��Ŀ����
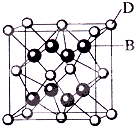 ��֪A��B��C��D��E����Ԫ�����ڱ���ǰ36�ŵ�Ԫ�أ����ǵ� ԭ��������������A������4��Ԫ�ؼȲ���ͬһ�����ֲ���ͬһ���壮B��C��ͬһ���壬D��E��ͬһ���ڣ���֪E�����ڱ���1-18���еĵ�7��Ԫ�أ�D��ԭ��������EС5��D��B���γ����ӻ������侧���ṹ��ͼ��
��֪A��B��C��D��E����Ԫ�����ڱ���ǰ36�ŵ�Ԫ�أ����ǵ� ԭ��������������A������4��Ԫ�ؼȲ���ͬһ�����ֲ���ͬһ���壮B��C��ͬһ���壬D��E��ͬһ���ڣ���֪E�����ڱ���1-18���еĵ�7��Ԫ�أ�D��ԭ��������EС5��D��B���γ����ӻ������侧���ṹ��ͼ����1��A��Ԫ�ط�����
��2��B���⻯��ľ���������
��3����ͼ�п��Կ�����D��B�γɵ����ӻ�����ĵ���ʽΪ
���㣺�����ļ���
ר�⣺��ѧ���뾧��ṹ
��������1����D��E�����ڱ���1-18����E�ŵ�7�п��ж�E�ǵ�4����VIIB���MnԪ�أ�ԭ������Ϊ25������DҲ�ڵ�4���ڣ�D��ԭ��������EС5����ԭ������Ϊ20��ӦΪCaԪ�أ�ͼ�����ӻ�����D��B�����Ӹ�����ֵΪ����8��
+6��
����8=1��2����DΪCa����B�Ļ��ϼ�Ϊ-1�ۣ�ӦΪ�ڢ�A��Ԫ�أ�B��C��ͬһ���壬B��������ǰ�棬BΪF��CΪCl��A������4��Ԫ�ؼȲ���ͬһ�����ֲ���ͬһ���壬����AΪH��
����ԭ�Ӻ�������Ų��ж�Ԫ�������ڱ��е�λ�ã������������ԭ���ͺ��ع�����д���ӵĵ����Ų�ʽ��
��2��BΪFԪ�أ�HF�۷е�ܵͣ����ڷ��Ӿ��壻��ɷ��ӵ�Ԫ�ص縺�Բ��Խ���ӵļ���Խ��FԪ���ǵ縺������Ԫ�أ���HF�ļ��Խϴ�
��3����ͼ�п��Կ����������к���Ca�����Ӹ���Ϊ8��
+6��
=4������F�����Ӹ���Ϊ8�����߱�ֵΪ1��2����ѧʽΪCaF2��CaF2Ϊ���ӻ������д������ʽ��
�����ܶȼ��㹫ʽ��=
���㣮
| 1 |
| 8 |
| 1 |
| 2 |
����ԭ�Ӻ�������Ų��ж�Ԫ�������ڱ��е�λ�ã������������ԭ���ͺ��ع�����д���ӵĵ����Ų�ʽ��
��2��BΪFԪ�أ�HF�۷е�ܵͣ����ڷ��Ӿ��壻��ɷ��ӵ�Ԫ�ص縺�Բ��Խ���ӵļ���Խ��FԪ���ǵ縺������Ԫ�أ���HF�ļ��Խϴ�
��3����ͼ�п��Կ����������к���Ca�����Ӹ���Ϊ8��
| 1 |
| 8 |
| 1 |
| 2 |
�����ܶȼ��㹫ʽ��=
| m |
| V |
���
�⣺��1������D��E�����ڱ���1-18����E�ŵ�7�п��ж�E�ǵ�4����VIIB���MnԪ�أ�ԭ������Ϊ25������DҲ�ڵ�4���ڣ�D��ԭ��������EС5����ԭ������Ϊ20��ӦΪCaԪ�أ�ͼ�����ӻ�����D��B�����Ӹ�����ֵΪ����8��
+6��
����8=1��2����DΪCa����B�Ļ��ϼ�Ϊ-1�ۣ�ӦΪ�ڢ�A��Ԫ�أ�B��C��ͬһ���壬B��������ǰ�棬BΪF��CΪCl��A������4��Ԫ�ؼȲ���ͬһ�����ֲ���ͬһ���壬����AΪH��
EΪMnԪ�أ�λ�����ڱ��������ڵ����У���Ӧλ��VIIB�壬ԭ�ӵĺ�����ӵ��Ų�ʽΪ��ʧȥ2������ 1s22s22p63s23p63d54s2�����ӵĵ����Ų�ʽΪ 1s22s22p63s23p63d5��
�ʴ�Ϊ��H���������ڵ�VIIB�壻 1s22s22p63s23p63d5��
��2��BΪFԪ�أ�CΪClԪ�أ�HF�۷е�ܵͣ����ڷ��Ӿ��壻��ɷ��ӵ�Ԫ�ص縺�Բ��Խ���ӵļ���Խ��FԪ���ǵ縺������Ԫ�أ���HF�ļ��Խϴ�
�ʴ�Ϊ�����ӣ�HF��
��3����ͼ�п��Կ����������к���Ca�����Ӹ���Ϊ8��
+6��
=4������F�����Ӹ���Ϊ8�����߱�ֵΪ1��2����ѧʽΪCaF2��CaF2Ϊ���ӻ�����������ʽΪ��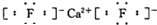 ��
��
��������=
=
�Tag?cm-3����V=
��
�ʴ�Ϊ��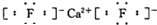 ��V=
��V=
��
| 1 |
| 8 |
| 1 |
| 2 |
EΪMnԪ�أ�λ�����ڱ��������ڵ����У���Ӧλ��VIIB�壬ԭ�ӵĺ�����ӵ��Ų�ʽΪ��ʧȥ2������ 1s22s22p63s23p63d54s2�����ӵĵ����Ų�ʽΪ 1s22s22p63s23p63d5��
�ʴ�Ϊ��H���������ڵ�VIIB�壻 1s22s22p63s23p63d5��
��2��BΪFԪ�أ�CΪClԪ�أ�HF�۷е�ܵͣ����ڷ��Ӿ��壻��ɷ��ӵ�Ԫ�ص縺�Բ��Խ���ӵļ���Խ��FԪ���ǵ縺������Ԫ�أ���HF�ļ��Խϴ�
�ʴ�Ϊ�����ӣ�HF��
��3����ͼ�п��Կ����������к���Ca�����Ӹ���Ϊ8��
| 1 |
| 8 |
| 1 |
| 2 |
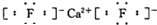 ��
����������=
| m |
| V |
| ||
| V |
| 4��78g/mol |
| ag?cm-3��6.02��1023mol-1 |
�ʴ�Ϊ��
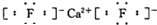 ��V=
��V=| 4��78g/mol |
| ag?cm-3��6.02��1023mol-1 |
������������Ԫ���ƶ�Ϊ������������Ԫ�����ơ�λ�ü������Ų�ʽ֮�⣬��������й�Ӱ�쾧���۷е�ߵ͵�ԭ������������⣮������ۺ��Խϴ����ϸ��������ʽṹ�����ʵ�����֪ʶ��

��ϰ��ϵ�д�
�����Ŀ
Ϊȷ�����о����ڿ����е������Ƿ���ʣ���ѡ�����Լ������������ʣ����ܴﵽĿ���ǣ�������
| A��Na2SO3��Һ��BaCl2�� |
| B��FeCl2��Һ��KSCN�� |
| C��KI��������Һ�� |
| D��NaOH��Һ�����ᣩ |
��������һ�ֿ���������ṹ��ͼ��ʾ������������ˮ�������ж������ȼ��飮����˵������ȷ���ǣ������� 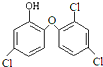

| A���������ķ���ʽ��C12H7Cl3O2 |
| B�����ȼ�����������ͬ���칹�� |
| C��1 mol�������������6 mol H2��Ӧ |
| D��1 mol�������������4mol NaOH��Ӧ |
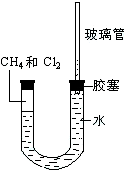 ��ͼ��ʾ��U�ܵ���˱�ˮ�ͽ�������м���������������Ϊ1��4���Ļ�����壬�ٶ�������ˮ���ܽ�ȿ��Ժ��ԣ�����װ�÷������й����ĵط����û�����建���ط�Ӧһ��ʱ�䣮
��ͼ��ʾ��U�ܵ���˱�ˮ�ͽ�������м���������������Ϊ1��4���Ļ�����壬�ٶ�������ˮ���ܽ�ȿ��Ժ��ԣ�����װ�÷������й����ĵط����û�����建���ط�Ӧһ��ʱ�䣮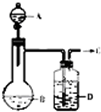 Ϊ����ȡ���﴿��������C����������ͼ��ʾװ�ã�����C���ٶȿ�ͨ�����ڵ���Һ��A���ٶȵķ��������ƣ���֪C��һ����ɫ��ζ������ʹ���ָʾ����ɫ��Ҳ�����ڿ�����ȼ�յ����壮
Ϊ����ȡ���﴿��������C����������ͼ��ʾװ�ã�����C���ٶȿ�ͨ�����ڵ���Һ��A���ٶȵķ��������ƣ���֪C��һ����ɫ��ζ������ʹ���ָʾ����ɫ��Ҳ�����ڿ�����ȼ�յ����壮