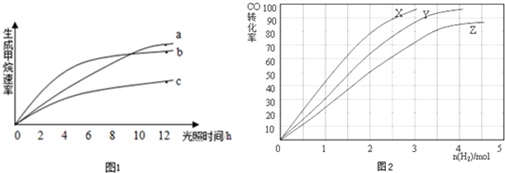
��Ŀ����
14�� һ���¶��£���2L�ܱ������м�������Cu2O��ͨ��10.0molˮ������������Ӧ��2H2O��g��?2H2��g��+O2��g����H=+484kJ/mol��T1�¶��²�ͬʱ�β���O2������������
һ���¶��£���2L�ܱ������м�������Cu2O��ͨ��10.0molˮ������������Ӧ��2H2O��g��?2H2��g��+O2��g����H=+484kJ/mol��T1�¶��²�ͬʱ�β���O2������������| ʱ��/min | 20 | 40 | 60 | 80 |
| n��O2��/mol | 1.0 | 1.6 | 2.0 | 2.0 |
��2��T1�¶��£��÷�Ӧ��ƽ�ⳣ������ʽΪ��K=$\frac{{c}^{2}��{H}_{2}����c��{O}_{2}��}{{c}^{2}��{H}_{2}O��}$
��3����T2�¶�ʱ��K=0.4��T1�� T2���������������=����
��4��T1�¶��£�t1ʱ�̴ﵽƽ��ʱ��ͼ�����Ϊ������ͼI��
����ͼI�л���������T0��t0min�ﵽƽ��ʱ�Ĵ���ͼ������Ҫ�ļ�ע��
��5��H2��������ȼ�ϵ�صķ�Ӧ���ʣ���д�������Խ����У�����ȼ�ϵ�صĸ�����Ӧʽ��H2-2e-=2H+��
���� ��1�����ݻ�ѧ��Ӧ����֮�ȵ��ڼ�����֮�ȼ��㷴Ӧ���ʣ�
��2����ƽ��ʱ��K=$\frac{����������ƽ��Ũ��ϵ������֮��}{������Ӧ��ƽ��Ũ��ϵ������֮��}$��
��3����������ʽ����ƽ�ⳣ���������¶Ⱥ�ƽ�ⳣ��֮��Ĺ�ϵ���ش�
��4���������ȵķ�Ӧ�������¶ȣ�ƽ�������ƶ������Ƿ�Ӧ���ʻ������
��5������ȼ�����Ե���У�����������ʧ�������������ӣ�
��� �⣺��1���ɱ������ݿ�֪ǰ20minʱv��O2��=$\frac{0.1mol}{2L��20min}$=2.5��10-2 mol��L-1��min -1����v��H2O��=2v��O2��=5.0��10-2 mol��L-1��min -1���ʴ�Ϊ��5.0��10-2 mol��L-1��min -1��
��2���÷�Ӧ��ƽ�ⳣ������ʽK=$\frac{{c}^{2}��{H}_{2}����c��{O}_{2}��}{{c}^{2}��{H}_{2}O��}$���ʴ�Ϊ��K=$\frac{{c}^{2}��{H}_{2}����c��{O}_{2}��}{{c}^{2}��{H}_{2}O��}$��
��3��2H2O��g��?2H2��g��+O2��g��
��ʼŨ�ȣ�5 0 0
�仯Ũ�ȣ�2 2 1
ƽ��Ũ�ȣ�3 2 1
������µ�ƽ�ⳣ��K=$\frac{1��{2}^{2}}{{3}^{2}}$=$\frac{4}{9}$����T2�¶�ʱ��K=0.4����Ӧ�����ȵģ�����T1��T2���ʴ�Ϊ��T1��T2��
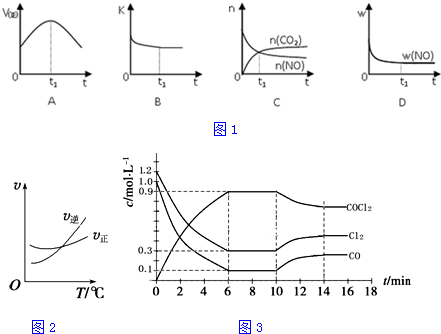
��4��2H2O��g��?2H2��g��+O2��g�������ȵķ�Ӧ�������¶ȣ�ƽ�������ƶ������������ʵ����ή�ͣ����Ƿ�Ӧ���ʻ������ͼ��Ϊ��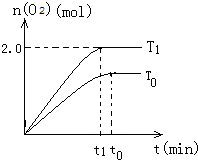 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��5������ȼ�����Ե���У�����������ʧ�������������ӣ�������ӦʽΪ��2H2-4e-�T4H+���ʴ�Ϊ��H2-2e-=2H+��
���� �����ۺϿ���ѧ�����ʺ�ƽ��ļ��㡢ƽ���ƶ�ԭ����Ӧ���Լ�ԭ��ع���ԭ����Ӧ��֪ʶ�������ۺ�֪ʶ�Ŀ��飬�Ѷ��еȣ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | 200mL2mol/LMgCl2��Һ | B�� | 1000mL2.5mol/LNaCl��Һ | ||
| C�� | 300mL5mol/LFeCl3��Һ | D�� | 250mL lmol/L AlCl3��Һ |
��1�����������������ڴ����еĺ������������ʱ�������������ȣ��漰���·�Ӧ��
��4NO2��g��+2NaCl��s��?2NaNO3��s��+2NO��g��+Cl2��g�� K1
��2NO2��g��+NaCl��s��?NaNO3��s��+ClNO��g�� K2
��2NO��g��+Cl2��g��?2ClNO��g�� K3
��K1��K2��K3֮��Ĺ�ϵΪK3=$\frac{{{K}_{2}}^{2}}{{K}_{1}}$��
��2��T��ʱ��2NO��g��+Cl2��g��?2ClNO��g��������Ӧ���ʱ���ʽΪv��=k cn��ClNO����������ʺ�Ũ�ȵĹ�ϵ�����
| ��� | c��ClNO��/mol•L-1 | v/mol•L-1•s-1 |
| �� | 0.30 | 3.6��10-8 |
| �� | 0.60 | 1.44��10-7 |
| �� | 0.90 | 3.24��10-7 |
��3����2L�ĺ����ܱ������г���4molNO��g����2molCl2��g�����ڲ�ͬ�¶��²��c��ClNO����ʱ��Ĺ�ϵ��ͼA��
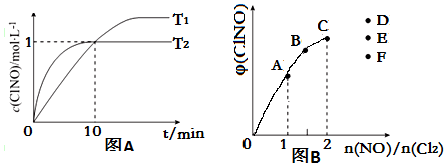
���¶�ΪT1ʱ������Ϊ�÷�Ӧ�ﵽƽ��ı�־����bdf��
a������������ֲ��� b������ѹǿ���ֲ��� c��ƽ�ⳣ��K���ֲ���
d��������ɫ���ֲ��� e��v��ClNO��=v��NO�� f��NO�� ClNO�����ʵ�����ֵ���ֲ���
�ڷ�Ӧ��ʼ��10minʱCl2��ƽ����Ӧ����v��Cl2��=0.05mol•L-1•min-1��
���¶�ΪT2ʱ��10min�Ѿ��ﵽƽ�⣬�÷�Ӧ��ƽ�ⳣ��K=2L/mol��ע����λ����
��4��һ���������ں��º��ݵ��ܱ������а�һ����������NO��g����Cl2��g����ƽ��ʱClNO�������������$\frac{n��NO��}{n��C{l}_{2}��}$�ı仯ͼ����ͼB����A��B��C��״̬�У�NO��ת������С����C�㣬��n��NO��/n��Cl2��=2.8ʱ���ﵽƽ��״̬ClNO����������տ�����D��E��F�����е�F�㣮
 �״���һ�ֿ�������Դ�����й㷺�Ŀ�����Ӧ��ǰ������ҵ��һ��ɲ������·�Ӧ���ϳɼ״���CO��g��+2H2��g��?CH3OH��g����H=-a kJ/mol�� ��T��ʱ����һ������̶�Ϊ1L���ܱ������м���1mol CO��2mol H2����Ӧ�ﵽƽ��ʱ�������ڵ�ѹǿ�ǿ�ʼʱ��3/5��
�״���һ�ֿ�������Դ�����й㷺�Ŀ�����Ӧ��ǰ������ҵ��һ��ɲ������·�Ӧ���ϳɼ״���CO��g��+2H2��g��?CH3OH��g����H=-a kJ/mol�� ��T��ʱ����һ������̶�Ϊ1L���ܱ������м���1mol CO��2mol H2����Ӧ�ﵽƽ��ʱ�������ڵ�ѹǿ�ǿ�ʼʱ��3/5��