��Ŀ����
8����������泥�H2NCOONH4����һ���ֽ⡢��ˮ��İ�ɫ���壬ij�о�С����ͼ1��ʾ��ʵ��װ�ã������������ƹ��塢Ũ��ˮ���ɱ�����ԭ���Ʊ���������泥���Ҫ��Ӧԭ����2NH3��g��+CO2��g��=H2NCOONH4��s����H��0��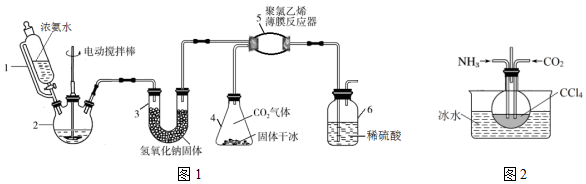
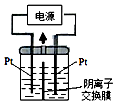
��1������2��������������ƿ������3��NaOH����������Ǹ��ﰱ������ֹ���ɵİ��������ˮ�⣮
��2������6��һ�������ǿ���ԭ��������Ӧ����ϵ����ַ�Ӧ������Ӧ���ڹ۲쵽װ����ϡ������Һ�в������ݣ�Ӧ�üӿ죨��ӿ족�������������ı䡱���������������٣�
��3������һ���Ʊ���������淋ķ�Ӧ����CCl4�䵱���Խ��ʣ���ͼ2��
�����ޱ�ˮ����������ֽ���������[��ѧʽΪCO��NH2��2]����д��������������ȷֽ�Ļ�ѧ��Ӧ����ʽ��H2NCOONH4$\frac{\underline{\;\;��\;\;}}{\;}$CO��NH2��2+H2O��
�ڵ�CCl4Һ���в����϶ྦྷ��������ʱ����ֹͣ�� Ӧ�����˷���õ��ֲ�Ʒ��Ϊ�˽����ôֲ�Ʒ����ɲ�ȡ�ķ�����C��
A������ B����ѹ���Ⱥ�� C������Ⱥ��
��4����֪��������刺���ȫˮ��Ϊ̼����泥���÷�Ӧ�Ļ�ѧ����ʽΪNH2COONH4+2H2O=NH4HCO3+NH3•H2O��
Ϊ�ⶨij��Ʒ�а�������淋�����������ij�о�С���ø���Ʒ����ʵ�飮��֪���ʲ����뷴Ӧ���벹�������ⶨij��Ʒ�а������������������ʵ�鷽��������ƽ��ȡһ����������Ʒ����ˮ�ܽ⣬������������ʯ��ˮ����ַ�Ӧ���ˡ�������ˮϴ�����ó��������������������������ظ�2��3�Σ�����������ȡƽ��ֵ���м��㣨��ѡ�Լ�������ˮ������ʯ��ˮ��BaCl2��Һ����
���� ��1���������������֪�������������ˮ�⣬U�����������Ƹ��ﰱ����
��2������Ӧ���ڹ۲쵽װ����ϡ������Һ�в������ݣ�˵���ж�����̼�ų���ͨ�백���������㣻
��3���ٰ�����������ȷֽ������CO��NH2��2����ԭ���غ��֪������ˮ��
�ڰ�������������ֽ⣬��ȡ��ѹ���º�ɣ�
��4�������������ȫˮ��Ϊ̼����狀�һˮ�ϰ�������Ʒ���ˮ�⣬�õ�̼����泥�������������ʯ��ˮ����̼������ӣ����ݳ�����������̼Ԫ�������غ�ɵ���Ʒ�а�������淋������������ݴ˷�����
��� �⣺��1������2Ϊ������ƿ�����ڰ����������ˮ�⣬U�����������Ƹ��ﰱ������ֹ���ɵİ��������ˮ�⣻
�ʴ�Ϊ��������ƿ�����ﰱ������ֹ���ɵİ��������ˮ�⣻
��2������Ӧ���ڹ۲쵽װ����ϡ������Һ�в������ݣ�˵���ж�����̼�ų���ͨ�백���������㣬Ӧ���ʵ��ӿ�������������٣�
�ʴ�Ϊ���ӿ죻
��3���ٰ�����������ȷֽ������CO��NH2��2����ԭ���غ��֪������ˮ����Ӧ����ʽΪNH2COONH4$\frac{\underline{\;\;��\;\;}}{\;}$CO��NH2��2+H2O��
�ʴ�Ϊ��H2NCOONH4$\frac{\underline{\;\;��\;\;}}{\;}$CO��NH2��2+H2O��
�ڰ�������������ֽ⣬��ȡ��ѹ���º�ɣ�������Ⱥ��ڣ�
�ʴ�Ϊ��C��
��4�������������ȫˮ��Ϊ̼����狀�һˮ�ϰ�����ӦΪ��NH2COONH4+2H2O=NH4HCO3+NH3•H2O������Ʒ���ˮ�⣬�õ�̼����泥�������������ʯ��ˮ����̼������ӣ����ݳ�����������̼Ԫ�������غ�ɵ���Ʒ�а�������淋������������ʲⶨij��Ʒ�а�����������������ķ���Ϊ������ƽ��ȡһ����������Ʒ����ˮ�ܽ⣬������������ʯ��ˮ����ַ�Ӧ���ˡ�������ˮϴ�����ó��������������������������ظ�2��3�Σ�����������ȡƽ��ֵ���м��㣻
�ʴ�Ϊ��NH2COONH4+2H2O=NH4HCO3+NH3•H2O��������������ʯ��ˮ����ַ�Ӧ���ˡ�������ˮϴ�����ó��������������������������ظ�2��3�Σ�
���� ���⿼�������Ʊ�ʵ�顢���ʵķ����ᴿ����װ�õķ������ۡ�����Ϣ�Ļ�ȡ��Ǩ��Ӧ�õȣ��ؼ���ע��̼Ԫ��ȫ��ת��Ϊ̼��ƣ���Ҫѧ���߱���ʵ�Ļ��������Ӧ����������Ŀ�Ѷ��еȣ�

 �������Ͽ�ʱͬ��ѵ��ϵ�д�
�������Ͽ�ʱͬ��ѵ��ϵ�д� �������ͬ����ϰϵ�д�
�������ͬ����ϰϵ�д�| A�� | FeS2$\stackrel{O_{2}/����}{��}$SO2$\stackrel{H_{2}O_{2}��aq��}{��}$H2SO4 | |
| B�� | Cu2��OH��2CO3$\stackrel{H_{2}SO_{4}}{��}$CuSO4��aq��$\stackrel{Na}{��}$Cu | |
| C�� | MgCl2��aq��$\stackrel{��}{��}$MgCl2��s��$\stackrel{���}{��}$Mg | |
| D�� | 1mol•L-1HCl��aq��$\stackrel{MnO_{2}/��}{��}$Cl2$\stackrel{ʯ����}{��}$Ca��ClO��2 |
| A�� | CH3COOH�����ԣ�������ͪ������û������ | |
| B�� | �ױ�ʹ���Ը��������ɫ����������ʹ����ɫ | |
| C�� | �����������ӷ�����ɫ��Ӧ���������� | |
| D�� | ��ȩ��HCN�����ӳɷ�Ӧ����ϩ������֮�ӳ� |
| ��ѧ���� | Ӧ�� | |
| A | O�������л�ԭ�� | ���ۿ�����ʳƷ�е������� |
| B | ������NaOH����Al��OH��3 | ����������ˮ�� |
| C | Fe�Ľ����Ա�Cu���� | FeCl3Ũ��Һ��ʴCu����ӡˢ��·�� |
| D | NaHCO3ˮ���Լ��� | ���ͷ��к���NaHCO3 |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | �÷�Ӧ����������ClO- | |
| B�� | �÷�Ӧ���������������� | |
| C�� | �μӷ�Ӧ���������뻹ԭ�������ʵ���֮��Ϊ2��3 | |
| D�� | �÷�Ӧ����1mol��ԭ��ʱ��ת�Ƶĵ���Ϊ3 mol |
| A�� | 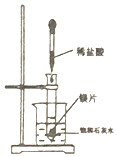 ����ͼװ�ã������о�Ca��OH��2���ܽ�����¶ȵı仯 | |
| B�� | 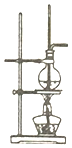 ����ͼװ�ã����������Ҵ��Ʊ���ϩ | |
| C�� |  ����ͼװ�ã�����������ʯ�Һ�NH4C1������NH3 | |
| D�� | 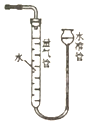 ����ͼװ�ã����ԲⶨŨ������ͭ��Ӧ���ɵ�NO2������� |
 Ԫ�����ڱ��еڢ�A��Ԫ�صĵ��ʼ��仯�������;�㷺��
Ԫ�����ڱ��еڢ�A��Ԫ�صĵ��ʼ��仯�������;�㷺��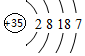 �������Ϻ�ɫ������ɫ���������ʣ���2�����ݵ���ʵ�������������ǻ���ʯ[Ca5��PO4��3OH]�ܽ�Ľ������մ���������ϣ���ø�������²����������ʣ������ȣ�ݣ�ʹ�ú�������ᱣ�����ݣ���Ϊ�����ӻ���ǻ���ʯ[Ca5��PO4��3OH]������Ӧ���ɷ���ʯ������ʯ���ǻ���ʯ���ֿܵ������ʴ���������ӷ���ʽ��ʾ��һ����Ca5��PO4��3OH��S��+F- ��aq��?Ca5��PO4��3 F��S��+OH-��aq������������ӻ������ƿ�ǻϸ�������ᣬ���������Ѿ��ձ�ʹ�ã�
�������Ϻ�ɫ������ɫ���������ʣ���2�����ݵ���ʵ�������������ǻ���ʯ[Ca5��PO4��3OH]�ܽ�Ľ������մ���������ϣ���ø�������²����������ʣ������ȣ�ݣ�ʹ�ú�������ᱣ�����ݣ���Ϊ�����ӻ���ǻ���ʯ[Ca5��PO4��3OH]������Ӧ���ɷ���ʯ������ʯ���ǻ���ʯ���ֿܵ������ʴ���������ӷ���ʽ��ʾ��һ����Ca5��PO4��3OH��S��+F- ��aq��?Ca5��PO4��3 F��S��+OH-��aq������������ӻ������ƿ�ǻϸ�������ᣬ���������Ѿ��ձ�ʹ�ã�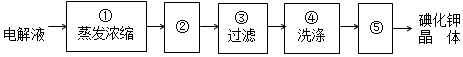
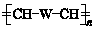 ��W�������еIJ��ֽṹ����һ����Ҫ�Ĺ��ܸ߷��Ӳ��ϣ��ۺ���X�����е�һ�֣�ת��·������
��W�������еIJ��ֽṹ����һ����Ҫ�Ĺ��ܸ߷��Ӳ��ϣ��ۺ���X�����е�һ�֣�ת��·������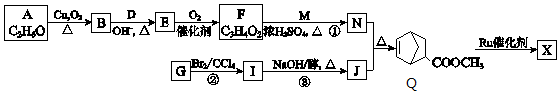
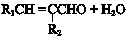
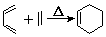
 +2NaOH$��_{��}^{��}$
+2NaOH$��_{��}^{��}$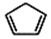 +2 NaBr+2 H2O
+2 NaBr+2 H2O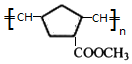
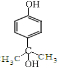 ��
��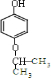 ����д������һ�ּ��ɣ�
����д������һ�ּ��ɣ�