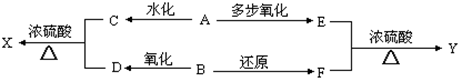
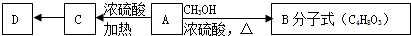��Ŀ����
19������X��ij���;�ˮ�����м��壬�����Կ������Ȼ�������180����������һ����A�����ʵ���֮��1��2��ɣ����ܱ������м���8.75g Xʹ֮��ȫ�ֽ⣬��ȴ��ɵõ�3.2g����������B��0.448L��ɫ����D�����������Ϊ��״������4.27g��Ͼ���E��B����ϡ����μ�KSCN��Һ�����Һ��Ѫ��ɫ��D������ʹƷ����ɫ����ش��������⣺��1��X�Ļ�ѧʽΪAlCl3•2FeSO4��
��2����A�����������������գ�ʹ��ֽ⣬���ɵ����ʵ�����B��D����һ�ֻ������A�ֽ�Ļ�ѧ����ʽΪ2FeSO4$\frac{\underline{\;����\;}}{\;}$SO2��+SO3+Fe2O3��
��3����E��Ͼ�������ˮ�����Һ����μ������ϡNaOH��Һ���ù��̵��ܷ�Ӧ�����ӷ���ʽΪAl3++2H++6OH-=AlO2-+4H2O��E��Ͼ�����ij��������һ���������ܺ�KI���巴Ӧ��д���÷���ʽSO3+2KI=I2+K2SO3��
��4�������£������ܱ������г�ʱ������X�������л�������һ�����壬�����ʽ��O2�������ʵ�鷽����֤֮������ͨ������NaOH��Һ�У��ռ���������һ�������ǵı����������У�����ȼ����˵����O2��
���� ����������B����ϡ����μ�KSCN��Һ�����Һ��Ѫ��ɫ��˵��B�к���+3��Fe����BΪFe2O3����ɫD������ʹƷ����ɫ����DΪSO2����Ԫ���غ��֪A�к���Fe��S��OԪ�أ�A���ȷֽ��������������Ͷ�����������AΪFeSO4��X�����ΪAlCl3•2FeSO4�������������ʵ���Ϊ$\frac{3.2g}{160g/mol}$=0.02mol�����ɶ�������Ϊ$\frac{0.448L}{22.4L/mol}$=0.02mol����Fe��Sԭ��Ϊ1��1��֪����SO3Ϊ0.02mol��4.27g��Ͼ���EΪAlCl3��SO3��AlCl3�����ʵ���Ϊ$\frac{4.27g-0.02mol��80g/mol}{133.5g/mol}$=0.02mol���Դ˽����⣮
��� �⣺����������B����ϡ����μ�KSCN��Һ�����Һ��Ѫ��ɫ��˵��B�к���+3��Fe����BΪFe2O3����ɫD������ʹƷ����ɫ����DΪSO2����Ԫ���غ��֪A�к���Fe��S��OԪ�أ�A���ȷֽ��������������Ͷ�����������AΪFeSO4��X�����ΪAlCl3•2FeSO4�������������ʵ���Ϊ$\frac{3.2g}{160g/mol}$=0.02mol�����ɶ�������Ϊ$\frac{0.448L}{22.4L/mol}$=0.02mol����Fe��Sԭ��Ϊ1��1��֪����SO3Ϊ0.02mol��4.27g��Ͼ���EΪAlCl3��SO3��AlCl3�����ʵ���Ϊ$\frac{4.27g-0.02mol��80g/mol}{133.5g/mol}$=0.02mol��
��1��X�Ļ�ѧʽΪAlCl3•2FeSO4���ʴ�Ϊ��AlCl3•2FeSO4��
��2��A�ֽ�Ļ�ѧ����ʽΪ��2FeSO4$\frac{\underline{\;����\;}}{\;}$SO2��+SO3+Fe2O3��
�ʴ�Ϊ��2FeSO4$\frac{\underline{\;����\;}}{\;}$SO2��+SO3+Fe2O3��
��3����E��Ͼ�������ˮ�����Һ����������Ӧ�������ᣬ���������Ȼ��������ʵ�����ȣ���μ������ϡNaOH��Һ���ù��̵��ܷ�Ӧ�����ӷ���ʽΪ��Al3++2H++6OH-=AlO2-+4H2O��
E�����е�SO3��һ�������ܺ�KI���巴Ӧ���÷�Ӧ����ʽΪSO3+2KI=I2+K2SO3��
�ʴ�Ϊ��Al3++2H++6OH-=AlO2-+4H2O��SO3+2KI=I2+K2SO3��
��4�����ڸ����³�ʱ������X�����ɵ����������ٷֽ����ɶ����������������һ���������ʽ��O2�����������ķ���Ϊ��������ͨ������NaOH��Һ�У��ռ���������һ�������ǵı����������У�����ȼ����˵����O2��
�ʴ�Ϊ��O2��������ͨ������NaOH��Һ�У��ռ���������һ�������ǵı����������У�����ȼ����˵����O2��
���� ���⿼��������ƶϣ�Ϊ�߿��������ͣ�������ѧ���ķ��������������Ŀ��飬��Ŀ�Ƚ��ۺϣ���Ҫѧ����������Ԫ�ػ�����֪ʶ��ȷ��A������ǽ���Ĺؼ�����Ŀ�ѶȽϴ�

 ֥�鿪���γ�������ϵ�д�
֥�鿪���γ�������ϵ�д� ����ѧ��ţ��Ӣ��ϵ�д�
����ѧ��ţ��Ӣ��ϵ�д����ռ�ڴ���۽����ơ��������������ᣮ
| A�� | ȫ�� | B�� | �٢ڢۢ� | C�� | �٢ڢۢ� | D�� | �٢ڢܢ� |
| A�� | ��5.6 LN2�к���n�������ӣ����ӵ�����һ��Ϊ4n | |
| B�� | ��״���£�1L����ȫȼ�����ɵ���̬����ķ�����Ϊ$\frac{6}{22.4}$NA | |
| C�� | 14 g��ϩ�ͱ�ϩ�Ļ��������������������Ϊ0.5NA | |
| D�� | 0.1molC3H6Cl2�л�ѧ������ΪNA |
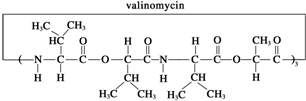
| A�� | �Ӱ�ù����һ�ֵ����� | |
| B�� | �Ӱ�ù����ȫˮ��ɵõ����ְ����� | |
| C�� | �Ӱ�ù����ȫˮ�⣬����һ�ֲ�������ͻ�Ϊͬ���칹�� | |
| D�� | �Ӱ�ù����ȫˮ���IJ����������ֲ��ﻥΪͬϵ�� |
| A�� | 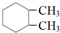 | B�� | 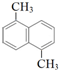 | C�� | ��CH3CO��2O | D�� | 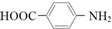 |