��Ŀ����
 �������ڹ���Ԫ��Fe��Ti����C��H��N��O�γɶ��ֻ����
�������ڹ���Ԫ��Fe��Ti����C��H��N��O�γɶ��ֻ������1����H��C��N��O����Ԫ�صĵ縺����С�����˳��Ϊ
��������������ȷ����
A����ΪHCHOΪ���Է��ӣ�ˮҲΪ���Է��ӣ�������������ԭ����HCHO������ˮ��
B��HCHO��CO2�����е�����ԭ�Ӿ�����sp2�ӻ�
C��C6H6�����к���6���Ҽ���1����м���C2H2�ǷǼ��Է���
D��CO2������۵㡢�е㶼�ȶ������辧��ĵ�
��2��Feԭ�ӻ�������Χ�н϶���������Ŀչ������һЩ���ӻ������γ�����
����Feԭ�ӻ������γ������ķ��ӻ�����Ӧ�߱��Ľṹ������
���������������[Fe��CN��6]4-�����
A�����ۼ� B���Ǽ��Լ� C����λ�� D���Ҽ� E���м�
д��һ���� CN-��Ϊ�ȵ�����ĵ��ʷ���ʽ
��3������Ԫ��ԭ�ӵ���Χ�����Ų��������ɽ����ڱ��ֳ������������Ti����
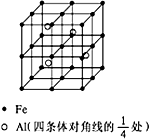
��4��һ��Al-Fe�Ͻ�����徧����ͼ��ʾ����ݴ˻ش��������⣺
��ȷ���úϽ�Ļ�ѧʽ
����������ܶ�=�� g/cm3����˺Ͻ������������Feԭ��֮��ľ��루�ú��ѵĴ���ʽ��ʾ�����ػ���Ϊ
���㣺�����ļ���,Ԫ�ص����ܡ��縺�Եĺ��弰Ӧ��,�����ijɼ����
ר�⣺
��������1���ٸ���Ԫ�����ڱ��縺�Եĵݱ���ɱȽϵ縺��ǿ����
�ڷ����к����ǻ�����ˮ�����γ������������̼Ϊsp�ӻ�������̼̼������6���Ҽ��������6��̼��Ҽ���
��2�����γ������Ӿ߱�������Ϊ������ԭ�Ӿ��пչ����������й¶Ե��Ӷԣ�
���������������[Fe��CN��6]4-��Fe2+��CN-�γ���λ����CN-�д���C��N��������������1���Ҽ�2���м���CN-��Cԭ����1����λ�������Nԭ���滻�ɵ�CN-�ȵ�����ĵ��ʣ�
��3������Tiԭ�Ӻ�������Ų�ʽ���
��4���ٸ��ݾ�̯�����㻯ѧʽ��
�ڼ���������߳������ɽ�һ��������������Feԭ��֮��ľ��룮
�ڷ����к����ǻ�����ˮ�����γ������������̼Ϊsp�ӻ�������̼̼������6���Ҽ��������6��̼��Ҽ���
��2�����γ������Ӿ߱�������Ϊ������ԭ�Ӿ��пչ����������й¶Ե��Ӷԣ�
���������������[Fe��CN��6]4-��Fe2+��CN-�γ���λ����CN-�д���C��N��������������1���Ҽ�2���м���CN-��Cԭ����1����λ�������Nԭ���滻�ɵ�CN-�ȵ�����ĵ��ʣ�
��3������Tiԭ�Ӻ�������Ų�ʽ���
��4���ٸ��ݾ�̯�����㻯ѧʽ��
�ڼ���������߳������ɽ�һ��������������Feԭ��֮��ľ��룮
���
�⣺��1����ͬ����Ԫ�ش�����Ԫ�صĵ縺�����������е縺��C��N��O��H�ĵ縺����С����縺��ΪH��C��N��O���ʴ�Ϊ��H��C��N��O��
��A����ȩ�к����ǻ�����ˮ�����γ��������A��ȷ��
B��������̼Ϊsp�ӻ�����B����
C������̼̼������6���Ҽ���ÿ��̼�������һ���Ҽ�����������һ������12������C����
D�������¶�����̼Ϊ���壬��������Ϊ���壬CO2������۵㡢�е㶼�ȶ������辧��ĵͣ���D��ȷ��
�ʴ�Ϊ��BC��
��2�����γ������Ӿ߱�������Ϊ������ԭ�Ӿ��пչ����������й¶Ե��Ӷԣ�
�ʴ�Ϊ�����й¶Ե��ӣ�
���������������[Fe��CN��6]4-��Fe2+��CN-�γ���λ����CN-�д���C��N������Ϊ���Թ��ۼ�����������1���Ҽ�2���м�������CN-�й��ۼ�����λ�����Ҽ����м���CN-��Cԭ����1����λ�������Nԭ���滻�ɵ�CN-�ȵ�����ĵ���ΪN2��
�ʴ�Ϊ��B��N2��
��3��Tiԭ�Ӻ�������Ų�Ϊ1s22s22p63s23p63d24s2����λ�ڵ������ڵڢ�B�壬����d�����ʴ�Ϊ��d��
��4����Feλ�ڶ��㡢���ġ����ĺ����ģ���ĿΪ8��
+6��
+1+12��
=8��Alλ�ھ������ģ���ĿΪ4��ԭ����Ŀ��ֵΪ2��1����ѧʽ��дΪ��Fe2Al��
�ʴ�Ϊ��Fe2Al��
�ھ�������Ϊ��
����56��2+27��g���辧���߳�Ϊa cm�������Ϊa3cm3�����ܶȹ�ʽ�ã��ѡ�a3=
����56��2+27�������a=2
������Feԭ��֮��ľ���Ϊ�߳���һ�룬�����Ϊ��
��a cm=
cm���ʴ�Ϊ��
��
��A����ȩ�к����ǻ�����ˮ�����γ��������A��ȷ��
B��������̼Ϊsp�ӻ�����B����
C������̼̼������6���Ҽ���ÿ��̼�������һ���Ҽ�����������һ������12������C����
D�������¶�����̼Ϊ���壬��������Ϊ���壬CO2������۵㡢�е㶼�ȶ������辧��ĵͣ���D��ȷ��
�ʴ�Ϊ��BC��
��2�����γ������Ӿ߱�������Ϊ������ԭ�Ӿ��пչ����������й¶Ե��Ӷԣ�
�ʴ�Ϊ�����й¶Ե��ӣ�
���������������[Fe��CN��6]4-��Fe2+��CN-�γ���λ����CN-�д���C��N������Ϊ���Թ��ۼ�����������1���Ҽ�2���м�������CN-�й��ۼ�����λ�����Ҽ����м���CN-��Cԭ����1����λ�������Nԭ���滻�ɵ�CN-�ȵ�����ĵ���ΪN2��
�ʴ�Ϊ��B��N2��
��3��Tiԭ�Ӻ�������Ų�Ϊ1s22s22p63s23p63d24s2����λ�ڵ������ڵڢ�B�壬����d�����ʴ�Ϊ��d��
��4����Feλ�ڶ��㡢���ġ����ĺ����ģ���ĿΪ8��
| 1 |
| 8 |
| 1 |
| 2 |
| 1 |
| 4 |
�ʴ�Ϊ��Fe2Al��
�ھ�������Ϊ��
| 4 |
| NA |
| 4 |
| NA |
| 3 |
| ||
| 1 |
| 2 |
| 3 |
| ||
| 3 |
| ||
�������⿼���˵縺�ԡ�ԭ�ӹ�����ӻ����ȵ����塢�����ṹ����������ȣ�ע��������ռ乹�͵��жϷ�������Ҫѧ���߱�֪ʶ�Ļ�������Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 �ο�������ϵ�д�
�ο�������ϵ�д� ������ѧ��ʱ��ҵϵ�д�
������ѧ��ʱ��ҵϵ�д� ���������ʱ��ѵϵ�д�
���������ʱ��ѵϵ�д� �㽭�¿γ���άĿ�������ʱ��ѵϵ�д�
�㽭�¿γ���άĿ�������ʱ��ѵϵ�д�
�����Ŀ
����˵����ȷ���ǣ�������
| A������H2S����д��H3S���ӣ�Υ���˹��ۼ��ı����� |
| B��s�����p����ص������γɦм� |
| C�����й��ۼ����з����� |
| D������ԭ��֮���γɹ��ۼ�ʱ�����γɶ���Ҽ� |
���й�����ȡ����ˮ��ʵ��װ���������˵���У�����ȷ���ǣ�������
| A���¶ȼƵ�ˮ����Ӧ����������ƿ�е�����ˮ�� |
| B���������е�ˮ�������Ǵ��¿ڽ��룬�Ͽ��ų� |
| C��ʵ������Ҫ��������ƿ�м��뼸�����Ƭ����ֹ���ֱ������� |
| D��������ƿ�����ʯ���� |
��������������Ӧ����PCl5��g��?PCl 3��g��+Cl2��g�� ��2HI��g��?H2��g��+I2��g����2NO2��g��?N2O4��g����һ�������£�����������Ӧ�ﵽ��ѧƽ��ʱ����Ӧ���ת���ʾ���b%�������ֺ��º��ݣ��ֱ��ټ���һ�����ĸ��Եķ�Ӧ���ת���ʣ�������
| A�������� |
| B�������� |
| C�������ڲ��䣬�ۼ��� |
| D���ټ��٣��ڲ��䣬������ |
���������У��Ⱥ����Ӽ��ֺ����ۼ����ǣ�������
| A��NaOH |
| B��NaCl |
| C��H2 |
| D��H2S |
 ij�¶�ʱ����5L���ܱ������У�X��Y��Z������������ʵ�����ʱ��ı仯������ͼ��ʾ������ͼ���������
ij�¶�ʱ����5L���ܱ������У�X��Y��Z������������ʵ�����ʱ��ı仯������ͼ��ʾ������ͼ���������