������
��������ڶ�����Һ����Ʒ����Һ����ɫ��ȥ����������ˮ�к��е�Ư�������ʴ�����������ã��÷�����������
���������������Һ����BaCl2��Һ��������ɫ������֤����Һ���ܳ�����������ӣ��Ƕ������������Ի����º���ˮ��Ӧ���ɵģ���SO2+Cl2+2H2O=4H++SO42-+2Cl-���÷���������
�ʴ�Ϊ����SO2+Cl2+2H2O=4H++SO42-+2Cl-��
�۽���������ͨ�뵽ʢ���������Ƶ��Թ�c�У�������Һ�д��ڵ���غ㣺c��Na+��+c��H+��=2c��SO32-��+c��HSO3-��+c��OH-������Һ��ʾ���ԣ�����c��H+��=c��OH-������c��Na+��=2c��SO32-��+c��HSO3-����
�ʴ�Ϊ��2c��SO32-��+c��HSO3-����
�ʴ�Ϊ����Һ©���ϿڵĻ�����������Һ©���������������μӣ�
��Ϊȷ��ʵ���ȷ�ȣ�Ҫ��֤���������������������IJⶨȷ����ֹͣͨ��SO2��Ϩ��ƾ��ƺ���Ҫ����ͨһ��ʱ���������������װ���еĶ�����������������ֱ������װ�����գ�
�ʴ�Ϊ��ʹ������װ���еĶ��������������������գ�
��װ��D���ӵ�����Ϊmg��˵���������������������mg����ת������������Ķ�����������ʵ���Ϊ��
| mg |
| 80g/mol |
| m |
| 80 |
| ng |
| 233g/mol |
| n |
| 233 |
���Զ��������ת����Ϊ��
| ||||
|
�ʴ�Ϊ��
| ||||
|
��2���ٶ���������������ԣ����Ժ���ͼ۵����Ʒ���������ԭ��Ӧ�����ɵ���ɫ�ij���S��
�ʴ�Ϊ���Թ�a�г��ֵ���ɫ���ǣ�
�ڷ���I�����һ����Һ�м���AgNO3��Һ���а�ɫ�������ɣ���������ˮ�е������Ӳ��������ã��÷�����������
��������ڶ�����Һ����Ʒ����Һ����ɫ��ȥ����������ˮ�к��е�Ư�������ʴ�����������ã��÷�����������
���������������Һ����BaCl2��Һ��������ɫ������֤����Һ���ܳ�����������ӣ��Ƕ������������Ի����º���ˮ��Ӧ���ɵģ���SO2+Cl2+2H2O=4H++SO42-+2Cl-���÷���������
�ʴ�Ϊ����SO2+Cl2+2H2O=4H++SO42-+2Cl-��
�۽���������ͨ�뵽ʢ���������Ƶ��Թ�c�У�������Һ�д��ڵ���غ㣺c��Na+��+c��H+��=2c��SO32-��+c��HSO3-��+c��OH-������Һ��ʾ���ԣ�����c��H+��=c��OH-������c��Na+��=2c��SO32-��+c��HSO3-����
�ʴ�Ϊ��2c��SO32-��+c��HSO3-����
���������⿼���˶����������ʼ����飬��Ŀ�Ѷ��еȣ�ע�����ն�������Ļ�ѧ���ʼ����鷽������ȷ���������ϢΪ�����Ĺؼ�������������ѧ���ķ�����������������ѧʵ��������

��ϰ��ϵ�д�
 Сѧѧϰ�ð���ϵ�д�
Сѧѧϰ�ð���ϵ�д� Сѧͬ�����������ܾ�ϵ�д�
Сѧͬ�����������ܾ�ϵ�д�
�����Ŀ
�������ӷ���ʽ��ȷ���ǣ�������
| A����NaAlO2��Һ��ͨ�����CO2��Al��OH��3��AlO2-+CO2+2H2O=Al��OH��3��+HCO3- |
| B��Fe��OH��3��������Fe��OH��3+3H+=Fe3++3H2O |
| C��AlCl3��Һ�м��������Ũ��ˮ��Al3++4NH3?H2O��AlO2-+4NH4++2H2O |
| D��H2SO4��Ba��OH��2��Һ��Ӧ��Ba2++2OH-+2H++SO42-=BaSO4��+H2O |
����˵����ȷ���ǣ�������
| A����һ���¶��£�AgClˮ��Һ��Ag+��Cl-Ũ�ȵij˻���һ������ |
| B�������������뻯ѧ��Ӧ����Ӧǰ����������ɺ����ʶ����ֲ��� |
| C��Ӧ�ø�˹���ɣ��ɼ�Ӽ���ijЩ����ֱ�Ӳ����ķ�Ӧ�� |
| D�����ڷ��Ӽ�����Ĵ��ڣ���VA���⻯��ķе��С��ϵΪ��NH3��SbH3��AsH3��PH3 |
���и�����Һ���������Լ����������ǣ�������
| A��BaCl2��Һ��ϡH2SO4 |
| B��NH4Cl��Һ��NaOH��Һ |
| C��K2CO3��Һ��ϡH2SO4 |
| D��KI��Һ��NaCl��Һ |
ijԪ��ԭ�ӵ�������Ϊ52��������Ϊ28�����̬ԭ��δ�ɶԵ�����Ϊ��������
| A��1 | B��3 | C��4 | D��6 |
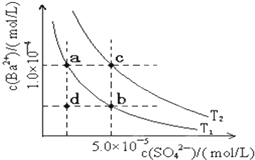 �±��Ǽ���������ʵĵ���ƽ�ⳣ�������ܵ���ʵ��ܶȻ�Ksp��25�棩��
�±��Ǽ���������ʵĵ���ƽ�ⳣ�������ܵ���ʵ��ܶȻ�Ksp��25�棩��