��Ŀ����
��2013?������һģ����������;�㷺�Ļ���ԭ�ϣ�������ˮ������ˮ�����������ʹ����ȣ�
��1����ҵ������ͭ�ķ����࣮ܶ
�ٷ���һ����Ũ�����ͭ��ȡ����ͭ���÷�Ӧ�Ļ�ѧ����ʽ��
�ڷ���������ϡ���ᡢͭ����������ȡ����ͭ����������Ҫ������ͼ��ʾ��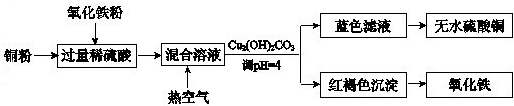
ϡ���ᡢͭ����������Ӧ�Ļ�ѧ����ʽ��
��2���������������������Ṥҵβ���еĶ�������ͬʱ�Ƶ�����泥���Ҫ�Ĺ���������ͼ��ʾ��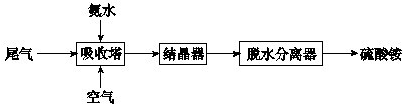
���������з�����Ӧ�Ļ�ѧ����ʽ��
�������ݱ���������������Һ��pH��5.5��6.0֮�䣬����Ч�ʽϸߣ�������һ��������β��ʱ��������Һ��pH�ķ�����
��1����ҵ������ͭ�ķ����࣮ܶ
�ٷ���һ����Ũ�����ͭ��ȡ����ͭ���÷�Ӧ�Ļ�ѧ����ʽ��
Cu+2H2SO4��Ũ��
CuSO4+SO2��+2H2O
| ||
Cu+2H2SO4��Ũ��
CuSO4+SO2��+2H2O
���˷������ȱ����
| ||
��������Ⱦ������
��������Ⱦ������
���ڷ���������ϡ���ᡢͭ����������ȡ����ͭ����������Ҫ������ͼ��ʾ��

ϡ���ᡢͭ����������Ӧ�Ļ�ѧ����ʽ��
Fe2O3+6H+�T2Fe3++3H2O��2Fe3++Cu�T2Fe2++Cu2+��
Fe2O3+6H+�T2Fe3++3H2O��2Fe3++Cu�T2Fe2++Cu2+��
��������Һ��ͨ���ȿ����ķ�Ӧ�����ӷ���ʽ��4Fe2++4H++O2�T4Fe2++2H2O
4Fe2++4H++O2�T4Fe2++2H2O
������Һ�õ���ˮ����ͭ��ʵ����������ȡ�����
���ȡ�����
����2���������������������Ṥҵβ���еĶ�������ͬʱ�Ƶ�����泥���Ҫ�Ĺ���������ͼ��ʾ��

���������з�����Ӧ�Ļ�ѧ����ʽ��
4NH3?H2O+2SO2+O2�T2��NH4��2SO4+2H2O
4NH3?H2O+2SO2+O2�T2��NH4��2SO4+2H2O
���������ݱ���������������Һ��pH��5.5��6.0֮�䣬����Ч�ʽϸߣ�������һ��������β��ʱ��������Һ��pH�ķ�����
���ڰ�ˮ������
���ڰ�ˮ������
����������1����ͭ��Ũ������ȷ�Ӧ��������ͭ�����������ˮ����Ӧ���ɵĶ�����������Ⱦ���������壻
����������ͼ�����жϣ�ϡ���ᡢͭ����������Ӧ����������ϡ���ᷴӦ������������ˮ����������ͭ��Ӧ�����Ȼ��������Ȼ�ͭ��ͨ���ȵĿ�����Ϊ��������������Ϊ���������ӣ�������ҺPH��ȥ�����ӣ���Һ�еõ����ʹ���ķ���������ˮ�ᾧ������
��2�����������Ṥҵβ���еĶ�������ͬʱ�Ƶ�����泥���������ͨ���������������������������Ϊ�����Σ�
������������Һ��pH��5.5��6.0֮�䣬����Ч�ʽϸߣ�������һ��������β��ʱ��������Һ��pH�ķ����ǿ��ư�����������
����������ͼ�����жϣ�ϡ���ᡢͭ����������Ӧ����������ϡ���ᷴӦ������������ˮ����������ͭ��Ӧ�����Ȼ��������Ȼ�ͭ��ͨ���ȵĿ�����Ϊ��������������Ϊ���������ӣ�������ҺPH��ȥ�����ӣ���Һ�еõ����ʹ���ķ���������ˮ�ᾧ������
��2�����������Ṥҵβ���еĶ�������ͬʱ�Ƶ�����泥���������ͨ���������������������������Ϊ�����Σ�
������������Һ��pH��5.5��6.0֮�䣬����Ч�ʽϸߣ�������һ��������β��ʱ��������Һ��pH�ķ����ǿ��ư�����������
����⣺��1����ͭ��Ũ������ȷ�Ӧ��������ͭ�����������ˮ����Ӧ�Ļ�ѧ����ʽΪ��Cu+2H2SO4��Ũ���TCuSO4+SO2��+2H2O����Ӧ���ɵĶ�����������Ⱦ���������壬����������Ⱦ�����壻�ʴ�Ϊ��Cu+2H2SO4��Ũ��
CuSO4+SO2��+2H2O������������Ⱦ�����壻
����������ת��ͼ������ͭ��Ҫ�������������Ӧ���ɵ��������ܽ�����ͭ���ӣ���ͨ�����������������Ϊ�����ӣ�������ҺPHʹ�����ӳ�����ȥ���õ�����ͭ��Һ����������Ũ���ᾧ�õ�����ͭ���壻��Ӧ�����ӷ���ʽΪ��Fe2O3+6H+�T2Fe3++3H2O��2Fe3++Cu�T2Fe2++Cu2+����Fe2O3+Cu+6H+�T3H2O+2Fe2++Cu2+����ͨ�����������������������������Ϊ�����ӵķ�Ӧ����Ӧ���ӷ���ʽΪ��4Fe2++4H++O2�T4Fe3++2H2O���õ�����ͭ��Һ���������õ�����ͭ���壻���ȡ�������
�ʴ�Ϊ��Fe2O3+6H+�T2Fe3++3H2O��2Fe3++Cu�T2Fe2++Cu2+��4Fe2++4H++O2�T4Fe2++2H2O�����ȡ�������
��2������������ͼ�������������з�����Ӧ�Ƕ�������һˮ��������Ӧ��������狀�ˮ����Ӧ�Ļ�ѧ����ʽΪ��4NH3?H2O+2SO2+O2�T2��NH4��2SO4+2H2O��
�ʴ�Ϊ��4NH3?H2O+2SO2+O2�T2��NH4��2SO4+2H2O��
������������Һ��pH��5.5��6.0֮�䣬����Ч�ʽϸߣ�������һ��������β��ʱ��������Һ��pH�ķ����ǣ����ڰ�ˮ���������ʴ�Ϊ�����ڰ�ˮ��������
| ||
����������ת��ͼ������ͭ��Ҫ�������������Ӧ���ɵ��������ܽ�����ͭ���ӣ���ͨ�����������������Ϊ�����ӣ�������ҺPHʹ�����ӳ�����ȥ���õ�����ͭ��Һ����������Ũ���ᾧ�õ�����ͭ���壻��Ӧ�����ӷ���ʽΪ��Fe2O3+6H+�T2Fe3++3H2O��2Fe3++Cu�T2Fe2++Cu2+����Fe2O3+Cu+6H+�T3H2O+2Fe2++Cu2+����ͨ�����������������������������Ϊ�����ӵķ�Ӧ����Ӧ���ӷ���ʽΪ��4Fe2++4H++O2�T4Fe3++2H2O���õ�����ͭ��Һ���������õ�����ͭ���壻���ȡ�������
�ʴ�Ϊ��Fe2O3+6H+�T2Fe3++3H2O��2Fe3++Cu�T2Fe2++Cu2+��4Fe2++4H++O2�T4Fe2++2H2O�����ȡ�������
��2������������ͼ�������������з�����Ӧ�Ƕ�������һˮ��������Ӧ��������狀�ˮ����Ӧ�Ļ�ѧ����ʽΪ��4NH3?H2O+2SO2+O2�T2��NH4��2SO4+2H2O��
�ʴ�Ϊ��4NH3?H2O+2SO2+O2�T2��NH4��2SO4+2H2O��
������������Һ��pH��5.5��6.0֮�䣬����Ч�ʽϸߣ�������һ��������β��ʱ��������Һ��pH�ķ����ǣ����ڰ�ˮ���������ʴ�Ϊ�����ڰ�ˮ��������
���������⿼����Ũ���ᡢ�����仯����ͭ���仯�������ʵ�Ӧ�ã����ӷ���ʽ����д���������ӵķ������Լ�ѡ��ԭ����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д�
���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д� ����ѵ��ϵ�д�
����ѵ��ϵ�д� ��ĩ�����ϵ�д�
��ĩ�����ϵ�д�
�����Ŀ
