��Ŀ����
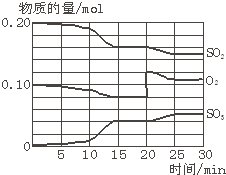
��֪��Na2S2O3+H2SO4=Na2SO4+SO2��+S��+H2O��ijͬѧ̽��������������ᷴӦ���ʵ�Ӱ�����أ������һϵ������ʵ�飺
��1��ʵ�������Ϊʲô���ò���һ��ʱ���ڷų�SO2�����������ʾ�÷�Ӧ�Ļ�ѧ��Ӧ���ʴ�С ��
��2����Ҫ̽���¶ȶԷ�Ӧ���ʵ�Ӱ�죬����ѡ��ʵ���е���Щ������Ƚ� ������ţ���
��3�����÷�Ӧ��I2+2Na2S2O3=Na2S4O6+2NaI �ɶ����ⶨ������������ƵĴ��ȣ��ֳ�ȡWg��������ƹ�����Ʒ���250mL����Һ��ȡV mL���ڴ���ƿ�У�����ָʾ�����ú�I2Ϊa mol?L-1�ĵ�ˮ�ζ������ĵ�ˮb mL���ش����и��⣺
��ʵ���еζ���Ӧѡ�� ������ʽ���ʽ���ζ��ܣ��ζ�ǰ����ѡ�� ��Ϊָʾ�����ζ��յ�ʱ������Ϊ
�����в��������������Ƶĺ���ƫ�����
A���ζ�ǰ�ζ����������ݣ��ζ���������ʧ
B����ȡ��ˮ����̶�ʱ���ζ�ǰƽ�ӣ��ζ�����
C���ζ������У���ƿ���ھ��ң���������Һ����
D���ζ���������ˮϴ����ֱ�Ӽ����ˮ��Һ
����Ʒ����������ƴ��ȵı���ʽΪ ��
| ʵ�� | ��Ӧ�¶�/�� | Na2S2O3��Һ | ϡH2SO4 | H2O | ||
| V/mL | c/��mol?L-1�� | V/mL | c/��mol?L-1�� | V/mL | ||
| A | 10 | 5 | 0.1 | 5 | 0.1 | 5 |
| B | 10 | 5 | 0.1 | 5 | 0.1 | 10 |
| C | 30 | 5 | 0.1 | 5 | 0.1 | 5 |
| D | 30 | 5 | 0.2 | 5 | 0.2 | 10 |
��2����Ҫ̽���¶ȶԷ�Ӧ���ʵ�Ӱ�죬����ѡ��ʵ���е���Щ������Ƚ�
��3�����÷�Ӧ��I2+2Na2S2O3=Na2S4O6+2NaI �ɶ����ⶨ������������ƵĴ��ȣ��ֳ�ȡWg��������ƹ�����Ʒ���250mL����Һ��ȡV mL���ڴ���ƿ�У�����ָʾ�����ú�I2Ϊa mol?L-1�ĵ�ˮ�ζ������ĵ�ˮb mL���ش����и��⣺
��ʵ���еζ���Ӧѡ��
�����в��������������Ƶĺ���ƫ�����
A���ζ�ǰ�ζ����������ݣ��ζ���������ʧ
B����ȡ��ˮ����̶�ʱ���ζ�ǰƽ�ӣ��ζ�����
C���ζ������У���ƿ���ھ��ң���������Һ����
D���ζ���������ˮϴ����ֱ�Ӽ����ˮ��Һ
����Ʒ����������ƴ��ȵı���ʽΪ
���㣺̽���¶ȡ�ѹǿ�Ի�ѧ��Ӧ���ʵ�Ӱ��,̽�����ʵ���ɻ�������ʵĺ���
ר�⣺ʵ�������
��������1�����ڶ�����������Һ���ܽ⣬�ⶨ�������������ȷ
��2����Ҫ̽���¶ȶԷ�Ӧ���ʵ�Ӱ�죬��Ҫʹ�������ͬ��
��3���ٵ�������ʴ���������������
�ڸ���c=
=
�жϲ�������ԭ��
�������������Ƶ���������Ʒ����������ƴ���=��
��100%��
��2����Ҫ̽���¶ȶԷ�Ӧ���ʵ�Ӱ�죬��Ҫʹ�������ͬ��
��3���ٵ�������ʴ���������������
�ڸ���c=
| n(���������) |
| V |
| 2n(I2) |
| V |
�������������Ƶ���������Ʒ����������ƴ���=��
| ������������� |
| ��Ʒ���� |
���
�⣺��1�����ڶ�����������Һ���ܽ�Ƚϴ��ֶ��������ܽ⣬�ⶨ�������������ȷ���ʴ�Ϊ�����ڶ�����������Һ���ܽ⣬�ⶨ�������������ȷ��
��2����Ҫ̽���¶ȶԷ�Ӧ���ʵ�Ӱ�죬��ҪNa2S2O3��Һ��ϡH2SO4Ũ����ͬ��A��C��Ũ����ͬ���ʴ�Ϊ��AC��
��3���ٵ�������ʴ����Ҫѡȡ��ʽ�ζ��ܣ��ζ���ɺ����ʣ�࣬�����õ�����Һ������ⵥ�ʣ����������ⵥ�ʱ���ɫ��
�ʴ�Ϊ����ʽ�����ۣ����۱�����
�ڸ���I2+2Na2S2O3=Na2S4O6+2NaI ֪n��Na2S2O3��=2n��I2��������c=
��
A���ζ�ǰ�ζ����������ݣ��ζ���������ʧ��ʹ�õζ��ĵ�ˮ��������������Ƶĺ���ƫ��ѡ��
B����ȡ��ˮ����̶�ʱ���ζ�ǰƽ�ӣ��ζ����ӣ�ʹ�õ�ˮ���ƫС����������Ƶĺ���ƫС���ʲ�ѡ��
C���ζ������У���ƿ���ھ��ң���������Һ������ʹ�õ�ˮ���ƫС����������Ƶĺ���ƫС���ʲ�ѡ��
D���ζ���������ˮϴ����ֱ�Ӽ����ˮ��Һ��ʹ�õζ��ĵ�ˮ��������������Ƶĺ���ƫ��ѡ��
�ʴ�Ϊ��AD��
�۸���I2+2Na2S2O3=Na2S4O6+2NaI��n��I2��=amol/L��b��10-3L��֪���ĵ�Na2S2O3�����ʵ���=2n��I2��=2amol/L��b��10-3L�������ֳ�ȡWg��������ƹ�����Ʒ���250mL����Һ��ȡV mL���ڴ���ƿ�У�֪���ζ����������������Ϊ
��Wg������Ʒ����������ƴ��ȵı���ʽΪ
��100%��
�ʴ�Ϊ��
��100%��
��2����Ҫ̽���¶ȶԷ�Ӧ���ʵ�Ӱ�죬��ҪNa2S2O3��Һ��ϡH2SO4Ũ����ͬ��A��C��Ũ����ͬ���ʴ�Ϊ��AC��
��3���ٵ�������ʴ����Ҫѡȡ��ʽ�ζ��ܣ��ζ���ɺ����ʣ�࣬�����õ�����Һ������ⵥ�ʣ����������ⵥ�ʱ���ɫ��
�ʴ�Ϊ����ʽ�����ۣ����۱�����
�ڸ���I2+2Na2S2O3=Na2S4O6+2NaI ֪n��Na2S2O3��=2n��I2��������c=
| n(���������) |
| V |
A���ζ�ǰ�ζ����������ݣ��ζ���������ʧ��ʹ�õζ��ĵ�ˮ��������������Ƶĺ���ƫ��ѡ��
B����ȡ��ˮ����̶�ʱ���ζ�ǰƽ�ӣ��ζ����ӣ�ʹ�õ�ˮ���ƫС����������Ƶĺ���ƫС���ʲ�ѡ��
C���ζ������У���ƿ���ھ��ң���������Һ������ʹ�õ�ˮ���ƫС����������Ƶĺ���ƫС���ʲ�ѡ��
D���ζ���������ˮϴ����ֱ�Ӽ����ˮ��Һ��ʹ�õζ��ĵ�ˮ��������������Ƶĺ���ƫ��ѡ��
�ʴ�Ϊ��AD��
�۸���I2+2Na2S2O3=Na2S4O6+2NaI��n��I2��=amol/L��b��10-3L��֪���ĵ�Na2S2O3�����ʵ���=2n��I2��=2amol/L��b��10-3L�������ֳ�ȡWg��������ƹ�����Ʒ���250mL����Һ��ȡV mL���ڴ���ƿ�У�֪���ζ����������������Ϊ
| VmL |
| 250mL |
| 2amol/L��b��10-3L | ||
|
�ʴ�Ϊ��
| 2amol/L��b��10-3L | ||
|
���������⿼�����¶ȶԻ�ѧ��Ӧ���ʵ�Ӱ������ʰٷֺ�����̽�������ض�ѧ�������������ͽ��ⷽ����ָ����ѵ����ּ�ڿ���ѧ��������û���֪ʶ���ʵ�������������

��ϰ��ϵ�д�
�����Ŀ
CuSO4��һ����Ҫ�Ļ���ԭ�ϣ����й��Ʊ�;����������ͼ��ʾ������˵����ȷ���ǣ�������
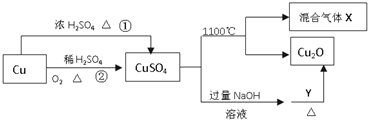

| A�������;���٣�;���ڸ��õ���������ɫ��ѧ˼�� |
| B��Y������������Һ |
| C��CuSO4��1100��ֽ���������X������SO2��SO3�Ļ������ |
| D����CuSO4��Һ��������ȴ���ᾧ�����Ƶõ������� |
2006��4��22���ǵ�37������������ա������������ǡ��ƴ�����--��ϧ��Դ��������չ�������л����Ϊ��������һ������ǣ�������
| A����չѭ�����ã�������Դ��Լ����ἰ����������� |
| B��������ɫ��ѧ�������գ����ԭ�������ʣ����������Ѻ��ͻ�����ϵ |
| C����������ɭ�֣����������Ƕ�ľ�ĵ����� |
| D������������������Դ���ƹ����Ҵ����ʹ��洫ͳ��ȼ�� |
CH4��H2�������10mL����ȫȼ�պ�����6mL CO2���壨��������Ϊ��ͬ״���²ⶨ��������������CH4��H2�������Ϊ��������
| A��2��3 | B��3��2 |
| C��2��1 | D��1��2 |
��֪��Cu2O+2H+=Cu2++Cu+H2OΪ����ij����ͭ��ĩ��Ʒ���Ƿ���Cu2O��ijͬѧ��������·��������в��������ǣ�������
| A��������COͨ��������Ʒ����ȴ�������������������С������Cu2O |
| B�������������ͨ�����ȵ���Ʒ����ͨ����ˮ����ͭ������ˮ����ͭ����������Cu2O |
| C��ȡ������Ʒ������ϡ���ᣬ��������۲쵽��Һ����ɫ������Cu2O |
| D��ȡ������Ʒ������ϡ���ᣬ��������۲쵽��Һ����ɫ������Cu2O |