��Ŀ����
17�����λ�����Ⱦ��������̬��������Ϊȫ��Ĺ�ʶ����1������Ӧ�����ռ������¡���ɫ��Ⱦ������������Ӧ����������a������ĸ����־������Ͳ�ڣ�

��2����ҵ��ˮ�账����������ŷţ����з�ˮ�����ķ�����������ad������ĸ����
a�����кͷ���ȥ��ˮ�е��� b���û�������ȥ��ˮ�е��ؽ�������
c����������ȥ��ˮ�е������� d����������ԭ�������ࡢ�軯��
��3��Ϊ�˼���úȼ�ղ�����SO2�Դ�����ɵ���Ⱦ������ú�м�������ʯ��ʯ��ʯ��ʯ��úȼ��ʱ������SO2�������е�O2��Ӧ������CaSO4��CO2��д���÷�Ӧ�Ļ�ѧ����2CaCO3+2SO2+O2=2CaSO4+2CO2��
��4��Ϊ�˸��ƿ���������������ƴ����ж�������������̳�����Ⱦ����ŷ�����Ϊ���ƴ����ж���������ŷţ�����ȡ�Ĵ�ʩ�ǶԻ�ʯȼ�Ͻ�������������������װβ��������װ�ã�ʹ���е��к�����NO��COת��Ϊ�����壬�÷�Ӧ�Ļ�ѧ����ʽΪ2NO+2CO$\frac{\underline{\;����\;}}{\;}$N2+2CO2��
���� ��1������ɫ��Ⱦ������������--�������ڿɻ���������
��2������������ԭ�������ڳ����ࡢ�軯������������ڳ�ȥ��ˮ�е�����������������ڳ�ȥ��ˮ�е��ؽ������ӣ��кͷ������ڳ�ȥ��ˮ�е��
��3��ʯ��ʯ��úȼ��ʱ������SO2�������е�O2��Ӧ������CaSO4��CO2��
��4������ú������ʯ��ʯ������������������װβ��������װ�ã�ʹ���е��к�����NO��COת��Ϊ�����壬����N2��CO2��
��� �⣺��1������ɫ��Ⱦ������������---�������ڿɻ�����������ѡ��a��
��2��������ԭ�������ڳ����ࡢ�軯������������ڳ�ȥ��ˮ�е�����������������ڳ�ȥ��ˮ�е��ؽ������ӣ��кͷ������ڳ�ȥ��ˮ�е��ᣬ��ѡ��ad��
��3������Ϣ֪ʯ��ʯ��úȼ��ʱ������SO2�������е�O2��Ӧ������CaSO4��CO2����ѧ����ʽΪ��2CaCO3+2SO2+O2=2CaSO4+2CO2���ʴ�Ϊ��2CaCO3+2SO2+O2=2CaSO4+2CO2��
��4������ú������ʯ��ʯ��������������������ƣ�������װβ��������װ�ã�ʹ���е��к�����NO��COת��Ϊ�����壬����N2��CO2��
��Ӧ�ķ���ʽΪ2NO+2CO$\frac{\underline{\;����\;}}{\;}$N2+2CO2���ʴ�Ϊ����������2NO+2CO$\frac{\underline{\;����\;}}{\;}$N2+2CO2��
���� ���⿼�黷����Ⱦ�������������ڻ�ѧ�����彡���Ŀ��飬Ϊ�߿��������ͺ�Ƶ���㣬����������ѧ�������ÿ�ѧ�������ѶȲ���ע����ػ���֪ʶ�Ļ��ۣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | ʵ�����ϵľƾ����㵹��ȼ��������������ˮ���� | |
| B�� | ����������Һ�������ڣ����������۾���������� | |
| C�� | Ƥ���Ͻ��н϶��Ũ���ᣬ�Ͻ���ˮ��ϴ | |
| D�� | ��Һʱ���²�Һ����¶˷ų����ϲ�Һ����Ͽڵ��� |
| A�� | ͬ������H2��Cl2��H2�ķ�����һ����Cl2�� | |
| B�� | 0.5mol���������0.5g | |
| C�� | Ħ�����������������Ӷ��ٵ�һ�������� | |
| D�� | 0.1mol H2SO4������ԭ�����ľ�ȷֵΪ1.204��1023 |
| A�� | CO2+H2O�TH2CO3���Ϸ�Ӧ | |
| B�� | Cu2��OH��2CO3�T2CuO+H2O+CO2�� �ֽⷴӦ | |
| C�� | CuO+CO�TCu+CO2 �û���Ӧ | |
| D�� | NaCl+AgNO3�TAgCl��+NaNO3���ֽⷴӦ |
| A�� | �۳� | B�� | �����ЧӦ | C�� | �����˶� | D�� | ������ |
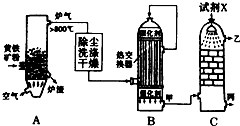 �ϳɰ���ҵ�����Ṥҵ�����Ṥҵ�ǻ�ѧ��ҵ����Ҫ��ɲ��֣���ش��������⣺
�ϳɰ���ҵ�����Ṥҵ�����Ṥҵ�ǻ�ѧ��ҵ����Ҫ��ɲ��֣���ش��������⣺