��Ŀ����
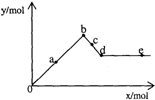 ��֪Ba��AlO2��2������ˮ����ͼ��ʾ������A12��SO4��3��Һ����μ���Ba��OH��2��Һʱ�����ɳ��������ʵ���y�����Ba��OH��2�����ʵ���x�Ĺ�ϵ�������й�������ȷ���ǣ�������
��֪Ba��AlO2��2������ˮ����ͼ��ʾ������A12��SO4��3��Һ����μ���Ba��OH��2��Һʱ�����ɳ��������ʵ���y�����Ba��OH��2�����ʵ���x�Ĺ�ϵ�������й�������ȷ���ǣ�������| A��a-bʱ���������ʵ�����A1��OH��3��BaSO4�� |
| B��c-dʱ��Һ�����ӵ����ʵ�����AlO2-��Ba2+�� |
| C��a-dʱ���������ʵ�����BaSO4����С��A1��OH��3 |
| D��d-eʱ��Һ�����ӵ����ʵ�����Ba2+�����ܵ���OH- |
���㣺���ӷ���ʽ���йؼ���
ר�⣺
������A������a-b�dz�����������Ĺ��̣�����A12��SO4��3+3Ba��OH��2=2Al��OH��3��+3BaSO4����b����������ʵ�������Լ��������ķ�Ӧ���ش�
B��c-d������Ӧ2Al��OH��3+Ba��OH��2=Ba��AlO2��2+4H2O��Ba��AlO2��2�����AlO2-��Ba2+��
C��a-b����A12��SO4��3+3Ba��OH��2=2Al��OH��3��+3BaSO4����b��d��Al��OH��3�������ܽ�ת��ΪBa��AlO2��2���ݴ���������
D��d��ʱAl��OH��3����ǡ����ȫ�ܽ⣬��ʱ��Һ��ֻ����Ba��AlO2��2���������Ba��OH��2�����ʵ�������Ba��AlO2��2�����ʵ���ʱ����Һ��Ba2+��OH-������ȣ�
B��c-d������Ӧ2Al��OH��3+Ba��OH��2=Ba��AlO2��2+4H2O��Ba��AlO2��2�����AlO2-��Ba2+��
C��a-b����A12��SO4��3+3Ba��OH��2=2Al��OH��3��+3BaSO4����b��d��Al��OH��3�������ܽ�ת��ΪBa��AlO2��2���ݴ���������
D��d��ʱAl��OH��3����ǡ����ȫ�ܽ⣬��ʱ��Һ��ֻ����Ba��AlO2��2���������Ba��OH��2�����ʵ�������Ba��AlO2��2�����ʵ���ʱ����Һ��Ba2+��OH-������ȣ�
���
�⣺A����a-b������ӦA12��SO4��3+3Ba��OH��2=2Al��OH��3��+3BaSO4�����ɷ���ʽ��֪�����������ʵ�����Al��OH��3��BaSO4����A����
B��c-d������Ӧ2Al��OH��3+Ba��OH��2=Ba��AlO2��2+4H2O��1molBa��AlO2��2�����2molAlO2-��1molBa2+����AlO2-��Ba2+�࣬��B��ȷ��
C��a-b����A12��SO4��3+3Ba��OH��2=2Al��OH��3��+3BaSO4�����ɷ���ʽ��֪�����������ʵ�����Al��OH��3��BaSO4��b��d��Al��OH��3�������ܽ�ת��ΪBa��AlO2��2����a-dʱ���������ʵ�����BaSO4����A1��OH��3����C����
D��d��ʱAl��OH��3����ǡ����ȫ�ܽ⣬��ʱ��Һ��ֻ����Ba[Al��OH��4]2���������Ba��OH��2�����ʵ�������Ba[Al��OH��4]2�����ʵ���ʱ����Һ��Ba2+��OH-������ȣ���D����
��ѡB��
B��c-d������Ӧ2Al��OH��3+Ba��OH��2=Ba��AlO2��2+4H2O��1molBa��AlO2��2�����2molAlO2-��1molBa2+����AlO2-��Ba2+�࣬��B��ȷ��
C��a-b����A12��SO4��3+3Ba��OH��2=2Al��OH��3��+3BaSO4�����ɷ���ʽ��֪�����������ʵ�����Al��OH��3��BaSO4��b��d��Al��OH��3�������ܽ�ת��ΪBa��AlO2��2����a-dʱ���������ʵ�����BaSO4����A1��OH��3����C����
D��d��ʱAl��OH��3����ǡ����ȫ�ܽ⣬��ʱ��Һ��ֻ����Ba[Al��OH��4]2���������Ba��OH��2�����ʵ�������Ba[Al��OH��4]2�����ʵ���ʱ����Һ��Ba2+��OH-������ȣ���D����
��ѡB��
���������⿼�����ӷ�Ӧ����ʽ��ͼ��ķ�����Ϊ��Ƶ���㣬���շ��������ӷ�Ӧ��ͼ���ж�Ӧ�����ӷ�ӦΪ���Ĺؼ������ط�����Ӧ���������ۺϿ��飬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
һ���¶��£���Ӧ2SO2��g��+O2��g��?2SO3��g�����ﵽƽ��ʱ��n��SO2����n��O2����n��SO3��=2��3��4����С�������Ӧ�ٴδﵽƽ��ʱ��n��O2��=0.8mol��n��SO3��=1.4mol����ʱSO2�����ʵ���Ӧ�ǣ�������
| A��0.4 mol |
| B��0.6 mol |
| C��0.8 mol |
| D��1.2 mol |
���и����ʵķ��ࡢ���ƣ�������������ѧʽ����ȷ���ǣ�������
| A������������ ������̼ CO2 |
| B���� ���� H2S |
| C������������ �������� Ca��OH��2 |
| D���� ���� NaOH |
����ʵ����������Ⱥ�˳�����ȷ���ǣ�������
| A����װ��ҩƷ���ټ��װ�õ������� |
| B������˫������Թܣ��ٽ����ܲ���ˮ�м��װ�õ������� |
| C��������ԭ����ͭ��ʵ�飬��ͨһ��ʱ���������������������ȣ��ټ��� |
| D��ϡ��Ũ����ʱ�������ձ��ﵹ��Ũ���ᣬ��С�ĵ���ˮ�����Ͻ��� |
2014��6��10���ǡ�ȫ����̼�ա��������ǡ�Я�ֽ��ܵ�̼��������ˮ���족���������������ϡ����ܵ�̼��������ǣ�������
| A���ᳫ˫��ʹ��ֽ�� |
| B���ƹ�һ������� |
| C���ᳫʹ��̫������ˮ�� |
| D���ƹ㹫�����г� |
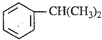 ������һ����Ҫ���л�����ԭ�ϣ������������������գ�
������һ����Ҫ���л�����ԭ�ϣ������������������գ�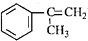 ��������������ABS��֬��һ�ֵ��壬��ҵ���������������õ���д�����������ȡ�õ������һ�ַ���
��������������ABS��֬��һ�ֵ��壬��ҵ���������������õ���д�����������ȡ�õ������һ�ַ���